1,3,5-三甲基-吡唑-4-胺 | 28466-21-9
中文名称
1,3,5-三甲基-吡唑-4-胺
中文别名
4-氨基-1,3,5-三甲基吡唑;4-氨基-1,3,5-三甲基-1H-吡唑;4-氨基-1,3,5-三甲基吡唑,97%;1,3,5-三甲基-4-氨基吡唑
英文名称
1,3,5-trimethyl-1H-pyrazol-4-amine
英文别名
4-amino-1,3,5-trimethyl-1H-pyrazole;1,3,5-trimethyl-4-aminopyrazole;4-amino-1,3,5-trimethylpyrazole;4-Amino-1,3,5-trimethylpyrazol;1,3,5-trimethyl-1H-pyrazol-4-ylamine;1,3,5-trimethylpyrazol-4-amine
CAS
28466-21-9
化学式
C6H11N3
mdl
MFCD00052883
分子量
125.173
InChiKey
SSDGMKHZMNTWLS-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:96 °C
-
沸点:236℃
-
密度:1.13
-
闪点:97℃
-
稳定性/保质期:
如果遵照规格使用和储存则不会分解,未有已知危险反应。避免与氧化物接触。
计算性质
-
辛醇/水分配系数(LogP):0.4
-
重原子数:9
-
可旋转键数:0
-
环数:1.0
-
sp3杂化的碳原子比例:0.5
-
拓扑面积:43.8
-
氢给体数:1
-
氢受体数:2
安全信息
-
危险品标志:Xi
-
RTECS号:UQ6125100
-
海关编码:2933199090
-
安全说明:S22,S24/25
-
危险性防范说明:P280,P305+P351+P338
-
危险性描述:H302
-
储存条件:室温
SDS
| Name: | 1 3 5-Trimethyl-1H-pyrazol-4-amine 97% Material Safety Data Sheet |
| Synonym: | |
| CAS: | 28466-21-9 |
Synonym:
Section 2 - COMPOSITION, INFORMATION ON INGREDIENTS
| CAS# | Chemical Name | content | EINECS# |
| 28466-21-9 | 1,3,5-Trimethyl-1H-pyrazol-4-amine | 97% | unlisted |
Risk Phrases: None Listed.
Section 3 - HAZARDS IDENTIFICATION
EMERGENCY OVERVIEW
Not available.
Potential Health Effects
Eye:
May cause eye irritation.
Skin:
May cause skin irritation. May be harmful if absorbed through the skin.
Ingestion:
May cause irritation of the digestive tract. May be harmful if swallowed.
Inhalation:
May cause respiratory tract irritation. May be harmful if inhaled.
Chronic:
Not available.
Section 4 - FIRST AID MEASURES
Eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid.
Skin:
Get medical aid. Flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes.
Ingestion:
Get medical aid. Wash mouth out with water.
Inhalation:
Remove from exposure and move to fresh air immediately.
Notes to Physician:
Treat symptomatically and supportively.
Section 5 - FIRE FIGHTING MEASURES
General Information:
As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear.
Extinguishing Media:
Use water spray, dry chemical, carbon dioxide, or chemical foam.
Section 6 - ACCIDENTAL RELEASE MEASURES
General Information: Use proper personal protective equipment as indicated in Section 8.
Spills/Leaks:
Vacuum or sweep up material and place into a suitable disposal container.
Section 7 - HANDLING and STORAGE
Handling:
Avoid breathing dust, vapor, mist, or gas. Avoid contact with skin and eyes.
Storage:
Store in a cool, dry place. Store in a tightly closed container.
Section 8 - EXPOSURE CONTROLS, PERSONAL PROTECTION
Engineering Controls:
Use adequate ventilation to keep airborne concentrations low.
Exposure Limits CAS# 28466-21-9: Personal Protective Equipment Eyes: Not available.
Skin:
Wear appropriate protective gloves to prevent skin exposure.
Clothing:
Wear appropriate protective clothing to prevent skin exposure.
Respirators:
Follow the OSHA respirator regulations found in 29 CFR 1910.134 or European Standard EN 149. Use a NIOSH/MSHA or European Standard EN 149 approved respirator if exposure limits are exceeded or if irritation or other symptoms are experienced.
Section 9 - PHYSICAL AND CHEMICAL PROPERTIES
Physical State: Solid
Color: beige
Odor: Not available.
pH: Not available.
Vapor Pressure: Not available.
Viscosity: Not available.
Boiling Point: Not available.
Freezing/Melting Point: 101 - 102 deg C
Autoignition Temperature: Not available.
Flash Point: Not available.
