2,5-二甲基苯甲醇 | 53957-33-8
中文名称
2,5-二甲基苯甲醇
中文别名
2,5-二甲基苄醇;2,5-二甲基苯甲基醇
英文名称
2,5-dimethylbenzyl alcohol
英文别名
2,5-Dimethyl-benzylalkohol;(2,5-dimethylphenyl)methanol;2,5-Dimethyl-hydroxymethyl benzene
CAS
53957-33-8
化学式
C9H12O
mdl
——
分子量
136.194
InChiKey
LEBQTCCCNMTXSF-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:40-42°C
-
沸点:232-234 °C
-
密度:1,011 g/cm3
-
闪点:107 °C
-
溶解度:很难混溶。
-
稳定性/保质期:
遵照规定使用和储存,则不会发生分解。
计算性质
-
辛醇/水分配系数(LogP):1.8
-
重原子数:10
-
可旋转键数:1
-
环数:1.0
-
sp3杂化的碳原子比例:0.33
-
拓扑面积:20.2
-
氢给体数:1
-
氢受体数:1
安全信息
-
安全说明:S24/25
-
海关编码:2906299090
-
危险性防范说明:P261,P305+P351+P338
-
危险性描述:H302,H315,H319,H335
-
储存条件:存于阴凉干燥处
SDS
| Name: | 2 5-Dimethylbenzyl alcohol 98% Material Safety Data Sheet |
| Synonym: | |
| CAS: | 53957-33-8 |
Synonym:
Section 2 - COMPOSITION, INFORMATION ON INGREDIENTS
| CAS# | Chemical Name | content | EINECS# |
| 53957-33-8 | 2,5-Dimethylbenzyl alcohol | 98 | 258-888-2 |
Risk Phrases: None Listed.
Section 3 - HAZARDS IDENTIFICATION
EMERGENCY OVERVIEW
The toxicological properties of this material have not been fully investigated.
Potential Health Effects
Eye:
May cause eye irritation.
Skin:
May cause skin irritation.
Ingestion:
May cause irritation of the digestive tract. The toxicological properties of this substance have not been fully investigated.
Inhalation:
May cause respiratory tract irritation. The toxicological properties of this substance have not been fully investigated.
Chronic:
No information found.
Section 4 - FIRST AID MEASURES
Eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid immediately.
Skin:
Get medical aid. Flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes. Wash clothing before reuse.
Ingestion:
If victim is conscious and alert, give 2-4 cupfuls of milk or water.
Never give anything by mouth to an unconscious person. Get medical aid immediately.
Inhalation:
Remove from exposure and move to fresh air immediately. If not breathing, give artificial respiration. If breathing is difficult, give oxygen. Get medical aid.
Notes to Physician:
Section 5 - FIRE FIGHTING MEASURES
General Information:
As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear. During a fire, irritating and highly toxic gases may be generated by thermal decomposition or combustion.
Extinguishing Media:
Use agent most appropriate to extinguish fire. Use water spray, dry chemical, carbon dioxide, or appropriate foam.
Section 6 - ACCIDENTAL RELEASE MEASURES
General Information: Use proper personal protective equipment as indicated in Section 8.
Spills/Leaks:
Absorb spill with inert material (e.g. vermiculite, sand or earth), then place in suitable container. Sweep up or absorb material, then place into a suitable clean, dry, closed container for disposal.
Provide ventilation.
Section 7 - HANDLING and STORAGE
Handling:
Wash thoroughly after handling. Remove contaminated clothing and wash before reuse. Use with adequate ventilation. Avoid contact with eyes, skin, and clothing. Keep container tightly closed. Avoid ingestion and inhalation.
Storage:
Store in a tightly closed container. Store in a cool, dry, well-ventilated area away from incompatible substances.
Section 8 - EXPOSURE CONTROLS, PERSONAL PROTECTION
Engineering Controls:
Facilities storing or utilizing this material should be equipped with an eyewash facility and a safety shower. Use adequate ventilation to keep airborne concentrations low.
Exposure Limits CAS# 53957-33-8: Personal Protective Equipment Eyes: Wear appropriate protective eyeglasses or chemical safety goggles as described by OSHA's eye and face protection regulations in 29 CFR 1910.133 or European Standard EN166.
