1-苯基-1-氰基环戊烷 | 77-57-6
中文名称
1-苯基-1-氰基环戊烷
中文别名
1-苯基环戊烷甲腈;1-苯基环戊腈;1-苯基-1-环戊烷甲腈
英文名称
1-phenyl-1-cyclopentanecarbonitrile
英文别名
1-phenylcyclopentane-1-carbonitrile;1-Phenylcyclopentanecarbonitrile
CAS
77-57-6
化学式
C12H13N
mdl
——
分子量
171.242
InChiKey
GDXMFFGTPGAGGX-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
沸点:135-140 °C (10 mmHg)
-
密度:1.03
-
稳定性/保质期:
与强氧化剂反应。
计算性质
-
辛醇/水分配系数(LogP):3
-
重原子数:13
-
可旋转键数:1
-
环数:2.0
-
sp3杂化的碳原子比例:0.42
-
拓扑面积:23.8
-
氢给体数:0
-
氢受体数:1
安全信息
-
危险品标志:Xn
-
安全说明:S26,S36/37/39
-
危险类别码:R20/21/22,R36/37/38
-
海关编码:29269095
-
WGK Germany:3
-
危险类别:6.1
-
包装等级:III
-
危险品运输编号:3276
-
危险性防范说明:P261,P280,P305+P351+P338
-
危险性描述:H302,H312,H315,H319,H332,H335
-
储存条件:密封贮藏,存放在阴凉干燥的地方。
SDS
| Name: | 1-PHENYL-1-CYCLOPENTANECARBONITRILE Material Safety Data Sheet |
| Synonym: | |
| CAS: | 77-57-6 |
Synonym:
Section 2 - COMPOSITION, INFORMATION ON INGREDIENTS
| CAS# | Chemical Name | content | EINECS# |
| 77-57-6 | 1-PHENYL-1-CYCLOPENTANECARBONITRILE | 201-038-2 |
Risk Phrases: 20/21/22
Section 3 - HAZARDS IDENTIFICATION
EMERGENCY OVERVIEW
Harmful by inhalation, in contact with skin and if swallowed.
Potential Health Effects
The toxicological properties of this material have not been investigated. Use appropriate procedures to prevent opportunities for direct contact with the skin or eyes and to prevent inhalation.
Section 4 - FIRST AID MEASURES
Eyes: Immediately flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids.
Skin:
Flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes.
Ingestion:
Do NOT induce vomiting. If conscious and alert, rinse mouth and drink 2-4 cupfuls of milk or water.
Inhalation:
Remove from exposure and move to fresh air immediately.
Notes to Physician:
Section 5 - FIRE FIGHTING MEASURES
General Information:
As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear.
Extinguishing Media:
In case of fire, use water, dry chemical, chemical foam, or alcohol-resistant foam.
Section 6 - ACCIDENTAL RELEASE MEASURES
General Information: Use proper personal protective equipment as indicated in Section 8.
Spills/Leaks:
Absorb spill with inert material (e.g. vermiculite, sand or earth), then place in suitable container.
Section 7 - HANDLING and STORAGE
Handling:
Not available.
Storage:
Not available.
Section 8 - EXPOSURE CONTROLS, PERSONAL PROTECTION
Engineering Controls:
Use adequate general or local exhaust ventilation to keep airborne concentrations below the permissible exposure limits.
Exposure Limits CAS# 77-57-6: Personal Protective Equipment Eyes: Not available.
Skin:
Wear appropriate protective gloves and clothing to prevent skin exposure.
Clothing:
Wear appropriate protective clothing to minimize contact with skin.
Respirators:
Wear a NIOSH/MSHA or European Standard EN 149 approved full-facepiece airline respirator in the positive pressure mode with emergency escape provisions.
Section 9 - PHYSICAL AND CHEMICAL PROPERTIES
Physical State: Not available.
Color: Not available.
Odor: None reported.
pH: Not available.
Vapor Pressure: Not available.
Viscosity: Not available.
