N-苄基马来酰亚胺 | 1631-26-1
中文名称
N-苄基马来酰亚胺
中文别名
苄基马来酰亚胺
英文名称
1-benzyl-1H-pyrrole-2,5-dione
英文别名
N-Benzylmaleimide;1-benzylpyrrole-2,5-dione
CAS
1631-26-1
化学式
C11H9NO2
mdl
MFCD00014540
分子量
187.198
InChiKey
MKRBAPNEJMFMHU-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:70 °C (lit.)
-
沸点:177 °C / 20mmHg
-
密度:1 g/cm3
-
溶解度:可溶于氯仿(少许)、甲醇(少许)
-
稳定性/保质期:
常温常压下稳定,避免与氧化物、酸和碱接触。
计算性质
-
辛醇/水分配系数(LogP):1
-
重原子数:14
-
可旋转键数:2
-
环数:2.0
-
sp3杂化的碳原子比例:0.09
-
拓扑面积:37.4
-
氢给体数:0
-
氢受体数:2
安全信息
-
危险等级:8
-
危险品标志:T
-
安全说明:S26,S36/37/39,S45
-
危险类别码:R20/21,R34,R25
-
WGK Germany:3
-
海关编码:2925190090
-
危险品运输编号:UN 1759 8/PG 2
-
危险类别:8
-
包装等级:III
-
危险性防范说明:P280,P305+P351+P338,P310
-
危险性描述:H314
-
储存条件:请将容器密封保存,并存放在阴凉干燥处。
SDS
| Name: | N-Benzylmaleimide Material Safety Data Sheet |
| Synonym: | None Known |
| CAS: | 1631-26-1 |
Synonym:None Known
Section 2 - COMPOSITION, INFORMATION ON INGREDIENTS
| CAS# | Chemical Name | content | EINECS# |
| 1631-26-1 | N-Benzylmaleimide | ca. 100 | 216-631-1 |
Risk Phrases: 34
Section 3 - HAZARDS IDENTIFICATION
EMERGENCY OVERVIEW
Causes burns.Corrosive.
Potential Health Effects
Eye:
Causes eye burns. May cause chemical conjunctivitis and corneal damage.
Skin:
Causes skin burns. May cause skin rash (in milder cases), and cold and clammy skin with cyanosis or pale color.
Ingestion:
May cause severe and permanent damage to the digestive tract. Causes gastrointestinal tract burns. May cause perforation of the digestive tract. The toxicological properties of this substance have not been fully investigated. May cause systemic effects.
Inhalation:
Causes chemical burns to the respiratory tract. The toxicological properties of this substance have not been fully investigated.
Aspiration may lead to pulmonary edema. May cause systemic effects.
Chronic:
Effects may be delayed.
Section 4 - FIRST AID MEASURES
Eyes: Get medical aid immediately. Do NOT allow victim to rub eyes or keep eyes closed. Extensive irrigation with water is required (at least 30 minutes).
Skin:
Get medical aid immediately. Immediately flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes. Wash clothing before reuse. Destroy contaminated shoes.
Ingestion:
Do not induce vomiting. If victim is conscious and alert, give 2-4 cupfuls of milk or water. Never give anything by mouth to an unconscious person. Get medical aid immediately.
Inhalation:
Get medical aid immediately. Remove from exposure and move to fresh air immediately. If breathing is difficult, give oxygen. Do NOT use mouth-to-mouth resuscitation. If breathing has ceased apply artificial respiration using oxygen and a suitable mechanical device such as a bag and a mask.
Notes to Physician:
Section 5 - FIRE FIGHTING MEASURES
General Information:
As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear. During a fire, irritating and highly toxic gases may be generated by thermal decomposition or combustion.
Extinguishing Media:
Use water spray, dry chemical, carbon dioxide, or chemical foam.
Section 6 - ACCIDENTAL RELEASE MEASURES
General Information: Use proper personal protective equipment as indicated in Section 8.