Skin:
Wear appropriate protective gloves to prevent skin exposure.
Clothing:
Wear appropriate protective clothing to prevent skin exposure.
Respirators:
A respiratory protection program that meets OSHA's 29 CFR 1910.134 and ANSI Z88.2 requirements or European Standard EN 149 must be followed whenever workplace conditions warrant respirator use.
Section 9 - PHYSICAL AND CHEMICAL PROPERTIES
Physical State: Liquid
Color: colorless
Odor: Not available.
pH: Not available.
Vapor Pressure: Not available.
Viscosity: Not available.
Boiling Point: 232.0 - 234.0 deg C @ 760.00m
Freezing/Melting Point: Not available.
Autoignition Temperature: Not available.
Flash Point: 107 deg C ( 224.60 deg F)
Explosion Limits, lower: Not available.
Explosion Limits, upper: Not available.
Decomposition Temperature:
Solubility in water:
Specific Gravity/Density:
Molecular Formula: C9H12O
Molecular Weight: 136.19
Section 10 - STABILITY AND REACTIVITY
Chemical Stability:
Stable under normal temperatures and pressures.
Conditions to Avoid:
Incompatible materials, strong oxidants.
Incompatibilities with Other Materials:
Acids, acid chlorides, acid anhydrides, strong oxidizing agents.
Hazardous Decomposition Products:
Carbon monoxide, carbon dioxide.
Hazardous Polymerization: Has not been reported
Section 11 - TOXICOLOGICAL INFORMATION
RTECS#:
CAS# 53957-33-8 unlisted.
LD50/LC50:
Not available.
Carcinogenicity:
2,5-Dimethylbenzyl alcohol - Not listed by ACGIH, IARC, or NTP.
Section 12 - ECOLOGICAL INFORMATION
Section 13 - DISPOSAL CONSIDERATIONS
Dispose of in a manner consistent with federal, state, and local regulations.
Section 14 - TRANSPORT INFORMATION
IATA
Not regulated as a hazardous material.
IMO
Not regulated as a hazardous material.
RID/ADR
Not regulated as a hazardous material.
Section 15 - REGULATORY INFORMATION
European/International Regulations
European Labeling in Accordance with EC Directives
Hazard Symbols: Not available.
Risk Phrases:
Safety Phrases:
S 24/25 Avoid contact with skin and eyes.
S 28A After contact with skin, wash immediately with
plenty of water.
S 37 Wear suitable gloves.
S 45 In case of accident or if you feel unwell, seek
medical advice immediately (show the label where
possible).
WGK (Water Danger/Protection)
CAS# 53957-33-8: No information available.
Canada
None of the chemicals in this product are listed on the DSL/NDSL list.
CAS# 53957-33-8 is not listed on Canada's Ingredient Disclosure List.
US FEDERAL
TSCA
CAS# 53957-33-8 is not listed on the TSCA inventory.
It is for research and development use only.
