2-丁炔酸甲酯 | 23326-27-4
中文名称
2-丁炔酸甲酯
中文别名
丁炔酸甲酯
英文名称
Methyl 2-butynoate
英文别名
methyl tetrolate;methyl but-2-ynoate
CAS
23326-27-4
化学式
C5H6O2
mdl
——
分子量
98.1014
InChiKey
UJQCANQILFWSDJ-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
沸点:76-77 °C (80 mmHg)
-
密度:0.98
-
闪点:45 °C
-
稳定性/保质期:
在常温常压下稳定,应避免接触强氧化剂。
计算性质
-
辛醇/水分配系数(LogP):1.1
-
重原子数:7
-
可旋转键数:1
-
环数:0.0
-
sp3杂化的碳原子比例:0.4
-
拓扑面积:26.3
-
氢给体数:0
-
氢受体数:2
安全信息
-
危险等级:3.2
-
危险品标志:Xn
-
安全说明:S16
-
危险类别码:R10
-
海关编码:29161900
-
危险品运输编号:1993
-
危险类别:3.2
-
包装等级:III
-
危险性防范说明:P210,P233,P240,P241+P242+P243,P264,P280,P302+P352+P332+P313+P362+P364,P305+P351+P338+P337+P313,P370+P378,P403+P235,P501
-
危险性描述:H225,H315,H319
-
储存条件:将物品存放在紧密封装的容器中,并储存在阴凉、干燥的地方。
SDS
丁炔酸甲酯 修改号码:5
模块 1. 化学品
产品名称: Methyl Tetrolate
修改号码: 5
模块 2. 危险性概述
GHS分类
物理性危害
易燃液体 第3级
健康危害
皮肤腐蚀/刺激 第2级
严重损伤/刺激眼睛 2A类
环境危害 未分类
GHS标签元素
图标或危害标志
信号词 警告
危险描述 易燃液体和蒸气
造成皮肤刺激
造成严重眼刺激
防范说明
[预防] 远离热源/火花/明火/热表面。禁烟。
保持容器密闭。
使用防爆的电气/通风/照明设备。采取预防措施以防静电和火花引起的着火。
处理后要彻底清洗双手。
穿戴防护手套/护目镜/防护面具。
[急救措施] 眼睛接触:用水小心清洗几分钟。如果方便,易操作,摘除隐形眼镜。继续冲洗。
眼睛接触:求医/就诊
皮肤接触:立即去除/脱掉所有被污染的衣物。用水清洗皮肤/淋浴。
若皮肤刺激:求医/就诊。
脱掉被污染的衣物,清洗后方可重新使用。
[储存] 存放于通风良好处。保持凉爽。
[废弃处置] 根据当地政府规定把物品/容器交与工业废弃处理机构。
丁炔酸甲酯 修改号码:5
模块 3. 成分/组成信息
单一物质/混和物 单一物质
化学名(中文名): 丁炔酸甲酯
百分比: >97.0%(GC)
CAS编码: 23326-27-4
俗名: 2-Butynoic Acid Methyl Ester , Methyl 2-Butynoate , Tetrolic Acid Methyl
Ester
分子式: C5H6O2
模块 4. 急救措施
吸入: 将受害者移到新鲜空气处,保持呼吸通畅,休息。若感不适请求医/就诊。
皮肤接触: 立即去除/脱掉所有被污染的衣物。用大量肥皂和水轻轻洗。
若皮肤刺激或发生皮疹:求医/就诊。
眼睛接触: 用水小心清洗几分钟。如果方便,易操作,摘除隐形眼镜。继续清洗。
如果眼睛刺激:求医/就诊。
食入: 若感不适,求医/就诊。漱口。
紧急救助者的防护: 救援者需要穿戴个人防护用品,比如橡胶手套和气密性护目镜。
模块 5. 消防措施
合适的灭火剂: 干粉,泡沫,雾状水,二氧化碳
特定方法: 从上风处灭火,根据周围环境选择合适的灭火方法。
非相关人员应该撤离至安全地方。
周围一旦着火:喷水,保持容器冷却。如果安全,消除一切火源。
消防员的特殊防护用具: 灭火时,一定要穿戴个人防护用品。
模块 6. 泄漏应急处理
个人防护措施,防护用具, 使用个人防护用品。远离溢出物/泄露处并处在上风处。确保足够通风。
紧急措施: 泄露区应该用安全带等圈起来,控制非相关人员进入。
环保措施: 防止进入下水道。
控制和清洗的方法和材料: 回收到密闭容器前用干砂或惰性吸收剂吸收泄漏物。一旦大量泄漏,筑堤控制。附着
物或收集物应该根据相关法律法规废弃处置。
副危险性的防护措施 移除所有火源。一旦发生火灾应该准备灭火器。使用防火花工具和防爆设备。
模块 7. 操作处置与储存