Spills/Leaks:
Vacuum or sweep up material and place into a suitable disposal container. Clean up spills immediately, observing precautions in the Protective Equipment section. Avoid generating dusty conditions.
Provide ventilation.
Section 7 - HANDLING and STORAGE
Handling:
Minimize dust generation and accumulation. Do not breathe dust, vapor, mist, or gas. Do not get in eyes, on skin, or on clothing.
Keep container tightly closed. Do not ingest or inhale. Use only in a chemical fume hood. Discard contaminated shoes.
Storage:
Store in a cool, dry place. Store in a tightly closed container.
Corrosives area.
Section 8 - EXPOSURE CONTROLS, PERSONAL PROTECTION
Engineering Controls:
Facilities storing or utilizing this material should be equipped with an eyewash facility and a safety shower. Use adequate ventilation to keep airborne concentrations low.
Exposure Limits CAS# 1631-26-1: Personal Protective Equipment Eyes: Wear appropriate protective eyeglasses or chemical safety goggles as described by OSHA's eye and face protection regulations in 29 CFR 1910.133 or European Standard EN166.
Skin:
Wear appropriate protective gloves to prevent skin exposure.
Clothing:
Wear appropriate protective clothing to prevent skin exposure.
Respirators:
Follow the OSHA respirator regulations found in 29 CFR 1910.134 or European Standard EN 149. Use a NIOSH/MSHA or European Standard EN 149 approved respirator if exposure limits are exceeded or if irritation or other symptoms are experienced.
Section 9 - PHYSICAL AND CHEMICAL PROPERTIES
Physical State: Solid
Color: Not available.
Odor: Not available.
pH: Not available.
Vapor Pressure: Not available.
Viscosity: Not available.
Boiling Point: Not available.
Freezing/Melting Point: 70 deg C
Autoignition Temperature: Not available.
Flash Point: Not available.
Explosion Limits, lower: Not available.
Explosion Limits, upper: Not available.
Decomposition Temperature:
Solubility in water:
Specific Gravity/Density:
Molecular Formula: C11H9NO2
Molecular Weight: 187.20
Section 10 - STABILITY AND REACTIVITY
Chemical Stability:
Stable at room temperature in closed containers under normal storage and handling conditions.
Conditions to Avoid:
Incompatible materials, dust generation.
Incompatibilities with Other Materials:
Oxidizing agents.
Hazardous Decomposition Products:
Nitrogen oxides, carbon monoxide, carbon dioxide.
Hazardous Polymerization: Has not been reported
Section 11 - TOXICOLOGICAL INFORMATION
RTECS#:
CAS# 1631-26-1 unlisted.
LD50/LC50:
Not available.
Carcinogenicity:
N-Benzylmaleimide - Not listed by ACGIH, IARC, or NTP.
Section 12 - ECOLOGICAL INFORMATION
Ecotoxicity:
Fish: Pseudomonas putida:
Section 13 - DISPOSAL CONSIDERATIONS
Dispose of in a manner consistent with federal, state, and local regulations.
Section 14 - TRANSPORT INFORMATION
IATA
Shipping Name: NOT REVIEWED.
Hazard Class:
UN Number:
Packing Group:
IMO
Shipping Name: NOT REVIEWED.
Hazard Class:
UN Number:
Packing Group:
RID/ADR
Shipping Name: NOT REVIEWED.
Hazard Class:
UN Number:
Packing group:
Section 15 - REGULATORY INFORMATION
European/International Regulations
European Labeling in Accordance with EC Directives
Hazard Symbols: C
Risk Phrases:
R 34 Causes burns.
Safety Phrases:
S 25 Avoid contact with eyes.
S 36/37/39 Wear suitable protective clothing, gloves
and eye/face protection.
S 45 In case of accident or if you feel unwell, seek
medical advice immediately (show the label where
possible).
WGK (Water Danger/Protection)
CAS# 1631-26-1: No information available.
