4-(2-硝基苯基)丁-3-烯-2-酮 | 20766-40-9
中文名称
4-(2-硝基苯基)丁-3-烯-2-酮
中文别名
——
英文名称
(E)-4-(2-nitrophenyl)but-3-en-2-one
英文别名
4-(2-Nitrophenyl)but-3-en-2-one
CAS
20766-40-9
化学式
C10H9NO3
mdl
——
分子量
191.186
InChiKey
FRPAGLRAZLFTGW-VOTSOKGWSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
计算性质
-
辛醇/水分配系数(LogP):1.7
-
重原子数:14
-
可旋转键数:2
-
环数:1.0
-
sp3杂化的碳原子比例:0.1
-
拓扑面积:62.9
-
氢给体数:0
-
氢受体数:3
SDS
上下游信息
反应信息
-
作为反应物:描述:参考文献:名称:光环化反应。第8部分†。通过光反应反式顺式异构化合成2-喹诺酮,喹啉和香豆素衍生物摘要:通过traans-o-氨基肉桂酰基衍生物trans - 1和trans-的光环化反应合成了2-喹诺酮2,喹啉3,香豆素(2 H -1-苯并吡喃-2-酮)5和2 H -1-苯并吡喃半缩醛6。ø -hydroxycinnamoyl衍生物反式- 4。反应通过反式-顺式异构化进行,然后进行分子内环化。DOI:10.1002/jhet.5570390108
-
作为产物:参考文献:名称:烯烃转化为硝基烯烃的有用方法摘要:硝酸三氟甘油很容易从CH 2 Cl 2溶液中的硝酸四正丁铵中生成,并作为有效的硝化剂,适用于各种不饱和底物以形成硝基烯烃。DOI:10.1021/acs.orglett.1c00868
文献信息
-
Synthesis of 2-Substituted Quinolines from 2-Aminostyryl Ketones Using Iodide as a Catalyst作者:So Young Lee、Jiye Jeon、Cheol-Hong CheonDOI:10.1021/acs.joc.8b00552日期:2018.5.4A new protocol for the synthesis of 2-substituted quinolines from 2-aminostyryl ketones has been developed using iodide as a nucleophilic catalyst. Conjugate addition of iodide to 2-aminostyryl ketones yielded the corresponding β-iodoketones, which could have a conformation where the amino and carbonyl groups are proximal through free rotation about the Cα–Cβ single bond. Subsequent condensation between
-
On-Water Synthesis of 2-Substituted Quinolines from 2-Aminochalcones Using Benzylamine as the Nucleophilic Catalyst作者:So Young Lee、Cheol-Hong CheonDOI:10.1021/acs.joc.8b01675日期:2018.11.2On-water synthesis of 2-substituted quinolines from 2-aminochalcone derivatives was developed using benzylamine as the nucleophilic catalyst. Various 2-aminochalcones could be applied to this protocol, and the desired 2-substituted quinoline products were isolated in excellent yields by simple filtration. Furthermore, we elucidated the role of benzylamine in this transformation and provided the detailed
-
Access to Spirocyclic Benzothiophenones with Multiple Stereocenters via an Organocatalytic Cascade Reaction作者:Bedřich Formánek、Jiří Tauchman、Ivana Císařová、Jan VeselýDOI:10.1021/acs.joc.0c00882日期:2020.7.2derivatives containing three stereocenters were prepared via one-step synthesis in yields ranging from 88 to 96% and in enantioselectivities (enantiomeric excess (ee)) ranging from 85 to 97%, with diastereoselectivities of approximately 14/2/1. Therefore, this method provides an efficient route for the synthesis of a new class of optically active 2-spirobenzothiophenones.
-
PPh<sub>3</sub>⋅HBr-DMSO Mediated Expedient Synthesis of γ-Substituted β,γ-Unsaturated α-Ketomethylthioesters and α-Bromo Enals: Application to the Synthesis of 2-Methylsulfanyl-3(<i>2 H</i>)-furanones作者:Kanchan Mal、Abhinandan Sharma、Prakas R. Maulik、Indrajit DasDOI:10.1002/chem.201303755日期:2014.1.13An efficient chemoselective general procedure for the synthesis of γ‐substituted β,γ‐unsaturated α‐ketomethylthioesters from α,β‐unsaturated ketones has been achieved through an unprecedented PPh3⋅HBr‐DMSO mediated oxidative bromination and Kornblum oxidation sequence. The newly developed reagent system serves admirably for the synthesis of α‐bromoenals from enals. Furthermore, AuCl3‐catalyzed efficient
