5-氯-1,3-二甲基-4-硝基-1H-吡唑 | 13551-73-0
分子结构分类
中文名称
5-氯-1,3-二甲基-4-硝基-1H-吡唑
中文别名
5-氯-1,3-二甲基-4-硝基吡唑
英文名称
5-chloro-1,3-dimethyl-4-nitro-1H-pyrazole
英文别名
5-chloro-1,3-dimethyl-4-nitropyrazole
CAS
13551-73-0
化学式
C5H6ClN3O2
mdl
MFCD00052534
分子量
175.575
InChiKey
WBMBDRIOUHCVAS-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:75 °C
-
沸点:275.7±35.0 °C(Predicted)
-
密度:1.55±0.1 g/cm3(Predicted)
-
稳定性/保质期:
在常温常压下稳定,应避免与氧化物接触。
计算性质
-
辛醇/水分配系数(LogP):1.4
-
重原子数:11
-
可旋转键数:0
-
环数:1.0
-
sp3杂化的碳原子比例:0.4
-
拓扑面积:63.6
-
氢给体数:0
-
氢受体数:3
安全信息
-
危险品标志:Xi
-
安全说明:S26,S36/37/39
-
危险类别码:R36/37/38
-
海关编码:2933199090
-
储存条件:常温密闭,阴凉通风干燥
SDS
| Name: | 5-Chloro-1 3-dimethyl-4-nitro-1h-pyrazole 97% Material Safety Data Sheet |
| Synonym: | |
| CAS: | 13551-73-0 |
Synonym:
Section 2 - COMPOSITION, INFORMATION ON INGREDIENTS
| CAS# | Chemical Name | content | EINECS# |
| 13551-73-0 | 5-Chloro-1,3-dimethyl-4-nitro-1H-pyraz | 97% | unlisted |
Risk Phrases: 36/37/38
Section 3 - HAZARDS IDENTIFICATION
EMERGENCY OVERVIEW
Irritating to eyes, respiratory system and skin.
Potential Health Effects
Eye:
Causes eye irritation.
Skin:
Causes skin irritation. May be harmful if absorbed through the skin.
Ingestion:
May cause irritation of the digestive tract. May be harmful if swallowed.
Inhalation:
Causes respiratory tract irritation. May be harmful if inhaled.
Chronic:
Not available.
Section 4 - FIRST AID MEASURES
Eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid.
Skin:
Get medical aid. Flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes.
Ingestion:
Get medical aid. Wash mouth out with water.
Inhalation:
Remove from exposure and move to fresh air immediately. If not breathing, give artificial respiration. If breathing is difficult, give oxygen. Get medical aid.
Notes to Physician:
Treat symptomatically and supportively.
Section 5 - FIRE FIGHTING MEASURES
General Information:
As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear.
Extinguishing Media:
Use water spray, dry chemical, carbon dioxide, or chemical foam.
Section 6 - ACCIDENTAL RELEASE MEASURES
General Information: Use proper personal protective equipment as indicated in Section 8.
Spills/Leaks:
Vacuum or sweep up material and place into a suitable disposal container.
Section 7 - HANDLING and STORAGE
Handling:
Avoid breathing dust, vapor, mist, or gas. Avoid contact with skin and eyes.
Storage:
Store in a cool, dry place. Store in a tightly closed container.
Section 8 - EXPOSURE CONTROLS, PERSONAL PROTECTION
Engineering Controls:
Facilities storing or utilizing this material should be equipped with an eyewash facility and a safety shower. Use adequate ventilation to keep airborne concentrations low.
Exposure Limits CAS# 13551-73-0: Personal Protective Equipment Eyes: Not available.
Skin:
Wear appropriate protective gloves to prevent skin exposure.
Clothing:
Wear appropriate protective clothing to prevent skin exposure.
Respirators:
Follow the OSHA respirator regulations found in 29 CFR 1910.134 or European Standard EN 149. Use a NIOSH/MSHA or European Standard EN 149 approved respirator if exposure limits are exceeded or if irritation or other symptoms are experienced.