SECTION 16 - ADDITIONAL INFORMATION
N/A
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 2,5-二甲基苯甲酸 2,5-dimethylbenzoic acid 610-72-0 C9H10O2 150.177 1,2,4-三甲基苯 1,2,4-Trimethylbenzene 95-63-6 C9H12 120.194 —— (2.5-Dimethyl-benzyl)-acetat 22184-23-2 C11H14O2 178.231 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 (1,3-二氢异苯并呋喃-5-基)甲醇 (1,3-dihydroisobenzofuran-5-yl)methanol 89424-84-0 C9H10O2 150.177 2,5-二甲基苯甲酸 2,5-dimethylbenzoic acid 610-72-0 C9H10O2 150.177 —— 2,5-dimethylbenzyl chloroformate 138556-74-8 C10H11ClO2 198.649 —— (2.5-Dimethyl-benzyl)-acetat 22184-23-2 C11H14O2 178.231 1,3-二氢-2-苯并呋喃-5-甲醛 1,3-dihydroisobenzofuran-5-carbaldehyde 89424-83-9 C9H8O2 148.161 —— 5-(bromomethyl)-1,3-dihydroisobenzofuran 188111-48-0 C9H9BrO 213.074 1,3-二氢-2-苯并呋喃-5-基甲基乙酸酯 1,3-dihydro-2-benzofuran-5-ylmethyl acetate 1187196-72-0 C11H12O3 192.214
反应信息
-
作为反应物:描述:2,5-二甲基苯甲醇 在 sodium peroxide 作用下, 以 乙醚 、 氯仿 、 水 为溶剂, 反应 4.5h, 生成 di(2,5-dimethylbenzyl) peroxydicarbonate参考文献:名称:Etlis, I.V.; Fomin, V.A.; Nozrina, F.D., Journal of Organic Chemistry USSR (English Translation), 1991, vol. 27, # 11.1, p. 2010 - 2015摘要:DOI:
-
作为产物:描述:2,5-dimethyl-benzoic acid-(N-methyl-anilide) 在 四氢呋喃 、 lithium aluminium tetrahydride 作用下, 生成 2,5-二甲基苯甲醇参考文献:名称:Weygand; Mitgau, Chemische Berichte, 1955, vol. 88, p. 301,307摘要:DOI:
文献信息
-
Aluminum Monohydride Catalyzed Selective Hydroboration of Carbonyl Compounds作者:Vineet Kumar Jakhar、Milan Kr. Barman、Sharanappa NembennaDOI:10.1021/acs.orglett.6b02310日期:2016.9.16The well-defined aluminum monohydride compound [(2,4,6-Me3-C6H2)NC(Me)}2(Me)(H)]AlH·(NMe2Et) (1) catalyzes hydroboration of a wide range of aldehydes and ketones under mild reaction conditions. Moreover, compound 1 displayed chemoselective hydroboration of aldehydes over ketones at rt.
-
Method of treating cancer申请人:——公开号:US20030220241A1公开(公告)日:2003-11-27The present invention relates to methods of treating cancer using a combination of a compound which is a PSA conjugate and a compound which is a inhibitor of prenyl-protein transferase, which methods comprise administering to said mammal, either sequentially in any order or simultaneously, amounts of at least two therapeutic agents selected from a group consisting of a compound which is a PSA conjugate and a compound which is a inhibitor of prenyl-protein transferase. The invention also relates to methods of preparing such compositions.
-
SUBSTITUTED QUINAZOLINES AS FUNGICIDES申请人:Quaranta Laura公开号:US20120129875A1公开(公告)日:2012-05-24The present invention relates to a compound of formula (I) wherein the substituents have the definitions as defined in claim 1 or a salt or a N-oxide thereof, their use and methods for the control and/or prevention of microbial infection, particularly fungal infection, in plants and to processes for the preparation of these compounds.本发明涉及一种具有如权利要求1中定义的取代基定义的化合物的公式(I),或其盐或N-氧化物,它们的用途以及用于控制和/或预防植物中微生物感染,特别是真菌感染的方法,以及制备这些化合物的方法。
-
[EN] COMPOUNDS USEFUL AS KINASE INHIBITORS<br/>[FR] COMPOSÉS UTILISÉS COMME INHIBITEURS DE KINASE申请人:REDX PHARMA PLC公开号:WO2017103611A1公开(公告)日:2017-06-22This invention relates to novel compounds. The compounds of the invention are tyrosine kinase inhibitors. Specifically, the compounds of the invention are useful as inhibitors of Bruton's tyrosine kinase (BTK).The invention also contemplates the use of the compounds for treating conditions treatable by the inhibition of Bruton's tyrosine kinase, for example cancer, lymphoma, leukemia and immunological diseases.