Explosion Limits, lower: Not available.
Explosion Limits, upper: Not available.
Decomposition Temperature:
Solubility in water:
Specific Gravity/Density:
Molecular Formula: C6H11N3
Molecular Weight: 125
Section 10 - STABILITY AND REACTIVITY
Chemical Stability:
Not available.
Conditions to Avoid:
Incompatible materials.
Incompatibilities with Other Materials:
Strong oxidizing agents.
Hazardous Decomposition Products:
Nitrogen oxides, carbon monoxide, carbon dioxide.
Hazardous Polymerization: Has not been reported
Section 11 - TOXICOLOGICAL INFORMATION
RTECS#:
CAS# 28466-21-9: UQ6125100 LD50/LC50:
Not available.
Carcinogenicity:
1,3,5-Trimethyl-1H-pyrazol-4-amine - Not listed by ACGIH, IARC, or NTP.
Other:
See actual entry in RTECS for complete information.
Section 12 - ECOLOGICAL INFORMATION
Section 13 - DISPOSAL CONSIDERATIONS
Dispose of in a manner consistent with federal, state, and local regulations.
Section 14 - TRANSPORT INFORMATION
IATA
No information available.
IMO
No information available.
RID/ADR
No information available.
Section 15 - REGULATORY INFORMATION
European/International Regulations
European Labeling in Accordance with EC Directives
Hazard Symbols: Not available.
Risk Phrases:
Safety Phrases:
S 24/25 Avoid contact with skin and eyes.
WGK (Water Danger/Protection)
CAS# 28466-21-9: No information available.
Canada
None of the chemicals in this product are listed on the DSL/NDSL list.
CAS# 28466-21-9 is not listed on Canada's Ingredient Disclosure List.
US FEDERAL
TSCA
CAS# 28466-21-9 is not listed on the TSCA inventory.
It is for research and development use only.
SECTION 16 - ADDITIONAL INFORMATION
N/A
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 1,3,5-三甲基-4-亚硝基-1H-吡唑 1,3,5-trimethyl-4-nitrosopyrazole 7171-70-2 C6H9N3O 139.157 4-硝基-1,3,5-三甲基吡唑 1,3,5-trimethyl-4-nitro-1H-pyrazole 1125-30-0 C6H9N3O2 155.156 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 —— 4-isothiocyanato-1,3,5-trimethyl-1H-pyrazole 63204-11-5 C7H9N3S 167.235 —— (3,5-dimethyl-1H-pyrazol-4-yl)-(1,3,5-trimethyl-1H-pyrazol-4-yl)-diazene —— C11H16N6 232.288 —— N-isobutyl-1,3,5-trimethyl-1H-pyrazol-4-amine 1152653-90-1 C10H19N3 181.281 —— N-(1,3,5-trimethylpyrazol-4-yl)acetamide 221089-46-9 C8H13N3O 167.211 —— (E)-1,3,5-trimethyl-4-(phenyldiazenyl)-1H-pyrazole —— C12H14N4 214.27 —— N-(4-bromobenzyl)-1,3,5-trimethyl-1H-pyrazol-4-amine 1152826-59-9 C13H16BrN3 294.194 —— 3-[(1,3,5-Trimethyl-1H-pyrazol-4-yl)-hydrazono]-pentane-2,4-dione —— C11H16N4O2 236.274 —— N-propyl-N'-(1,3,5-trimethylpyrazole-4-yl)thiourea —— C10H18N4S 226.346