处理
技术措施: 在通风良好处进行处理。穿戴合适的防护用具。防止烟雾产生。远离热源/火花/明火
/热表面。禁烟。采取措施防止静电积累。使用防爆设备。处理后彻底清洗双手和脸。
注意事项: 如果可能,使用封闭系统。如果蒸气或浮质产生,使用通风、局部排气。
操作处置注意事项: 避免接触皮肤、眼睛和衣物。
贮存
储存条件: 保持容器密闭。存放于凉爽、阴暗、通风良好处。
存放于惰性气体环境中。
远离不相容的材料比如氧化剂存放。
气敏
包装材料: 依据法律。
模块 8. 接触控制和个体防护
工程控制: 尽可能安装封闭体系或局部排风系统。同时安装淋浴器和洗眼器。
个人防护用品
呼吸系统防护: 防毒面具。依据当地和政府法规。
丁炔酸甲酯 修改号码:5
模块 8. 接触控制和个体防护
手部防护: 防护手套。
眼睛防护: 安全防护镜。如果情况需要,佩戴面具。
皮肤和身体防护: 防护服。如果情况需要,穿戴防护靴。
模块 9. 理化特性
液体
外形(20°C):
外观: 透明
颜色: 无色-微浅黄色
气味: 无资料
pH: 无数据资料
熔点: 无资料
沸点/沸程 148 °C
闪点: 45°C
爆炸特性
爆炸下限: 无资料
爆炸上限: 无资料
密度: 1.00
溶解度:
[水] 无资料
[其他溶剂] 无资料
模块 10. 稳定性和反应性
化学稳定性: 一般情况下稳定。
危险反应的可能性: 未报道特殊反应性。
避免接触的条件: 火花, 明火, 静电
须避免接触的物质 氧化剂
危险的分解产物: 一氧化碳, 二氧化碳
模块 11. 毒理学信息
急性毒性: 无资料
对皮肤腐蚀或刺激: 无资料
对眼睛严重损害或刺激: 无资料
生殖细胞变异原性: 无资料
致癌性:
IARC = 无资料
NTP = 无资料
生殖毒性: 无资料
模块 12. 生态学信息
生态毒性:
鱼类: 无资料
甲壳类: 无资料
藻类: 无资料
残留性 / 降解性: 无资料
潜在生物累积 (BCF): 无资料
土壤中移动性
log水分配系数: 无资料
土壤吸收系数 (Koc): 无资料
亨利定律 无资料
constaNT(PaM3/mol):
丁炔酸甲酯 修改号码:5
模块 13. 废弃处置
如果可能,回收处理。请咨询当地管理部门。建议在装有后燃和洗涤装置的化学焚烧炉中焚烧。废弃处置时请遵守
国家、地区和当地的所有法规。
模块 14. 运输信息
联合国分类: 第3类 易燃液体 。
UN编号: 3272
正式运输名称: 酯类, 不另作详细说明
包装等级: III
模块 15. 法规信息
《危险化学品安全管理条例》(2002年1月26日国务院发布,2011年2月16日修订): 针对危险化学品的安全使用、
生产、储存、运输、装卸等方面均作了相应的规定。
模块16 - 其他信息
N/A
模块 1. 化学品
产品名称: Methyl Tetrolate
修改号码: 5
模块 2. 危险性概述
GHS分类
物理性危害
易燃液体 第3级
健康危害
皮肤腐蚀/刺激 第2级
严重损伤/刺激眼睛 2A类
环境危害 未分类
GHS标签元素
图标或危害标志
信号词 警告
危险描述 易燃液体和蒸气
造成皮肤刺激
造成严重眼刺激
防范说明
[预防] 远离热源/火花/明火/热表面。禁烟。
保持容器密闭。
使用防爆的电气/通风/照明设备。采取预防措施以防静电和火花引起的着火。
处理后要彻底清洗双手。
穿戴防护手套/护目镜/防护面具。
[急救措施] 眼睛接触:用水小心清洗几分钟。如果方便,易操作,摘除隐形眼镜。继续冲洗。
眼睛接触:求医/就诊
皮肤接触:立即去除/脱掉所有被污染的衣物。用水清洗皮肤/淋浴。
若皮肤刺激:求医/就诊。
脱掉被污染的衣物,清洗后方可重新使用。
[储存] 存放于通风良好处。保持凉爽。
[废弃处置] 根据当地政府规定把物品/容器交与工业废弃处理机构。
丁炔酸甲酯 修改号码:5
模块 3. 成分/组成信息
单一物质/混和物 单一物质
化学名(中文名): 丁炔酸甲酯
百分比: >97.0%(GC)
CAS编码: 23326-27-4
俗名: 2-Butynoic Acid Methyl Ester , Methyl 2-Butynoate , Tetrolic Acid Methyl
Ester
分子式: C5H6O2
模块 4. 急救措施
吸入: 将受害者移到新鲜空气处,保持呼吸通畅,休息。若感不适请求医/就诊。
皮肤接触: 立即去除/脱掉所有被污染的衣物。用大量肥皂和水轻轻洗。
若皮肤刺激或发生皮疹:求医/就诊。
眼睛接触: 用水小心清洗几分钟。如果方便,易操作,摘除隐形眼镜。继续清洗。