Boiling Point: 135.0 - 142.0 deg C @ 10.00mmHg
Freezing/Melting Point: 0 deg C
Autoignition Temperature: Not available.
Flash Point: > 110 deg C (> 230.00 deg F)
Explosion Limits, lower: Not available.
Explosion Limits, upper: Not available.
Decomposition Temperature:
Solubility in water:
Specific Gravity/Density:
Molecular Formula: C12H13N
Molecular Weight: 171.24
Section 10 - STABILITY AND REACTIVITY
Chemical Stability:
Stable under normal temperatures and pressures.
Conditions to Avoid:
Not available.
Incompatibilities with Other Materials:
Strong oxidizing agents - strong bases.
Hazardous Decomposition Products:
Not available.
Hazardous Polymerization: Has not been reported.
Section 11 - TOXICOLOGICAL INFORMATION
RTECS#:
CAS# 77-57-6 unlisted.
LD50/LC50:
Not available.
Carcinogenicity:
1-PHENYL-1-CYCLOPENTANECARBONITRILE - Not listed by ACGIH, IARC, or NTP.
Section 12 - ECOLOGICAL INFORMATION
Section 13 - DISPOSAL CONSIDERATIONS
Dispose of in a manner consistent with federal, state, and local regulations.
Section 14 - TRANSPORT INFORMATION
IATA
Shipping Name: TOXIC LIQUID, ORGANIC, N.O.S.*
Hazard Class: 6.1
UN Number: 2810
Packing Group: III
IMO
Shipping Name: TOXIC LIQUID, ORGANIC, N.O.S.
Hazard Class: 6.1
UN Number: 2810
Packing Group: III
RID/ADR
Shipping Name: TOXIC LIQUID, ORGANIC, N.O.S.
Hazard Class: 6.1
UN Number: 2810
Packing group: III
Section 15 - REGULATORY INFORMATION
European/International Regulations
European Labeling in Accordance with EC Directives
Hazard Symbols: XN
Risk Phrases:
R 20/21/22 Harmful by inhalation, in contact with
skin and if swallowed.
Safety Phrases:
S 24/25 Avoid contact with skin and eyes.
WGK (Water Danger/Protection)
CAS# 77-57-6: No information available.
Canada
None of the chemicals in this product are listed on the DSL/NDSL list.
CAS# 77-57-6 is not listed on Canada's Ingredient Disclosure List.
US FEDERAL
TSCA