Canada
None of the chemicals in this product are listed on the DSL/NDSL list.
CAS# 1631-26-1 is not listed on Canada's Ingredient Disclosure List.
US FEDERAL
TSCA
CAS# 1631-26-1 is not listed on the TSCA inventory.
It is for research and development use only.
SECTION 16 - ADDITIONAL INFORMATION
N/A
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 —— 3-(benzylcarbamoyl)acrylic acid 134575-94-3 C11H11NO3 205.213 N-苯基马来酸 N-benzylmaleamic acid 15329-69-8 C11H11NO3 205.213 —— fumaric acid monobenzyl amide 685543-77-5 C11H11NO3 205.213 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 1-苄基-1,5-二氢-吡咯-2-酮 1-benzyl-1H-pyrrol-2(5H)-one 64330-46-7 C11H11NO 173.214 —— N-benzyl-3-bromomaleimide 112749-49-2 C11H8BrNO2 266.094 —— 1-benzyl-3-chloro-1H-pyrrole-2,5-dione 112749-59-4 C11H8ClNO2 221.643 —— 1-benzyl-5-hydroxy-1,5-dihydro-2H-pyrrol-2-one 323204-69-9 C11H11NO2 189.214 N-苄基-2,3-二溴马来酰亚胺 N-benzyl-2,3-dibromo-maleimide 91026-00-5 C11H7Br2NO2 344.99 1-苄基-3-吡咯啉 1-benzyl-2,5-dihydro-1H-pyrrole 6913-92-4 C11H13N 159.231 1-苄基吡咯烷-2,5-二酮 1-(phenylmethyl)-2,5-pyrrolidinedione 2142-06-5 C11H11NO2 189.214 —— fumaric acid monobenzyl amide 685543-77-5 C11H11NO3 205.213 —— (E)-1-benzyl-3-ethylidenepyrrolidine-2,5-dione 1215092-36-6 C13H13NO2 215.252
反应信息
-
作为反应物:参考文献:名称:一种一锅法合成3-氨基吡咯烷盐酸盐的方法摘要:本发明涉及精细化工领域,具体涉及一种一锅法合成3‑氨基吡咯烷盐酸盐的方法。该方法利用马来酰亚胺剂化合物先与廉价的叠氮化合物发生步骤一加成反应,再在催化剂作用下,在同一反应过程中发生步骤二羰基氢化还原,叠氮还原,脱除氮原子上连接基团的反应,得到3‑氨基吡咯烷盐酸盐。该合成方法采用一锅法,步骤一结束不需要后处理,反应液可直接转移到反应釜中,进行步骤二的反应,反应路线简单,实验条件宽松,同时环境友好,同时实验操作简单、成本较低。公开号:CN112094215B
-
作为产物:描述:3-(Benzylamino)prop-2-enoic acid 在 sodium acetate 、 乙酸酐 作用下, 反应 1.0h, 以80%的产率得到N-苄基马来酰亚胺参考文献:名称:自挥发性聚合物的可变温度触发的热不稳定端盖小文库的制备和表征摘要:呋喃与马来酰亚胺之间的反应已越来越成为一种令人感兴趣的方法,因为它的可逆性使其成为自修复材料,自毁聚合物,细胞培养用水凝胶和骨修复制剂等应用的有用工具。然而,这些应用中的大多数都依赖于简单的单取代呋喃和简单的马来酰亚胺,并且还没有广泛评估通过简单的底物修饰可实现的固有的潜在热变异性。准备了一个适合上述应用的小型环加合物库,并通过原位1确定了Diels-Alder逆过程的温度依赖性。1 H NMR分析辅以计算计算。所报道的系统的实际温度范围为40至> 110°C。环还原反应比基于材料前沿分子轨道分析所预期的简单趋势所预期的复杂得多。DOI:10.1021/acs.joc.8b00135
-
作为试剂:描述:2H-1,3-benzoxazine-3-(4H)-acetonitrile 在 N-苄基马来酰亚胺 、 三氟甲磺酸三甲基硅酯 、 N,N-二异丙基乙胺 作用下, 以 甲苯 为溶剂, 以42%的产率得到[[(2-hydroxyphenyl)methyl]imino]diacetonitrile参考文献:名称:A New General Route to Novel Azomethine Ylides for Cycloaddition Reactions摘要:DOI:10.3987/com-02-9484
文献信息
-
Synthesis of Azabicyclo[3.1.0]amine Analogues of Anacardic Acid as Potent Antibacterial Agents作者:Ravi Kumar Vempati、N.S. Reddy、Srinivasa Rao Alapati、P.K. DubeyDOI:10.14233/ajchem.2013.13338日期:——Azabicyclo[3.1.0]amine analogues of anacardic acid (16a, 16b, 18a, 18b, 19 and 19b) were synthesized from anacardic acid and tested for their antibacterial activity against Gram positive and Gram negative bacteria. Most of the compounds are having potency at par with ampicillin and inferior with other standard drugs.