如果眼睛刺激:求医/就诊。
食入: 若感不适,求医/就诊。漱口。
紧急救助者的防护: 救援者需要穿戴个人防护用品,比如橡胶手套和气密性护目镜。
模块 5. 消防措施
合适的灭火剂: 干粉,泡沫,雾状水,二氧化碳
特定方法: 从上风处灭火,根据周围环境选择合适的灭火方法。
非相关人员应该撤离至安全地方。
周围一旦着火:喷水,保持容器冷却。如果安全,消除一切火源。
消防员的特殊防护用具: 灭火时,一定要穿戴个人防护用品。
模块 6. 泄漏应急处理
个人防护措施,防护用具, 使用个人防护用品。远离溢出物/泄露处并处在上风处。确保足够通风。
紧急措施: 泄露区应该用安全带等圈起来,控制非相关人员进入。
环保措施: 防止进入下水道。
控制和清洗的方法和材料: 回收到密闭容器前用干砂或惰性吸收剂吸收泄漏物。一旦大量泄漏,筑堤控制。附着
物或收集物应该根据相关法律法规废弃处置。
副危险性的防护措施 移除所有火源。一旦发生火灾应该准备灭火器。使用防火花工具和防爆设备。
模块 7. 操作处置与储存
处理
技术措施: 在通风良好处进行处理。穿戴合适的防护用具。防止烟雾产生。远离热源/火花/明火
/热表面。禁烟。采取措施防止静电积累。使用防爆设备。处理后彻底清洗双手和脸。
注意事项: 如果可能,使用封闭系统。如果蒸气或浮质产生,使用通风、局部排气。
操作处置注意事项: 避免接触皮肤、眼睛和衣物。
贮存
储存条件: 保持容器密闭。存放于凉爽、阴暗、通风良好处。
存放于惰性气体环境中。
远离不相容的材料比如氧化剂存放。
气敏
包装材料: 依据法律。
模块 8. 接触控制和个体防护
工程控制: 尽可能安装封闭体系或局部排风系统。同时安装淋浴器和洗眼器。
个人防护用品
呼吸系统防护: 防毒面具。依据当地和政府法规。
丁炔酸甲酯 修改号码:5
模块 8. 接触控制和个体防护
手部防护: 防护手套。
眼睛防护: 安全防护镜。如果情况需要,佩戴面具。
皮肤和身体防护: 防护服。如果情况需要,穿戴防护靴。
模块 9. 理化特性
液体
外形(20°C):
外观: 透明
颜色: 无色-微浅黄色
气味: 无资料
pH: 无数据资料
熔点: 无资料
沸点/沸程 148 °C
闪点: 45°C
爆炸特性
爆炸下限: 无资料
爆炸上限: 无资料
密度: 1.00
溶解度:
[水] 无资料
[其他溶剂] 无资料
模块 10. 稳定性和反应性
化学稳定性: 一般情况下稳定。
危险反应的可能性: 未报道特殊反应性。
避免接触的条件: 火花, 明火, 静电
须避免接触的物质 氧化剂
危险的分解产物: 一氧化碳, 二氧化碳
模块 11. 毒理学信息
急性毒性: 无资料
对皮肤腐蚀或刺激: 无资料
对眼睛严重损害或刺激: 无资料
生殖细胞变异原性: 无资料
致癌性:
IARC = 无资料
NTP = 无资料
生殖毒性: 无资料
模块 12. 生态学信息
生态毒性:
鱼类: 无资料
甲壳类: 无资料
藻类: 无资料
残留性 / 降解性: 无资料
潜在生物累积 (BCF): 无资料
土壤中移动性
log水分配系数: 无资料
土壤吸收系数 (Koc): 无资料
亨利定律 无资料
constaNT(PaM3/mol):
丁炔酸甲酯 修改号码:5
模块 13. 废弃处置
如果可能,回收处理。请咨询当地管理部门。建议在装有后燃和洗涤装置的化学焚烧炉中焚烧。废弃处置时请遵守
国家、地区和当地的所有法规。
模块 14. 运输信息
联合国分类: 第3类 易燃液体 。
UN编号: 3272
正式运输名称: 酯类, 不另作详细说明
包装等级: III
模块 15. 法规信息
《危险化学品安全管理条例》(2002年1月26日国务院发布,2011年2月16日修订): 针对危险化学品的安全使用、
生产、储存、运输、装卸等方面均作了相应的规定。
模块16 - 其他信息
N/A
制备方法与用途
合成制备方法
暂无相关描述。
用途暂无相关描述。
上下游信息
反应信息
-
作为反应物:参考文献:名称:(NH4)2S2O8-Mediated Diiodination of Alkynes with Iodide in Water: Stereospecific Synthesis of (E)-Diiodoalkenes摘要:A new approach to the stereospecific diiodination of alkynes under mild conditions has been developed. This protocol employs ammonium persulfate as an oxidant and iodide as an iodine source, and provides a highly efficient and general method for the preparation of a broad range of (E)-diiodoalkenes in water.DOI:10.1055/s-0034-1379910