-
[EN] TRIAZOLONE COMPOUNDS AS PERK INHIBITORS<br/>[FR] COMPOSÉS DE TRIAZOLONE COMME INHIBITEURS DE PERK申请人:GLAXOSMITHKLINE INTELLECTUAL PROPERTY (NO 2) LTD公开号:WO2017046738A1公开(公告)日:2017-03-23The invention is directed to substituted triazolidinone derivatives. Specifically, the invention is directed to compounds according to Formula I: (I) wherein R1, R2, R3, R4, R5, X, Y, Y1, and Z are as defined herein. The compounds of the invention are inhibitors of PERK and can be useful in the treatment of cancer, pre-cancerous syndromes, as Alzheimer's disease, neuropathic pain, spinal cord injury, traumatic brain injury, ischemic stroke, stroke, Parkinson disease, diabetes, metabolic syndrome, metabolic disorders, Huntington's disease, Creutzfeldt-Jakob Disease, fatal familial insomnia, Gerstmann-Sträussler-Scheinker syndrome, and related prion diseases, amyotrophic lateral sclerosis, progressive supranuclear palsy, myocardial infarction, cardiovascular disease, inflammation, organ fibrosis, chronic and acute diseases of the liver, fatty liver disease, liver steatosis, liver fibrosis, chronic and acute diseases of the lung, lung fibrosis, chronic and acute diseases of the kidney, kidney fibrosis, chronic traumatic encephalopathy (CTE), neurodegeneration, dementias, frontotemporal dementias, tauopathies, Pick's disease, Neimann-Pick's disease, amyloidosis, cognitive impairment, atherosclerosis, ocular diseases, arrhythmias, in organ transplantation and in the transportation