Section 9 - PHYSICAL AND CHEMICAL PROPERTIES
Physical State: Solid
Color: white
Odor: Not available.
pH: Not available.
Vapor Pressure: Not available.
Viscosity: Not available.
Boiling Point: Not available.
Freezing/Melting Point: 75 - 77 deg C
Autoignition Temperature: Not available.
Flash Point: Not available.
Explosion Limits, lower: Not available.
Explosion Limits, upper: Not available.
Decomposition Temperature:
Solubility in water:
Specific Gravity/Density:
Molecular Formula: C5H6ClN3O2
Molecular Weight: 176
Section 10 - STABILITY AND REACTIVITY
Chemical Stability:
Not available.
Conditions to Avoid:
Incompatible materials.
Incompatibilities with Other Materials:
Strong oxidizing agents.
Hazardous Decomposition Products:
Hydrogen chloride, chlorine, nitrogen oxides, carbon monoxide, carbon dioxide.
Hazardous Polymerization: Has not been reported
Section 11 - TOXICOLOGICAL INFORMATION
RTECS#:
CAS# 13551-73-0 unlisted.
LD50/LC50:
Not available.
Carcinogenicity:
5-Chloro-1,3-dimethyl-4-nitro-1H-pyrazole - Not listed by ACGIH, IARC, or NTP.
Section 12 - ECOLOGICAL INFORMATION
Section 13 - DISPOSAL CONSIDERATIONS
Dispose of in a manner consistent with federal, state, and local regulations.
Section 14 - TRANSPORT INFORMATION
IATA
No information available.
IMO
No information available.
RID/ADR
No information available.
Section 15 - REGULATORY INFORMATION
European/International Regulations
European Labeling in Accordance with EC Directives
Hazard Symbols: XI
Risk Phrases:
R 36/37/38 Irritating to eyes, respiratory system
and skin.
Safety Phrases:
S 26 In case of contact with eyes, rinse immediately
with plenty of water and seek medical advice.
S 37/39 Wear suitable gloves and eye/face
protection.
WGK (Water Danger/Protection)
CAS# 13551-73-0: No information available.
Canada
None of the chemicals in this product are listed on the DSL/NDSL list.