-
作为产物:描述:[acetyl(methoxycarbonyl)methylene]triphenylphosphorane 以71%的产率得到2-丁炔酸甲酯参考文献:名称:Synthesis and Spectroscopic Characterization of 1-13C- and 4-13C-Plastoquinone-9摘要:DOI:10.1002/1099-0690(200207)2002:13<2094::aid-ejoc2094>3.0.co;2-e
-
作为试剂:描述:甲醇 、 3-bromo-2-phenylsulfinyl-1-propene 、 alkaline earth salt of/the/ methylsulfuric acid 在 2-丁炔酸甲酯 作用下, 以10%的产率得到phenyl isopropenyl sulfoxide参考文献:名称:Ni(0)促进的烯丙基卤化物和乙炔的环羰基化反应中的环戊烯酮的不对称方法摘要:报道了通过Ni(CO)4促进的炔-烯丙基卤化物环化-羰基化来不对称合成环戊烯酮的不同方法。乙炔亚砜1的使用已证明对合成无环,稠合[5 + 8]和螺[5 + 5],[5 + 7]和[5 + 8]环戊烯有效,而芳基亚砜的放置烯丙基体系上的环糊精无法提供任何环加合物。通过这种方法已经获得了手性稠合的[5 + 8],螺[5 + 7]和[5 + 8]环戊烯酮。最后,当在我们的反应体系中使用不同的同手性α-取代的羧酸盐作为配体时,观察到适度的对映选择性。DOI:10.1016/0040-4020(96)00573-x
文献信息
-
Regioselective Synthesis of 2,3,4-Trisubstituted Pyrroles via Pd(II)-Catalyzed Three-Component Cascade Reactions of Amines, Alkyne Esters, and Alkenes作者:Xu Zhang、Xuefeng Xu、Gong Chen、Wei YiDOI:10.1021/acs.orglett.6b02325日期:2016.10.7three-component cascade reaction of diverse amines, alkyne esters, and alkenes is disclosed for the direct synthesis of diverse 2,3,4-trisubstituted pyrroles with broad functional group tolerance and in good to excellent yields. This transformation is supposed to proceed through the cascade formation of C(sp2)–C(sp2) and C(sp2)–N bonds via Pd(II)-catalyzed regioselective alkene migratory insertion, intramolecular
-
Selective Synthesis of Dihydrophenanthridine and Phenanthridine Derivatives from the Cascade Reactions of <i>o</i>-Arylanilines with Alkynoates through C–H/N–H/C–C Bond Cleavage作者:Yuanshuang Xu、Caiyun Yu、Xinying Zhang、Xuesen FanDOI:10.1021/acs.joc.1c00256日期:2021.4.16unprecedented selective synthesis of dihydrophenanthridine and phenanthridine derivatives through