CAS# 77-57-6 is not listed on the TSCA inventory.
It is for research and development use only.
SECTION 16 - ADDITIONAL INFORMATION
N/A
制备方法与用途
上下游信息
-
下游产品
中文名称 英文名称 CAS号 化学式 分子量 1-(4-氯苯基)-1-环戊烷甲腈 1-(4-chlorophenyl)cyclopentanecarbonitrile 64399-26-4 C12H12ClN 205.687 1-(4-氨基苯基)环戊甲腈 1-(4-aminophenyl)cyclopentane-1-carbonitrile 115279-73-7 C12H14N2 186.257 —— 3-oxo-1-phenylcyclopentane-1-carbonitrile 84409-26-7 C12H11NO 185.225 1-(4-硝基苯基)环戊烷甲腈 1-(4-nitrophenyl)cyclopentanecarbonitrile 91392-33-5 C12H12N2O2 216.239 2-(1-苯基环戊基)乙腈 β-phenyl-β,β-tetramethylenepropionitrile 5407-84-1 C13H15N 185.269 (1-乙炔基环戊基)苯 (1-ethynylcyclopentyl)benzene 1092352-06-1 C13H14 170.254 (1-苯基环戊基)甲胺 (1-phenylcyclopentyl)methylamine 17511-89-6 C12H17N 175.274 1-苯基环戊烷甲醛 1-phenyl-1-cyclopentanecarboxaldehyde 21573-69-3 C12H14O 174.243 —— 1-hydroxymethyl-1-phenylcyclopentane 59115-90-1 C12H16O 176.258 环戊基苯 cyclopentylbenzene 700-88-9 C11H14 146.232 —— 1-(1-phenyl-cyclopentyl)-ethylamine —— C13H19N 189.301 —— <1-Phenyl-cyclopentyl>-methyl-keton 4046-09-7 C13H16O 188.269 - 1
- 2
反应信息
-
作为反应物:描述:1-苯基-1-氰基环戊烷 在 sodium hydride 、 sodium iodide 作用下, 以 四氢呋喃 、 mineral oil 为溶剂, 反应 17.0h, 以61 mg的产率得到环戊基苯参考文献:名称:氢化物-碘化物复合材料的氢化物还原摘要:氢化钠(NaH)在化学合成中被广泛用作布朗斯台德碱,并与各种布朗斯台德酸发生反应,而它几乎不通过将氢化物传递给极性π亲电试剂来充当还原剂。这项研究提出了在LiI或NaI存在下,NaH使腈,酰胺和亚胺发生的一系列还原反应。这种非常简单的方案使NaH具有前所未有的独特的氢化物-给体化学反应性。DOI:10.1002/anie.201600305
-
作为产物:描述:参考文献:名称:Aminoesters of Substituted Alicylic Carboxylic Acids1摘要:DOI:10.1021/ja01203a077
文献信息
-
Inhibitors of acyl-CoA:cholesterol acyltransferase. 4. A novel series of urea ACAT inhibitors as potential hypocholesterolemic agents作者:Bharat K. Trivedi、Ann Holmes、Terri L. Stoeber、C. John Blankley、W. Howard Roark、Joseph A. Picard、Mary K. Shaw、Arnold D. Essenburg、Richard L. Stanfield、Brian R. KrauseDOI:10.1021/jm00074a011日期:1993.10We have synthesized a series of N-phenyl-N'-aralkyl and N-phenyl-N'-(1-phenylcycloalkyl)ureas as