-
Copper-Catalyzed Intermolecular Thioamination of Maleimides with Thiols and Formamides: A One-Step Construction of 3-Amino-4-thiomaleimides Using Formamides as Nitrogen Sources作者:Sheng-Yin Zhao、Zhen-Hua Yang、Jia-Nan Zhu、Ze-Hui Jin、Jian ZhengDOI:10.1055/s-0037-1610536日期:2018.12Abstract A highly efficient copper-catalyzed intermolecular C(sp2)–H thioamination of maleimides with thiols and formamides in the presence of fluoroboric acid is reported using various readily available formamides as nitrogen sources and solvents. A diverse range of 3-amino-4-thiomaleimides is obtained with good yields under mild conditions, involving C–N and C–S bond formation. This methodology enriches摘要 据报道,在氟硼酸存在下,使用各种现成的甲酰胺作为氮源和溶剂,高效铜催化马来酰亚胺与硫醇和甲酰胺的分子间C(sp 2)–H硫胺化反应。在温和条件下,涉及C–N和C–S键的形成,可以得到各种多样的3-氨基-4-硫代马来酰亚胺,并具有良好的收率。这种方法学丰富了当前的C–N和C–S键形成化学,并且具有操作简便和出色的官能团耐受性的特点。 据报道,在氟硼酸存在下,使用各种现成的甲酰胺作为氮源和溶剂,高效铜催化马来酰亚胺与硫醇和甲酰胺的分子间C(sp 2)–H硫胺化反应。在温和条件下,涉及C–N和C–S键的形成,可以得到各种多样的3-氨基-4-硫代马来酰亚胺,并具有良好的收率。这种方法学丰富了当前的C–N和C–S键形成化学,并且具有操作简便和出色的官能团耐受性的特点。
-
[EN] COMPOUNDS<br/>[FR] COMPOSÉS申请人:GLAXOSMITHKLINE IP DEV LTD公开号:WO2018137593A1公开(公告)日:2018-08-02Provided are novel compounds that inhibit LRRK2 kinase activity, processes for their preparation, compositions containing them and their use in the treatment of or prevention of diseases associated with or characterized by LRRK2 kinase activity, for example Parkinson's disease, Alzheimer's disease and amyotrophic lateral sclerosis (ALS).提供了抑制LRRK2激酶活性的新化合物,以及它们的制备方法、含有它们的组合物以及它们在治疗或预防与LRRK2激酶活性相关或以其为特征的疾病中的用途,例如帕金森病、阿尔茨海默病和肌萎缩侧索硬化症(ALS)。
-
Acid/Phosphide-Induced Radical Route to Alkyl and Alkenyl Sulfides and Phosphonothioates from Sodium Arylsulfinates in Water作者:Ya-mei Lin、Guo-ping Lu、Gui-xiang Wang、Wen-bin YiDOI:10.1021/acs.joc.6b02459日期:2017.1.6can in situ generate arylsulfenyl radicals. These radicals have high reactivity to react with alkynes, alkenes, and H-phosphine oxides for the synthesis of alkyl and alkenyl sulfides and phosphonothioates. The control experiments and quantum calculations are also performed to gain insights into the generation mechanism of arylsulfenyl radicals. Notably, the chemistry is free of thiol odors, organic solvents
-
[EN] DOPAMINE D3 RECEPTOR ANTAGONISTS HAVING A BICYCLO MOIETY<br/>[FR] ANTAGONISTES DU RÉCEPTEUR D3 DE LA DOPAMINE AYANT UN FRAGMENT BICYCLO申请人:INDIVIOR UK LTD公开号:WO2017021920A1公开(公告)日:2017-02-09The disclosure provides compounds having formula (I), wherein the substituents are as defined herein. The compounds are useful for modulating the dopamine D3 receptor and for treating conditions associated therewith, such as addictions, drug dependency, and psychiatric conditions.