反应信息
-
作为反应物:描述:参考文献:名称:Knorr, Chemische Berichte, 1895, vol. 28, p. 688摘要:DOI:
-
作为产物:描述:2,3,4-戊三酮肟 (6ci,7ci,8ci,9ci) 在 lithium aluminium tetrahydride 作用下, 以 乙醚 、 乙醇 为溶剂, 反应 3.0h, 生成 1,3,5-三甲基-吡唑-4-胺参考文献:名称:吡唑和吡唑鎓盐与复杂的金属氢化物和有机金属试剂的反应。吡唑啉和吡唑烷的合成摘要:2-吡唑啉-4-肟是通过4-亚硝基和4-硝基吡唑与复杂的金属氢化物和有机金属试剂的反应合成的。此外,4-硝基吡唑鎓四氟硼酸盐是有机锂和格氏试剂的反应性底物,从而导致新的和高度取代的4-硝基-3-吡唑啉和4-硝基吡唑烷。3-吡唑啉的形成是区域选择性的,并且当可能存在非对映异构体吡唑烷时,仅获得最稳定的3,4-反式和/或4,5-反式异构体。DOI:10.1016/s0040-4020(97)00514-0
文献信息
-
Synthesis and biological evaluation of new N-substituted-N′-(3,5-di/1,3,5-trimethylpyrazole-4-yl)thiourea/urea derivatives作者:Bedia Koçyiğit Kaymakçıoğlu、Sevim Rollas、Eylem Körceğez、Feyza ArıcıoğluDOI:10.1016/j.ejps.2005.05.005日期:2005.9Several thiourea and urea derivatives were prepared by the reaction of 4-aminopyrazoles with substituted isothiocyanates or isocyanates. The novel compounds were tested anticonvulsant activity using by pentylenetetrazole-induced seizure (PTZ) and maximal electroshock seizure (MES) tests. Among the tested compounds, thiourea derivatives of 4b were afforded 90 and 100% protection in PTZ and MES tests
-
CHEMICAL SUBSTANCES WHICH INHIBIT THE ENZYMATIC ACTIVITY OF HUMAN KALLIKREIN-RELATED PEPTIDASE 6 (KLK6)申请人:Deutsches Krebsforschungszentrum公开号:EP3305781A1公开(公告)日:2018-04-11The invention relates to compounds which are suitable for the treatment of a disease associated with kallikrein-like peptidase 6 overexpression and to pharmaceutical compositions containing such compounds. The invention further relates to a kit of parts comprising such compounds or pharmaceutical compositions.这项发明涉及适用于治疗与kallikrein样肽酶6过度表达相关疾病的化合物,以及含有这些化合物的药物组合物。该发明还涉及包括这些化合物或药物组合物的配套工具包。
-
PYRIMIDINE INHIBITORS OF KINASES申请人:Curtin Michael公开号:US20100317680A1公开(公告)日:2010-12-16The present invention relates to compounds of formula (I) or pharmaceutical acceptable salts, wherein A 1 , A 2 , A 3 , A 4 , X and Y are defined in the description. The present invention relates also to methods of making said compounds, and compositions containing said compounds which are useful for inhibiting kinases such as aurora and KDR.本发明涉及式(I)的化合物或药用可接受的盐,其中A1、A2、A3、A4、X和Y在描述中有定义。本发明还涉及制备所述化合物的方法,以及含有所述化合物的组合物,用于抑制aurora和KDR等激酶。