inhibitors of acyl-CoA:cholesterol acyltransferase (ACAT). This intracellular enzyme is thought to be responsible for the esterification of dietary cholesterol; hence inhibition of this enzyme could reduce diet-induced hypercholesterolemia. For this series of compounds, the in vitro ACAT inhibitory activity我们合成了一系列的N-苯基-N'-芳烷基和N-苯基-N'-(1-苯基环烷基)脲作为酰基辅酶A:胆固醇酰基转移酶(ACAT)的抑制剂。人们认为这种细胞内酶负责饮食中胆固醇的酯化。因此抑制这种酶可以减少饮食引起的高胆固醇血症。对于这一系列化合物,通过增加苯环上2,6-取代基的体积来提高体外ACAT抑制活性。此外,我们发现芳环的间距对于ACAT抑制活性至关重要。离必需的2,6-二异丙基苯基部分5个原子的苯环对于体外活性而言是最佳的。N'-苯基部分被α取代增强了体外效能。就苯基环烷基脲而言,ACAT抑制活性与环烷基环的大小无关。从该系列类似物中,发现在高胆固醇血症动物模型中以50 mg / kg的饮食施用时,具有出色的体外抑制ACAT效力的化合物25在体内可使血浆胆固醇降低73%。在此模型中,化合物25依赖于降低血浆胆固醇剂量,并且与Lederle ACAT抑制剂CL 277082一样有效。
-
Synthesis of α-Aryl Nitriles through Palladium-Catalyzed Decarboxylative Coupling of Cyanoacetate Salts with Aryl Halides and Triflates作者:Rui Shang、Dong-Sheng Ji、Ling Chu、Yao Fu、Lei LiuDOI:10.1002/anie.201006763日期:2011.5.2Worth its salt: The palladium‐catalyzed decarboxylative coupling of the cyanoacetate salt as well as its mono‐ and disubstituted derivatives with aryl chlorides, bromides, and triflates is described (see scheme). This reaction is potentially useful for the preparation of a diverse array of α‐aryl nitriles and has good functional group tolerance. S‐Phos=2‐(2,6‐dimethoxybiphenyl)dicyclohexylphosphine)
-
Palladium-Catalyzed Distal <i>m</i>-C–H Functionalization of Arylacetic Acid Derivatives作者:Dasari Srinivas、Gedu SatyanarayanaDOI:10.1021/acs.orglett.1c02460日期:2021.10.1Herein, we present m-C–H olefination on derivatives of phenylacetic acids by tethering with a simple nitrile-based template through palladium catalysis. Notably, the versatility of the method is evaluated with a wide range of phenylacetic acid derivatives for obtaining the meta-olefination products in fair to excellent yields with outstanding selectivities under mild conditions. Significantly, the