CAS# 13551-73-0 is not listed on Canada's Ingredient Disclosure List.
US FEDERAL
TSCA
CAS# 13551-73-0 is not listed on the TSCA inventory.
It is for research and development use only.
SECTION 16 - ADDITIONAL INFORMATION
N/A
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 5-氨基-1,3-二甲基-4-硝基吡唑 5-amino-1,3-dimethyl-4-nitro-1H-pyrazole 76689-64-0 C5H8N4O2 156.144 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 3-(溴甲基)-5-氯-1-甲基-4-硝基吡唑 3-Bromomethyl-5-chloro-1-methyl-4-nitropyrazole 132026-53-0 C5H5BrClN3O2 254.471 —— 5-chloro-3-(hydroxymethyl)-1-methyl-4-nitropyrazole 132026-57-4 C5H6ClN3O3 191.574 —— 5-chloro-3-<(2-methoxyethoxy)methyl>-1-methyl-4-nitropyrazole 132026-54-1 C8H12ClN3O4 249.654 —— 5-chloro-3-<<(2-methyl-1-propyl)oxy>methyl>-1-methyl-4-nitropyrazole 132026-56-3 C9H14ClN3O3 247.681 5-氯-1,3-二甲基吡唑-4-胺 5-chloro-1,3-dimethyl-pyrazol-4-amine 100674-51-9 C5H8ClN3 145.592
反应信息
-
作为反应物:参考文献:名称:5-Substituted derivatives of摘要:新的5-取代二嘧啶并[1,5-a:4',3'-e]吡嗪-6-羧酸衍生物,酯及其盐的化学式为 R.sup.1为氢、较低的烷基或苯基-较低的烷基;R.sup.2和R.sup.3分别为氢或较低的烷基;X为氧或硫;R.sup.4为氢、较低的烷基、苯基-较低的烷基或氨基-较低的烷基。这些新化合物可用作抗炎药和免疫抑制剂。公开号:US04077956A1
-
作为产物:描述:参考文献:名称:5-Substituted derivatives of摘要:新的5-取代二嘧啶并[1,5-a:4',3'-e]吡嗪-6-羧酸衍生物,酯及其盐的化学式为 R.sup.1为氢、较低的烷基或苯基-较低的烷基;R.sup.2和R.sup.3分别为氢或较低的烷基;X为氧或硫;R.sup.4为氢、较低的烷基、苯基-较低的烷基或氨基-较低的烷基。这些新化合物可用作抗炎药和免疫抑制剂。公开号:US04077956A1
文献信息
-
[EN] THIOPHENE DERIVATIVES FOR THE TREATMENT OF DISORDERS CAUSED BY IGE<br/>[FR] DÉRIVÉS DE THIOPHÈNE POUR LE TRAITEMENT DE TROUBLES PROVOQUÉS PAR IGE申请人:UCB BIOPHARMA SRL公开号:WO2019243550A1公开(公告)日:2019-12-26Thiophene derivatives of formula (I) and a pharmaceutically acceptable salt thereof are provided. These compounds have utility for the treatment or prevention of disorders caused by IgE, such as allergy, type 1 hypersensitivity or familiar sinus inflammation.
-