表征谱图
-
氢谱1HNMR
-
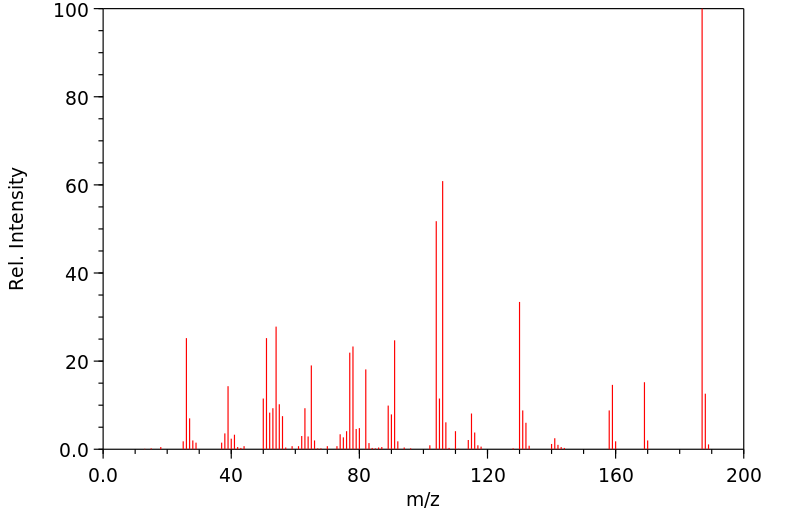
质谱MS
-
碳谱13CNMR
-
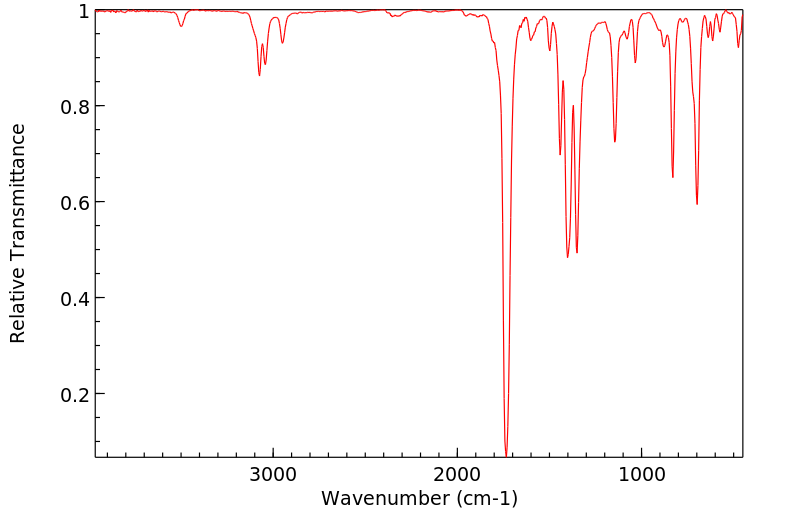
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(5R,Z)-3-(羟基((1R,2S,6S,8aS)-1,3,6-三甲基-2-((E)-prop-1-en-1-yl)-1,2,4a,5,6,7,8,8a-八氢萘-1-基)亚甲基)-5-(羟甲基)-1-甲基吡咯烷-2,4-二酮
(2R,2''R)-(-)-2,2''-联吡咯烷
麦角甾-7,22-二烯-3-基亚油酸酯
马来酰亚胺霉素
马来酰亚胺基酰肼盐酸盐
马来酰亚胺基甲基-3-马来酰亚胺基丙酸酯
马来酰亚胺丙酰基-dPEG4-NHS
马来酰亚胺-酰胺-PEG6-琥珀酰亚胺酯
马来酰亚胺-酰胺-PEG6-丙酸
马来酰亚胺-酰胺-PEG24-丙酸
马来酰亚胺-酰胺-PEG12-丙酸
马来酰亚胺-四聚乙二醇-羧酸
马来酰亚胺-四聚乙二醇-丙酸叔丁酯
马来酰亚胺-四聚乙二醇-丙烯酸琥珀酰亚胺酯
马来酰亚胺-六聚乙二醇-羧酸
马来酰亚胺-六聚乙二醇-丙酸叔丁酯
马来酰亚胺-八聚乙二醇-丙酸叔丁酯
马来酰亚胺-二聚乙二醇-丙酸叔丁酯
马来酰亚胺-三(乙烯乙二醇)-丙酸
马来酰亚胺-一聚乙二醇-羧酸
马来酰亚胺-一聚乙二醇-丙烯酸琥珀酰亚胺酯