-
Dihydroxypyrimidine carbonic acid derivatives and their use in the treatment, amelioration or prevention of a viral disease申请人:SAVIRA PHARMACEUTICALS GMBH公开号:US20140038990A1公开(公告)日:2014-02-06The present invention relates to a compound having the general formula (Di), (Dii), or (Diii), optionally in the form of a pharmaceutically acceptable salt, solvate, polymorph, codrug, cocrystal, prodrug, tautomer, racemate, enantiomer, or diastereomer or mixture thereof, which are useful in treating, ameloriating or preventing a viral disease. Furthermore, specific combination therapies are disclosed.本发明涉及具有一般式(Di)、(Dii)或(Diii)的化合物,可选地为药学上可接受的盐、溶剂合物、多型体、共药、共晶、前药、互变异构体、消旋体、对映体或二对映体或其混合物,这些化合物在治疗、改善或预防病毒性疾病方面是有用的。此外,还披露了特定的联合疗法。
-
Hydration of Cyanohydrins by Highly Active Cationic Pt Catalysts: Mechanism and Scope作者:Chengcheng Li、Xiao-Yong Chang、Luqiong Huo、Haibo Tan、Xiangyou Xing、Chen XuDOI:10.1021/acscatal.1c02254日期:2021.7.16Cyanohydrins (α-hydroxy nitriles) are a special type of nitriles that readily decompose into hydrogen cyanide (HCN) and the corresponding carbonyl compounds. Hydration of cyanohydrins that are readily available through cyanation of aldehydes and ketones provides the most straightforward route to valuable α-hydroxyamides. However, due to low stability of cyanohydrins and deactivation of the catalysts氰醇(α-羟基腈)是一种特殊类型的腈,很容易分解成氰化氢 (HCN) 和相应的羰基化合物。可通过醛和酮的氰化轻松获得的氰醇的水合提供了获得有价值的 α-羟基酰胺的最直接途径。然而,由于氰醇的低稳定性和释放的 HCN 使催化剂失活,氰醇的催化直接水合仍然在很大程度上未解决。一般来说,含有较大取代基的氰醇,如α,α-二芳基氰醇,降解速度更快,因此更难水合。在这里,我们报告了对各种氰醇的水合表现出高反应性的阳离子铂催化剂的开发。2 OH)X(OTf) 揭示了一个催化循环,包括形成五元金属环中间体,然后通过 H 2 O攻击二级氧化膦 (PR 2 OH) 配体的磷进行水解。我们发现 Pt 催化剂甲轴承富电子,适当小的咬角双膦配体提供了一种用于氰醇的水合反应性超级。由A催化的水合反应在环境温度下进行,并与多种氰醇一起发生,包括最难的 α,α-二芳基氰醇,具有良好的转化率。
表征谱图
-
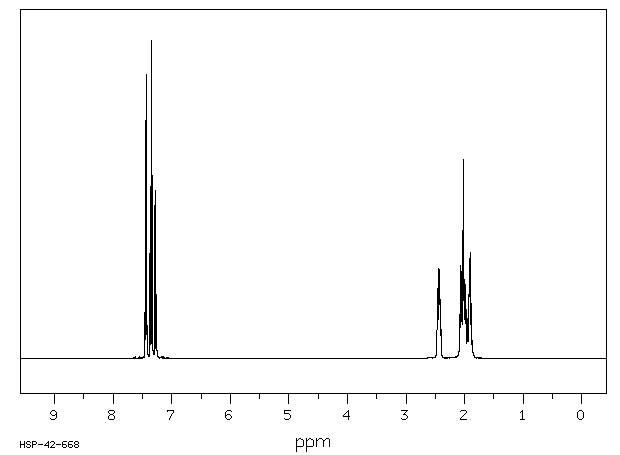
氢谱1HNMR
-
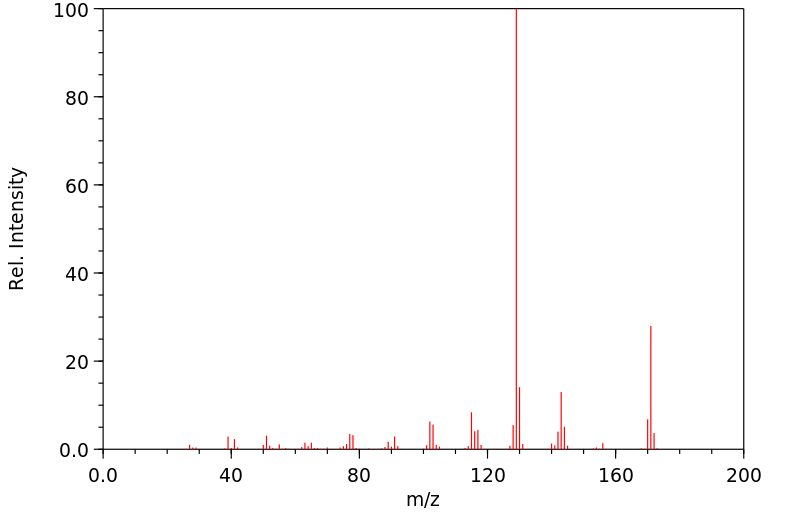
质谱MS
-
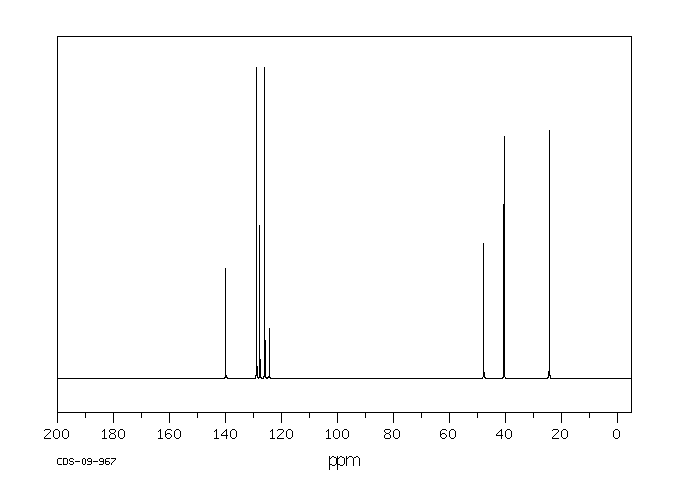
碳谱13CNMR
-
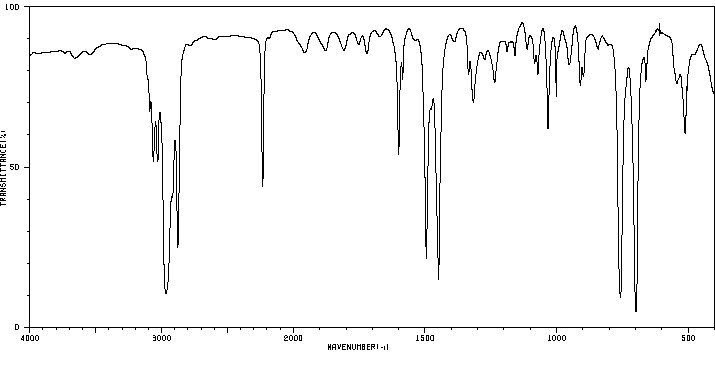