-
Substituted 1-aryl-3-heteroaryl-thioureas (or isothioureas) as antiatherosclerotic agents申请人:Wyeth公开号:US06455566B1公开(公告)日:2002-09-24Antiatherosclerotic agents are provided which are represented by Formulas I or II: wherein R is wherein R9, R10, R11, R12, R13, and R14 are each, independently, hydrogen or a lower alkyl of 1-6 carbon atoms; R6, and R7 are each, independently, hydrogen, lower alkyl of 1-6 carbon atoms, or CH2COOR8, where R8 is a lower alkyl of 1-6 carbon atoms; and X is O or S; R1 is hydrogen or a lower alkyl of 1-6 carbon atoms; R2, R3, and R4 are each, independently, hydrogen or halogen; and R5 is a lower alkyl of 1-6 carbon atoms; or a pharmaceutically acceptable salt thereof.提供了代表为Formula I或II的抗动脉粥样硬化药物: 其中 R为 其中R9、R10、R11、R12、R13和R14分别独立地为氢或1-6个碳原子的较低烷基; R6和R7分别独立地为氢、1-6个碳原子的较低烷基或CH2COOR8,其中R8为1-6个碳原子的较低烷基;以及 X为O或S; R1为氢或1-6个碳原子的较低烷基; R2、R3和R4分别独立地为氢或卤素;以及 R5为1-6个碳原子的较低烷基; 或其药用可接受盐。
-
[EN] PYRIDINE COMPOUNDS<br/>[FR] COMPOSÉS PYRIDINES申请人:ASTRAZENECA AB公开号:WO2009153589A1公开(公告)日:2009-12-23The present invention relates to compounds that inhibit of focal adhesion kinase function, processes for their preparation, pharmaceutical compositions containing them as the active ingredient, to their use as medicaments and to their use in the manufacture of medicaments for use in the treatment in warm-blooded animals such as humans of diseases such as cancer.本发明涉及抑制细胞焦点粘附激酶功能的化合物,其制备方法,含有其作为活性成分的药物组合物,以及它们作为药物的用途,以及用于制造用于治疗温血动物(如人类)癌症等疾病的药物。
表征谱图
-
氢谱1HNMR
-
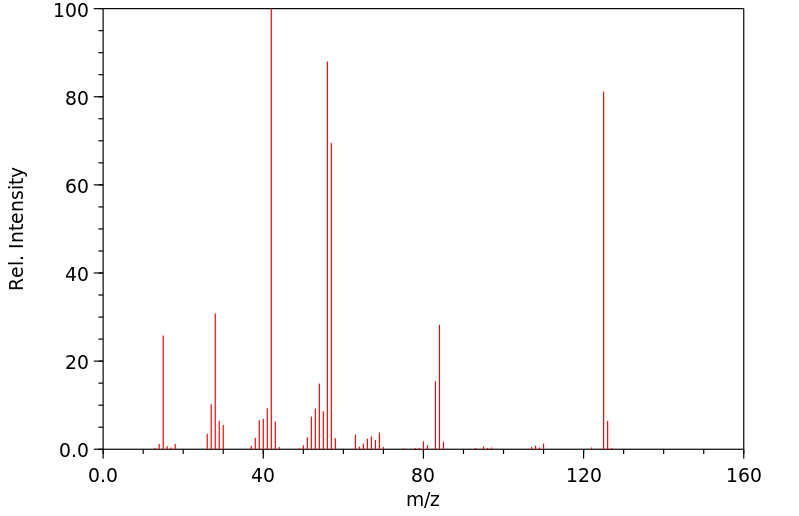
质谱MS
-
碳谱13CNMR
-
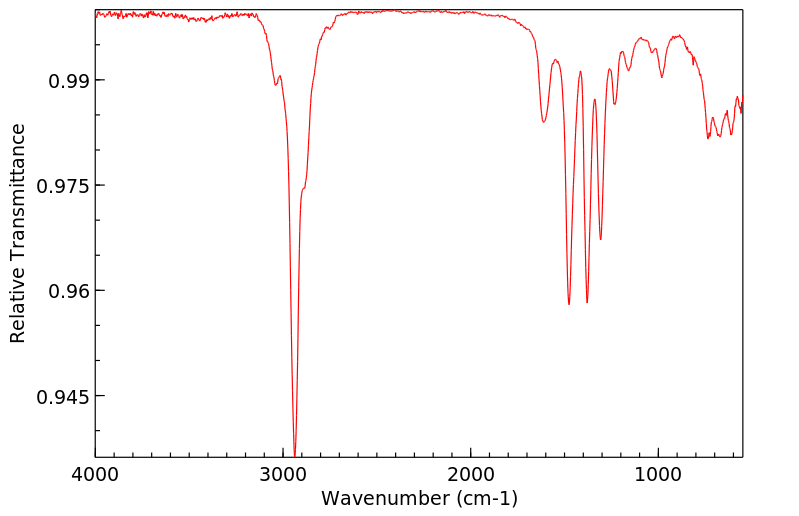
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(SP-4-1)-二氯双(1-苯基-1H-咪唑-κN3)-钯
(5aS,6R,9S,9aR)-5a,6,7,8,9,9a-六氢-6,11,11-三甲基-2-(2,3,4,5,6-五氟苯基)-6,9-甲基-4H-[1,2,4]三唑[3,4-c][1,4]苯并恶嗪四氟硼酸酯
(5-氨基-1,3,4-噻二唑-2-基)甲醇
齐墩果-2,12-二烯[2,3-d]异恶唑-28-酸
黄曲霉毒素H1
高效液相卡套柱
非昔硝唑
非布索坦杂质Z19
非布索坦杂质T
非布索坦杂质K
非布索坦杂质E
非布索坦杂质D
非布索坦杂质67
非布索坦杂质65
非布索坦杂质64
非布索坦杂质61
非布索坦代谢物67M-4
非布索坦代谢物67M-2
非布索坦代谢物 67M-1
非布索坦-D9
非布索坦
非唑拉明
雷非那酮-d7
雷西那德杂质2
雷西纳德杂质L
雷西纳德杂质H
雷西纳德杂质B
雷西纳德
雷西奈德杂质
阿西司特
阿莫奈韦
阿考替胺杂质9
阿米苯唑
阿米特罗13C2,15N2
阿瑞匹坦杂质
阿格列扎
阿扎司特
阿尔吡登
阿塔鲁伦中间体
阿培利司N-1
阿哌沙班杂质26
阿哌沙班杂质15
阿可替尼
阿作莫兰
阿佐塞米
镁(2+)(Z)-4'-羟基-3'-甲氧基肉桂酸酯
锌1,2-二甲基咪唑二氯化物
锌(II)(苯甲醇)(四苯基卟啉)
锌(II)(正丁醇)(四苯基卟啉)
锌(II)(异丁醇)(四苯基卟啉)