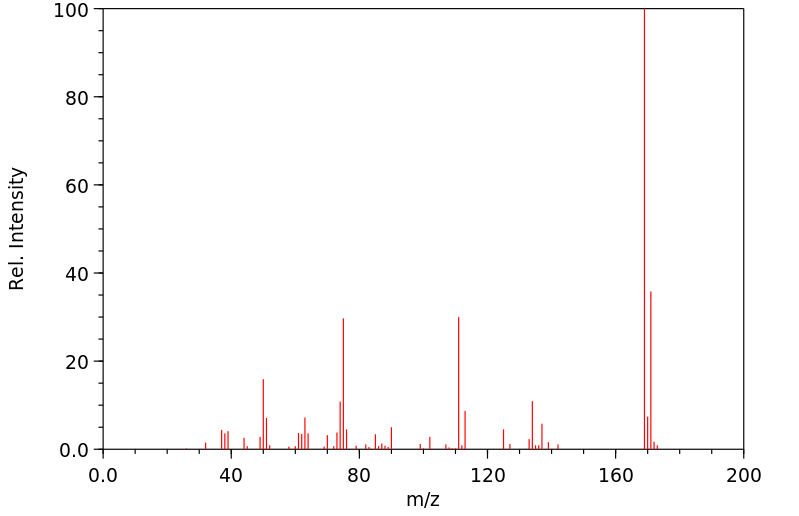
| Name: | 1-(2-Chlorophenyl)-2-thiourea 98% Material Safety Data Sheet |
| Synonym: | |
| CAS: | 5344-82-1 |
Section 1 - Chemical Product MSDS Name:1-(2-Chlorophenyl)-2-thiourea 98% Material Safety Data Sheet
Synonym:
Section 2 - COMPOSITION, INFORMATION ON INGREDIENTS | CAS# | Chemical Name | content | EINECS# |
| 5344-82-1 | 1-(2-Chlorophenyl)-2-thiourea | 98% | 226-291-6 |
Hazard Symbols: T+
Risk Phrases: 28
Section 3 - HAZARDS IDENTIFICATION EMERGENCY OVERVIEW
Very toxic if swallowed.
Potential Health Effects
Eye:
May cause eye irritation.
Skin:
May cause skin irritation. May be harmful if absorbed through the skin.
Ingestion:
May be fatal if swallowed. May cause irritation of the digestive tract.
Inhalation:
May cause respiratory tract irritation. May be harmful if inhaled.
Chronic:
Not available.
Section 4 - FIRST AID MEASURES Eyes: Immediately flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid immediately.
Skin:
Get medical aid immediately. Immediately flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes.
Ingestion:
Get medical aid immediately. Wash mouth out with water.
Inhalation:
Get medical aid immediately. Remove from exposure and move to fresh air immediately. If not breathing, give artificial respiration. If breathing is difficult, give oxygen.
Notes to Physician:
Section 5 - FIRE FIGHTING MEASURES General Information:
As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear.
Extinguishing Media:
Use water spray, dry chemical, carbon dioxide, or chemical foam.
Section 6 - ACCIDENTAL RELEASE MEASURES General Information: Use proper personal protective equipment as indicated in Section 8.
Spills/Leaks:
Vacuum or sweep up material and place into a suitable disposal container.
Section 7 - HANDLING and STORAGE Handling:
Do not breathe dust, vapor, mist, or gas. Do not get in eyes, on skin, or on clothing. Use only in a chemical fume hood.
Storage:
Store in a cool, dry place. Store in a tightly closed container.
Poison room locked.
Section 8 - EXPOSURE CONTROLS, PERSONAL PROTECTION Engineering Controls:
Use adequate ventilation to keep airborne concentrations low.
Exposure Limits CAS# 5344-82-1: Personal Protective Equipment Eyes: Not available.
Skin:
Wear appropriate protective gloves to prevent skin exposure.
Clothing:
Wear appropriate protective clothing to prevent skin exposure.
Respirators:
Follow the OSHA respirator regulations found in 29 CFR 1910.134 or European Standard EN 149. Use a NIOSH/MSHA or European Standard EN 149 approved respirator if exposure limits are exceeded or if irritation or other symptoms are experienced.
Section 9 - PHYSICAL AND CHEMICAL PROPERTIES Physical State: Powder
Color: white to tan
Odor: Not available.
pH: Not available.
Vapor Pressure: Not available.
Viscosity: Not available.
Boiling Point: Not available.
Freezing/Melting Point: 143 - 146 deg C
Autoignition Temperature: Not available.
Flash Point: Not available.
Explosion Limits, lower: Not available.
Explosion Limits, upper: Not available.
Decomposition Temperature:
Solubility in water:
Specific Gravity/Density:
Molecular Formula: C7H7ClN2S
Molecular Weight: 186.66
Section 10 - STABILITY AND REACTIVITY Chemical Stability:
Not available.
Conditions to Avoid:
Incompatible materials.
Incompatibilities with Other Materials:
Strong oxidizing agents, strong acids, strong bases.
Hazardous Decomposition Products:
Hydrogen chloride, nitrogen oxides, carbon monoxide, carbon dioxide, sulfur oxides (SOx), including sulfur oxide and sulfur dioxide.
Hazardous Polymerization: Has not been reported
Section 11 - TOXICOLOGICAL INFORMATION RTECS#:
CAS# 5344-82-1: YS7100000 LD50/LC50:
CAS# 5344-82-1: Oral, rat: LD50 = 4600 ug/kg.
Carcinogenicity:
1-(2-Chlorophenyl)-2-thiourea - Not listed by ACGIH, IARC, or NTP.
Other:
See actual entry in RTECS for complete information.
Section 12 - ECOLOGICAL INFORMATION
Section 13 - DISPOSAL CONSIDERATIONS Dispose of in a manner consistent with federal, state, and local regulations.
Section 14 - TRANSPORT INFORMATION IATA
Shipping Name: TOXIC SOLID, ORGANIC, N.O.S.*
Hazard Class: 6.1
UN Number: 2811
Packing Group: II
IMO
Shipping Name: TOXIC SOLID, ORGANIC, N.O.S.
Hazard Class: 6.1
UN Number: 2811
Packing Group: II
RID/ADR
Shipping Name: TOXIC SOLID, ORGANIC, N.O.S.
Hazard Class: 6.1
UN Number: 2811
Packing group: II
USA RQ: CAS# 5344-82-1: 100 lb final RQ; 45.4 kg final RQ
Section 15 - REGULATORY INFORMATION European/International Regulations
European Labeling in Accordance with EC Directives
Hazard Symbols: T+
Risk Phrases:
R 28 Very toxic if swallowed.
Safety Phrases:
S 1 Keep locked up.
S 28A After contact with skin, wash immediately with
plenty of water.
S 45 In case of accident or if you feel unwell, seek
medical advice immediately (show the label where
possible).
WGK (Water Danger/Protection)
CAS# 5344-82-1: No information available.
Canada
CAS# 5344-82-1 is listed on Canada's NDSL List.
CAS# 5344-82-1 is not listed on Canada's Ingredient Disclosure List.
US FEDERAL
TSCA
CAS# 5344-82-1 is listed on the TSCA inventory.
SECTION 16 - ADDITIONAL INFORMATION N/A