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(βS)-β-氨基-4-(4-羟基苯氧基)-3,5-二碘苯甲丙醇
(S,S)-邻甲苯基-DIPAMP
(S)-(-)-7'-〔4(S)-(苄基)恶唑-2-基]-7-二(3,5-二-叔丁基苯基)膦基-2,2',3,3'-四氢-1,1-螺二氢茚
(S)-盐酸沙丁胺醇
(S)-3-(叔丁基)-4-(2,6-二甲氧基苯基)-2,3-二氢苯并[d][1,3]氧磷杂环戊二烯
(S)-2,2'-双[双(3,5-三氟甲基苯基)膦基]-4,4',6,6'-四甲氧基联苯
(S)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(R)富马酸托特罗定
(R)-(-)-盐酸尼古地平
(R)-(-)-4,12-双(二苯基膦基)[2.2]对环芳烷(1,5环辛二烯)铑(I)四氟硼酸盐
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[((6-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(4-叔丁基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(3-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-4,7-双(3,5-二-叔丁基苯基)膦基-7“-[(吡啶-2-基甲基)氨基]-2,2”,3,3'-四氢1,1'-螺二茚满
(R)-3-(叔丁基)-4-(2,6-二苯氧基苯基)-2,3-二氢苯并[d][1,3]氧杂磷杂环戊烯
(R)-2-[((二苯基膦基)甲基]吡咯烷
(R)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(N-(4-甲氧基苯基)-N-甲基-3-(1-哌啶基)丙-2-烯酰胺)
(5-溴-2-羟基苯基)-4-氯苯甲酮
(5-溴-2-氯苯基)(4-羟基苯基)甲酮
(5-氧代-3-苯基-2,5-二氢-1,2,3,4-oxatriazol-3-鎓)
(4S,5R)-4-甲基-5-苯基-1,2,3-氧代噻唑烷-2,2-二氧化物-3-羧酸叔丁酯
(4S,4''S)-2,2''-亚环戊基双[4,5-二氢-4-(苯甲基)恶唑]
(4-溴苯基)-[2-氟-4-[6-[甲基(丙-2-烯基)氨基]己氧基]苯基]甲酮
(4-丁氧基苯甲基)三苯基溴化磷
(3aR,8aR)-(-)-4,4,8,8-四(3,5-二甲基苯基)四氢-2,2-二甲基-6-苯基-1,3-二氧戊环[4,5-e]二恶唑磷
(3aR,6aS)-5-氧代六氢环戊基[c]吡咯-2(1H)-羧酸酯
(2Z)-3-[[(4-氯苯基)氨基]-2-氰基丙烯酸乙酯
(2S,3S,5S)-5-(叔丁氧基甲酰氨基)-2-(N-5-噻唑基-甲氧羰基)氨基-1,6-二苯基-3-羟基己烷
(2S,2''S,3S,3''S)-3,3''-二叔丁基-4,4''-双(2,6-二甲氧基苯基)-2,2'',3,3''-四氢-2,2''-联苯并[d][1,3]氧杂磷杂戊环
(2S)-(-)-2-{[[[[3,5-双(氟代甲基)苯基]氨基]硫代甲基]氨基}-N-(二苯基甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[((1S,2S)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[[((1R,2R)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2-硝基苯基)磷酸三酰胺
(2,6-二氯苯基)乙酰氯
(2,3-二甲氧基-5-甲基苯基)硼酸
(1S,2S,3S,5S)-5-叠氮基-3-(苯基甲氧基)-2-[(苯基甲氧基)甲基]环戊醇
(1S,2S,3R,5R)-2-(苄氧基)甲基-6-氧杂双环[3.1.0]己-3-醇
(1-(4-氟苯基)环丙基)甲胺盐酸盐
(1-(3-溴苯基)环丁基)甲胺盐酸盐
(1-(2-氯苯基)环丁基)甲胺盐酸盐
(1-(2-氟苯基)环丙基)甲胺盐酸盐
(1-(2,6-二氟苯基)环丙基)甲胺盐酸盐
(-)-去甲基西布曲明
龙蒿油
龙胆酸钠
龙胆酸叔丁酯
龙胆酸
龙胆紫-d6
龙胆紫