马来酰亚胺-PEG3-羟基
马来酰亚胺-PEG2-胺三氟醋酸盐
马来酰亚胺-PEG2-琥珀酰亚胺酯
马来酰亚胺
频哪醇硼酸酯
顺式草酸双(-3,8-二氮杂双环[4.2.0]辛烷-8-羧酸叔丁酯)
顺式4-甲基吡咯烷酮-3-醇盐酸盐
顺式4-氟吡咯烷酮-3-醇盐酸盐
顺式3,4-二羟基吡咯烷盐酸盐
顺式3,4-二氨基吡咯烷-1-羧酸叔丁酯
顺式-二甲基 1-苄基吡咯烷-3,4-二羧酸
顺式-N-[2-(2,6-二甲基-1-哌啶基)乙基]-2-氧代-4-苯基-1-吡咯烷乙酰胺
顺式-N-Boc-吡咯烷-3,4-二羧酸
顺式-5-苄基-2-叔丁氧羰基六氢吡咯并[3,4-c]吡咯
顺式-5-甲基-1H-六氢吡咯并[3,4-b]吡咯二盐酸盐
顺式-5-氧代六氢环戊二烯并[c]吡咯-2(1H)-羧酸叔丁酯
顺式-5-乙氧羰基-1H-六氢吡咯并[3,4-B]吡咯盐酸盐
顺式-5-(碘甲基)-4-苯基-2-吡咯烷酮
顺式-5-(碘甲基)-4-甲基-2-吡咯烷酮
顺式-4-氧代-六氢-吡咯并[3,4-C]吡咯-2-甲酸叔丁酯
顺式-3-氟-4-羟基吡咯烷-1-羧酸叔丁酯
顺式-3-氟-4-甲基吡咯烷盐酸盐
顺式-2-甲基六氢吡咯并[3,4-c]吡咯
顺式-2,5-二甲基吡咯烷
顺式-1-苄基-3,4-吡咯烷二甲酸二乙酯
顺式-1-甲基六氢吡咯并[3,4-b]吡咯
顺式-(9CI)-3,4-二乙烯-1-(三氟乙酰基)-吡咯烷
顺-八氢环戊[c]吡咯-5-酮盐酸盐
非星匹宁