-
Blue-light-promoted carbon–carbon double bond isomerization and its application in the syntheses of quinolines作者:Xinzheng Chen、Shuxian Qiu、Sasa Wang、Huifei Wang、Hongbin ZhaiDOI:10.1039/c7ob00558j日期:——A blue-light-promoted carbon-carbon double bond isomerization in the absence of any photoredox catalyst is reported. It provides rapid access to a series of quinolines in good to excellent yields under simple aerobic conditions. The protocol is direct, catalyst-free and operationally convenient.据报道在没有任何光氧化还原催化剂的情况下,蓝光促进的碳-碳双键异构化。在简单的有氧条件下,它可以快速获得一系列喹啉,收率好至极好。该方案是直接,无催化剂且操作方便的。
表征谱图
-
氢谱1HNMR
-
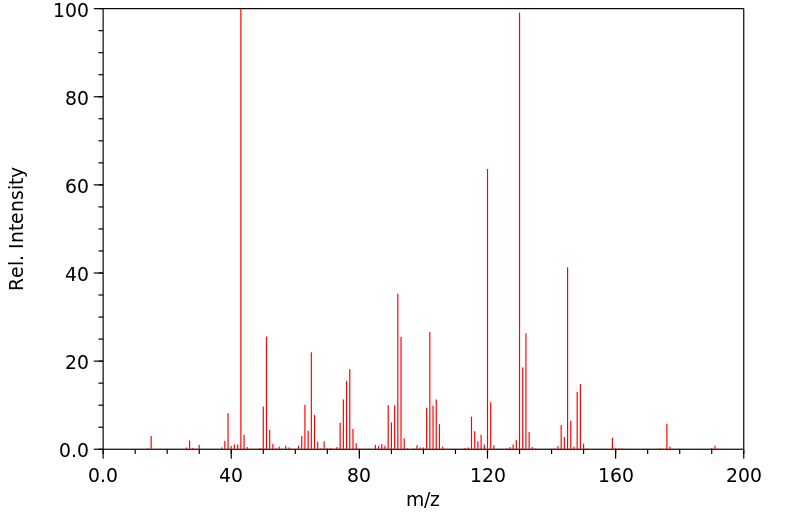
质谱MS
-
碳谱13CNMR
-
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(βS)-β-氨基-4-(4-羟基苯氧基)-3,5-二碘苯甲丙醇
(S,S)-邻甲苯基-DIPAMP
(S)-(-)-7'-〔4(S)-(苄基)恶唑-2-基]-7-二(3,5-二-叔丁基苯基)膦基-2,2',3,3'-四氢-1,1-螺二氢茚
(S)-盐酸沙丁胺醇
(S)-3-(叔丁基)-4-(2,6-二甲氧基苯基)-2,3-二氢苯并[d][1,3]氧磷杂环戊二烯
(S)-2,2'-双[双(3,5-三氟甲基苯基)膦基]-4,4',6,6'-四甲氧基联苯
(S)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(R)富马酸托特罗定
(R)-(-)-盐酸尼古地平
(R)-(-)-4,12-双(二苯基膦基)[2.2]对环芳烷(1,5环辛二烯)铑(I)四氟硼酸盐
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[((6-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(4-叔丁基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(3-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-4,7-双(3,5-二-叔丁基苯基)膦基-7“-[(吡啶-2-基甲基)氨基]-2,2”,3,3'-四氢1,1'-螺二茚满
(R)-3-(叔丁基)-4-(2,6-二苯氧基苯基)-2,3-二氢苯并[d][1,3]氧杂磷杂环戊烯
(R)-2-[((二苯基膦基)甲基]吡咯烷
(R)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(N-(4-甲氧基苯基)-N-甲基-3-(1-哌啶基)丙-2-烯酰胺)
(5-溴-2-羟基苯基)-4-氯苯甲酮
(5-溴-2-氯苯基)(4-羟基苯基)甲酮
(5-氧代-3-苯基-2,5-二氢-1,2,3,4-oxatriazol-3-鎓)
(4S,5R)-4-甲基-5-苯基-1,2,3-氧代噻唑烷-2,2-二氧化物-3-羧酸叔丁酯
(4S,4''S)-2,2''-亚环戊基双[4,5-二氢-4-(苯甲基)恶唑]
(4-溴苯基)-[2-氟-4-[6-[甲基(丙-2-烯基)氨基]己氧基]苯基]甲酮
(4-丁氧基苯甲基)三苯基溴化磷
(3aR,8aR)-(-)-4,4,8,8-四(3,5-二甲基苯基)四氢-2,2-二甲基-6-苯基-1,3-二氧戊环[4,5-e]二恶唑磷
(3aR,6aS)-5-氧代六氢环戊基[c]吡咯-2(1H)-羧酸酯
(2Z)-3-[[(4-氯苯基)氨基]-2-氰基丙烯酸乙酯
(2S,3S,5S)-5-(叔丁氧基甲酰氨基)-2-(N-5-噻唑基-甲氧羰基)氨基-1,6-二苯基-3-羟基己烷
(2S,2''S,3S,3''S)-3,3''-二叔丁基-4,4''-双(2,6-二甲氧基苯基)-2,2'',3,3''-四氢-2,2''-联苯并[d][1,3]氧杂磷杂戊环
(2S)-(-)-2-{[[[[3,5-双(氟代甲基)苯基]氨基]硫代甲基]氨基}-N-(二苯基甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[((1S,2S)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[[((1R,2R)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2-硝基苯基)磷酸三酰胺
(2,6-二氯苯基)乙酰氯
(2,3-二甲氧基-5-甲基苯基)硼酸
(1S,2S,3S,5S)-5-叠氮基-3-(苯基甲氧基)-2-[(苯基甲氧基)甲基]环戊醇
(1S,2S,3R,5R)-2-(苄氧基)甲基-6-氧杂双环[3.1.0]己-3-醇
(1-(4-氟苯基)环丙基)甲胺盐酸盐
(1-(3-溴苯基)环丁基)甲胺盐酸盐
(1-(2-氯苯基)环丁基)甲胺盐酸盐
(1-(2-氟苯基)环丙基)甲胺盐酸盐
(1-(2,6-二氟苯基)环丙基)甲胺盐酸盐
(-)-去甲基西布曲明
龙蒿油
龙胆酸钠
龙胆酸叔丁酯
龙胆酸
龙胆紫-d6
龙胆紫