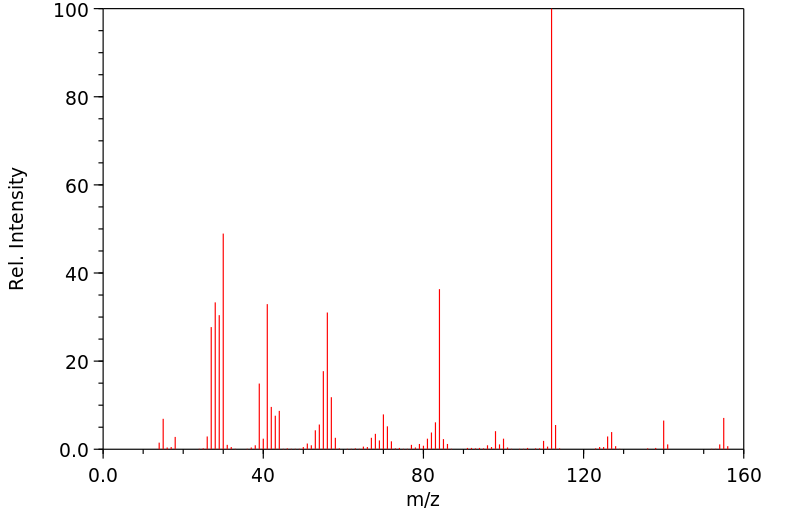
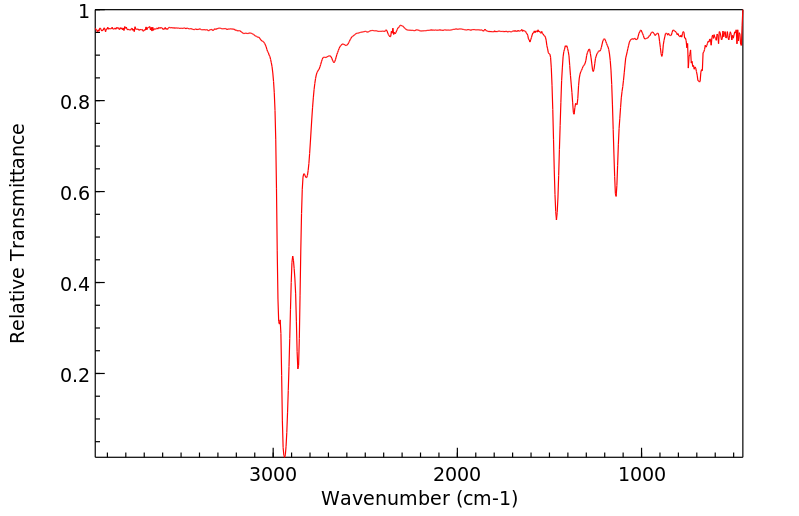
毒理性
丁环己胺对兔眼刺激性评分为9。/在兔眼外部进行测试,根据24小时后观察到的伤害程度,按照1到10的等级进行数值评分,特别关注角膜的状况。最严重的伤害评为10分。
N-BUTYLCYCLOHEXYLAMINE RATED 9 ON RABBIT EYES. /TESTED EXTERNALLY ON EYES OF RABBITS, RATED NUMERICALLY ON SCALE OF 1 TO 10 ACCORDING TO DEGREE OF INJURY OBSERVED AFTER 24 HR, PAYING PARTICULAR ATTENTION TO CONDITION OF CORNEA. MOST SEVERE INJURIES HAVE BEEN RATED 10/.
来源:Hazardous Substances Data Bank (HSDB)