[EN] TRPML MODULATORS<br/>[FR] MODULATEURS DE TRPML申请人:CASMA THERAPEUTICS INC公开号:WO2021127337A1公开(公告)日:2021-06-24The present invention provides compounds, pharmaceutically acceptable compositions thereof, and methods of using the same.本发明提供了化合物、药学上可接受的组合物以及使用这些化合物的方法。
-
[EN] (5,6-DIHYDRO)PYRIMIDO[4,5-E]INDOLIZINES<br/>[FR] (5,6-DIHYDRO)PYRIMIDO[4,5-E]INDOZILINES申请人:NETHERLANDS TRANSLATIONAL RES CT B V公开号:WO2015155042A1公开(公告)日:2015-10-15The invention relates to a compound of Formula (I) wherein, R1 and R2 independently are selected from the group consisting of optionally substituted (6-10C)aryl and (1-5C)heteroaryl groups. The compounds can be used in pharmaceutical compositions, in particular in the treatment of cancer.本发明涉及一种公式(I)的化合物,其中R1和R2独立地选自包括可选择性取代的(6-10C)芳基和(1-5C)杂芳基基团在内的组。这些化合物可用于药物组合物中,特别是在癌症治疗中。
-
Method of treating a patient having precancerous lesions with phenyl purinone derivatives
-
Pyrazolopyrimidinone antianginal agents申请人:Pfizer Inc.公开号:US05250534A1公开(公告)日:1993-10-05Compounds of the formula: ##STR1## wherein R.sup.1 is H, C.sub.1 -C.sub.3 alkyl, C.sub.3 -C.sub.5 cycloalkyl or C.sub.1 -C.sub.3 perfluoroalkyl; R.sup.2 is H, C.sub.1 -C.sub.6 alkyl optionally substituted by OH, C.sub.1 -C.sub.3 alkoxy or C.sub.3 -C.sub.6 cycloalkyl, or C.sub.1 -C.sub.3 perfluoroalkyl; R.sup.3 is C.sub.1 -C.sub.6 alkyl, C.sub.3 -C.sub.6 alkenyl, C.sub.3 -C.sub.6 alkynyl, C.sub.3 -C.sub.7 cycloalkyl, C.sub.1 -C.sub.6 perfluoroalkyl or (C.sub.3 -C.sub.6 cycloalkyl)C.sub.1 -C.sub.6 alkyl; R.sup.4 taken together with the nitrogen atom to which it is attached completes a pyrrolidinyl, piperidino, morpholino, or 4-N-(R.sup.6)-piperazinyl group; R.sup.5 is H, C.sub.1 -C.sub.4 alkyl, C.sub.1 -C.sub.3 alkoxy, NR.sup.7 R.sup.8, or CONR.sup.7 R.sup.8 ; R.sup.6 is H, C.sub.1 -C.sub.6 alkyl, (C.sub.1 -C.sub.3 alkoxy) C.sub.2 - C.sub.6 alkyl, hydroxy C.sub.2 -C.sub.6 alkyl, (R.sup.7 R.sup.8 N)C.sub.2 -C.sub.6 alkyl, (R.sup.7 R.sup.8 NCO)C.sub.1 -C.sub.6 alkyl, CONR.sup.7 R.sup.8, CSNR.sup.7 