of organs for transplantation. Accordingly, the invention is further directed to pharmaceutical compositions comprising a compound of the invention. The invention is still further directed to methods of inhibiting PERK activity and treatment of disorders associated therewith using a compound of the invention or a pharmaceutical composition comprising a compound of the invention.这项发明涉及取代三唑烷酮衍生物。具体而言,该发明涉及符合以下式(I)的化合物:其中R1、R2、R3、R4、R5、X、Y、Y1和Z如本文所定义。该发明的化合物是PERK的抑制剂,可用于治疗癌症、癌前综合征、阿尔茨海默病、神经病痛、脊髓损伤、创伤性脑损伤、缺血性中风、中风、帕金森病、糖尿病、代谢综合征、代谢紊乱、亨廷顿病、克雅氏病、致命性家族性失眠、格斯特曼-施特劳斯勒-谢因克症候群及相关朊蛋白病、肌萎缩侧索硬化、进行性核上性麻痹、心肌梗死、心血管疾病、炎症、器官纤维化、肝脏慢性和急性疾病、脂肪肝病、肝脂肪变性、肝纤维化、肺部慢性和急性疾病、肺纤维化、肾脏慢性和急性疾病、肾脏纤维化、慢性创伤性脑病(CTE)、神经退行性疾病、痴呆症、额颞叶痴呆症、tau蛋白病、皮克氏病、尼曼-皮克氏病、淀粉样变性、认知障碍、动脉粥样硬化、眼部疾病、心律失常、器官移植以及器官移植用途中的运输。因此,该发明进一步涉及包含该发明化合物的药物组合物。该发明还进一步涉及使用该发明化合物或包含该发明化合物的药物组合物抑制PERK活性和治疗相关疾病的方法。
表征谱图
-
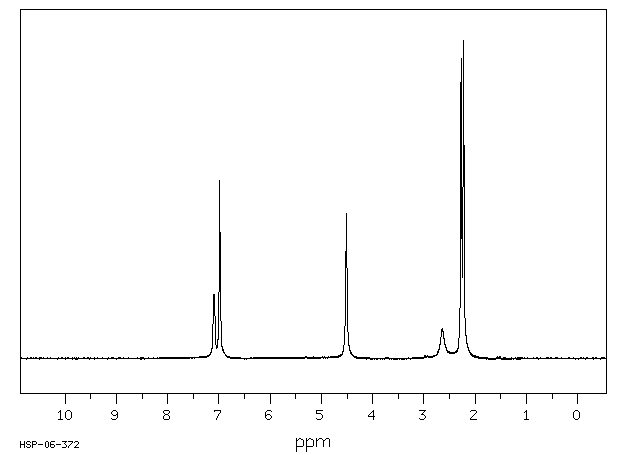
氢谱1HNMR
-
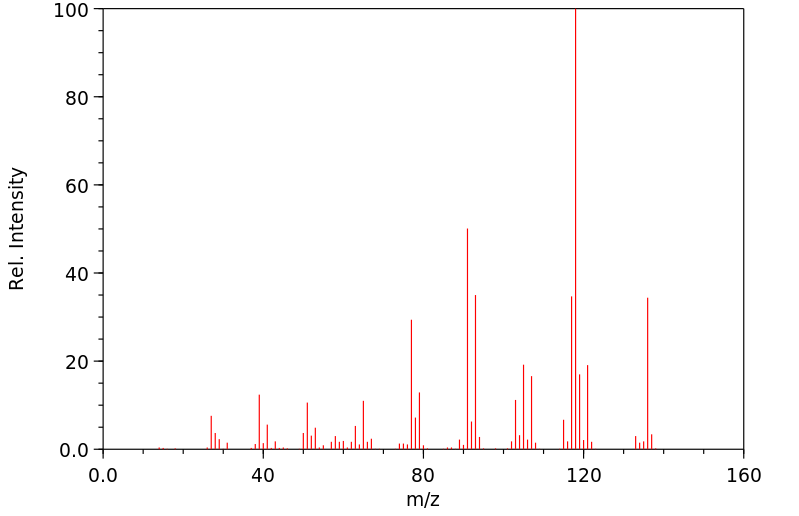
质谱MS
-
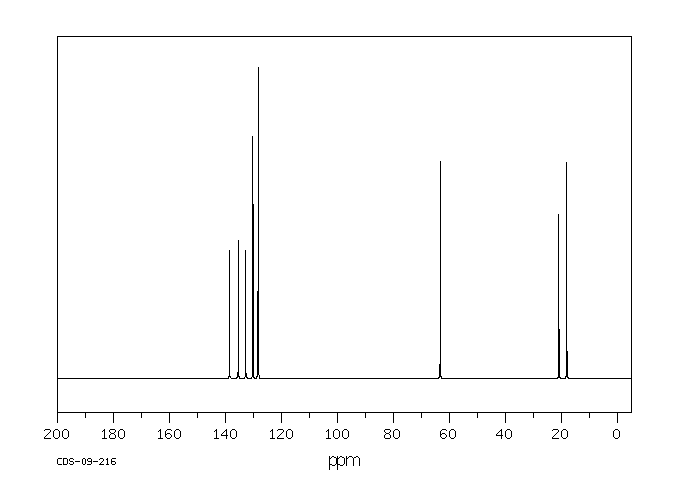
碳谱13CNMR
-
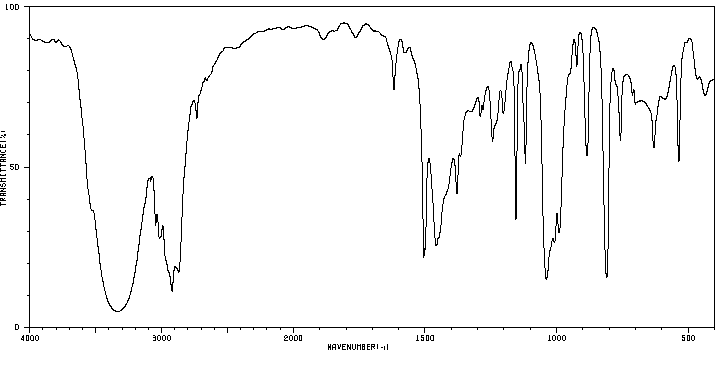
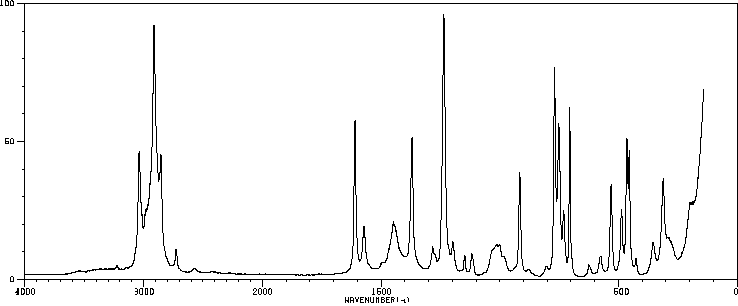
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(βS)-β-氨基-4-(4-羟基苯氧基)-3,5-二碘苯甲丙醇
(S,S)-邻甲苯基-DIPAMP
(S)-(-)-7'-〔4(S)-(苄基)恶唑-2-基]-7-二(3,5-二-叔丁基苯基)膦基-2,2',3,3'-四氢-1,1-螺二氢茚
(S)-盐酸沙丁胺醇
(S)-3-(叔丁基)-4-(2,6-二甲氧基苯基)-2,3-二氢苯并[d][1,3]氧磷杂环戊二烯
(S)-2,2'-双[双(3,5-三氟甲基苯基)膦基]-4,4',6,6'-四甲氧基联苯
(S)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(R)富马酸托特罗定
(R)-(-)-盐酸尼古地平
(R)-(-)-4,12-双(二苯基膦基)[2.2]对环芳烷(1,5环辛二烯)铑(I)四氟硼酸盐