R.sup.8 or C(NH)NR.sup.7 R.sup.8 ; R.sup.7 and R.sup.8 are each independently H, C.sub.1 -C.sub.4 alkyl, (C.sub.1 -C.sub.3 alkoxy)C.sub.2 -C.sub.4 alkyl or hydroxy C.sub.2 -C.sub.4 alkyl; and pharmaceutically acceptable salts thereof, are selective cGMP PDE inhibitors useful in the treatment of cardiovascular disorders such as angina, hypertension, heart failure and atherosclerosis.该化合物的结构式为:##STR1## 其中R.sup.1为H,C.sub.1 -C.sub.3烷基,C.sub.3 -C.sub.5环烷基或C.sub.1 -C.sub.3全氟烷基;R.sup.2为H,C.sub.1 -C.sub.6烷基,可以选择性地被OH,C.sub.1 -C.sub.3烷氧基或C.sub.3 -C.sub.6环烷基,或C.sub.1 -C.sub.3全氟烷基取代;R.sup.3为C.sub.1 -C.sub.6烷基,C.sub.3 -C.sub.6烯基,C.sub.3 -C.sub.6炔基,C.sub.3 -C.sub.7环烷基,C.sub.1 -C.sub.6全氟烷基或(C.sub.3 -C.sub.6环烷基)C.sub.1 -C.sub.6烷基;R.sup.4与其连接的氮原子一起形成吡咯啉基、哌啶基、吗啉基或4-N-(R.sup.6)-哌嗪基;R.sup.5为H,C.sub.1 -C.sub.4烷基,C.sub.1 -C.sub.3烷氧基,NR.sup.7 R.sup.8,或CONR.sup.7 R.sup.8;R.sup.6为H,C.sub.1 -C.sub.6烷基,(C.sub.1 -C.sub.3烷氧基)C.sub.2 - C.sub.6烷基,羟基C.sub.2 -C.sub.6烷基,(R.sup.7 R.sup.8 N)C.sub.2 -C.sub.6烷基,(R.sup.7 R.sup.8 NCO)C.sub.1 -C.sub.6烷基,CONR.sup.7 R.sup.8,CSNR.sup.7 R.sup.8或C(NH)NR.sup.7 R.sup.8;R.sup.7和R.sup.8各自独立地为H,C.sub.1 -C.sub.4烷基,(C.sub.1 -C.sub.3烷氧基)C.sub.2 -C.sub.4烷基或羟基C.sub.2 -C.sub.4烷基;以及其药学上可接受的盐,是用于治疗心血管疾病如心绞痛、高血压、心力衰竭和动脉粥样硬化的选择性cGMP PDE抑制剂。
表征谱图
-
氢谱1HNMR
-
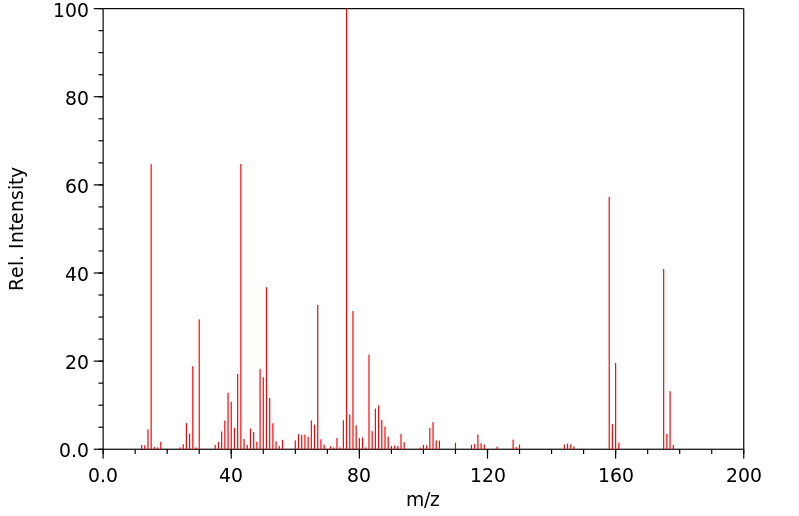
质谱MS
-
碳谱13CNMR
-
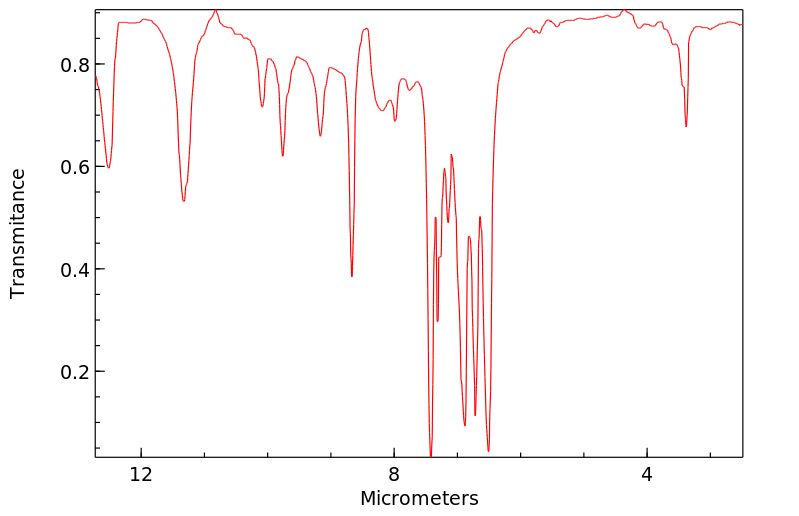
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
顺式-2-硝基环己基乙酸酯
顺式-2-硝基-6-甲基环己酮
雷尼替丁杂质18
铝硝基甲烷三氯化物
钾离子载体III
重氮(硝基)甲烷
醛基-七聚乙二醇-叠氮
过氧亚甲基
辛腈,4-氟-4-硝基-7-羰基-
辛烷,1,2-二氯-1-硝基-
赤霉素A4+7(GA4:GA7=65:35)
苄哒唑
羟胺-四聚乙二醇-叠氮
羟胺-三乙二醇-叠氮
米索硝唑
磷酸十二醇酯
碘硝基甲烷
碘化e1,1-二甲基-4-羰基-3,5-二(3-苯基-2-亚丙烯基)哌啶正离子
硝酰胺
硝基脲银(I)复合物
硝基甲醇
硝基甲烷-d3
硝基甲烷-13C,d3
硝基甲烷-13C
硝基甲烷-(15)N
硝基甲烷
硝基甲基甲醇胺
硝基环辛烷
硝基环戊烷
硝基环戊基阴离子
硝基环庚烷
硝基环己烷锂盐
硝基环己烷钾盐
硝基环己烷
硝基环丁烷
硝基氨基甲酸
硝基新戊烷
硝基二乙醇胺
硝基乙醛缩二甲醇
硝基乙醛缩二乙醇
硝基乙腈
硝基乙烷-D5
硝基乙烷-1,1-d2
硝基乙烷
硝基乙烯
硝基丙烷
硝基丙二醛(E,E)-二肟
硝基丙二腈
硝基-(3-硝基-[4]吡啶基)-胺
硝乙醛肟