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[((6-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(4-叔丁基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(3-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-4,7-双(3,5-二-叔丁基苯基)膦基-7“-[(吡啶-2-基甲基)氨基]-2,2”,3,3'-四氢1,1'-螺二茚满
(R)-3-(叔丁基)-4-(2,6-二苯氧基苯基)-2,3-二氢苯并[d][1,3]氧杂磷杂环戊烯
(R)-2-[((二苯基膦基)甲基]吡咯烷
(R)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(N-(4-甲氧基苯基)-N-甲基-3-(1-哌啶基)丙-2-烯酰胺)
(5-溴-2-羟基苯基)-4-氯苯甲酮
(5-溴-2-氯苯基)(4-羟基苯基)甲酮
(5-氧代-3-苯基-2,5-二氢-1,2,3,4-oxatriazol-3-鎓)
(4S,5R)-4-甲基-5-苯基-1,2,3-氧代噻唑烷-2,2-二氧化物-3-羧酸叔丁酯
(4S,4''S)-2,2''-亚环戊基双[4,5-二氢-4-(苯甲基)恶唑]
(4-溴苯基)-[2-氟-4-[6-[甲基(丙-2-烯基)氨基]己氧基]苯基]甲酮
(4-丁氧基苯甲基)三苯基溴化磷
(3aR,8aR)-(-)-4,4,8,8-四(3,5-二甲基苯基)四氢-2,2-二甲基-6-苯基-1,3-二氧戊环[4,5-e]二恶唑磷
(3aR,6aS)-5-氧代六氢环戊基[c]吡咯-2(1H)-羧酸酯
(2Z)-3-[[(4-氯苯基)氨基]-2-氰基丙烯酸乙酯
(2S,3S,5S)-5-(叔丁氧基甲酰氨基)-2-(N-5-噻唑基-甲氧羰基)氨基-1,6-二苯基-3-羟基己烷
(2S,2''S,3S,3''S)-3,3''-二叔丁基-4,4''-双(2,6-二甲氧基苯基)-2,2'',3,3''-四氢-2,2''-联苯并[d][1,3]氧杂磷杂戊环
(2S)-(-)-2-{[[[[3,5-双(氟代甲基)苯基]氨基]硫代甲基]氨基}-N-(二苯基甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[((1S,2S)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[[((1R,2R)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2-硝基苯基)磷酸三酰胺
(2,6-二氯苯基)乙酰氯
(2,3-二甲氧基-5-甲基苯基)硼酸
(1S,2S,3S,5S)-5-叠氮基-3-(苯基甲氧基)-2-[(苯基甲氧基)甲基]环戊醇
(1S,2S,3R,5R)-2-(苄氧基)甲基-6-氧杂双环[3.1.0]己-3-醇
(1-(4-氟苯基)环丙基)甲胺盐酸盐
(1-(3-溴苯基)环丁基)甲胺盐酸盐
(1-(2-氯苯基)环丁基)甲胺盐酸盐
(1-(2-氟苯基)环丙基)甲胺盐酸盐
(1-(2,6-二氟苯基)环丙基)甲胺盐酸盐
(-)-去甲基西布曲明
龙蒿油
龙胆酸钠
龙胆酸叔丁酯
龙胆酸
龙胆紫-d6
龙胆紫