the cascade reactions of 2-arylanilines with alkynoates is presented. Mechanistic studies showed that the formation of the dihydrophenanthridine scaffold involves an initial C(sp2)–H alkenylation of 2-arylaniline with alkynoate followed by an intramolecular aza-Michael addition. When this reaction is carried out
-
Rhodium-Catalyzed Regioselective Domino Azlactone-Alkyne Coupling/Aza-Cope Rearrangement: Facile Access to 2-Allyl-3-oxazolin-5-ones and Trisubstituted Pyridines作者:Jinqiang Kuang、Shaista Parveen、Bernhard BreitDOI:10.1002/anie.201704022日期:2017.7.10Rhodium‐catalyzed regioselective addition of azlactones to internal alkynes combined with aza‐Cope rearrangement provides efficient atom economic access to 2‐allyl‐3‐oxazolin‐5‐one derivatives. Extension to a triple domino process, in which the above process is combined with in situ azlactone formation starting from amino acids renders this process even more attractive. Subsequent thermolysis of the
-
Iron-Catalyzed Indolizine Synthesis from Pyridines, Diazo Compounds, and Alkynes作者:Tim Douglas、Anca Pordea、James DowdenDOI:10.1021/acs.orglett.7b03252日期:2017.12.1The iron(III)-catalyzed synthesis of indolizines from commercially available alkyne, pyridine, and diazo precursors is reported. This mild, expedient method is tolerant of various solvents and proceeds with as little as 0.25 mol % [Fe(TPP)Cl]. Significantly, this multicomponent reaction is compatible with electrophilic alkynes; control experiments demonstrate the importance of the catalyst in promoting
-
Acetylenic esters. Part III. Reactions of thiocarbonyl compounds with methyl propiolate, methyl methylpropiolate, and methyl phenylpropiolate作者:G. Dallas、J. W. Lown、J. C. N. MaDOI:10.1039/j39680002510日期:——Non-terminal acetylenic esters react with many thiocarbonyl compounds to form heterocycles or 1 : 1 addition products. Propiolic esters in most cases form isomeric mixtures of dimethyl 3,3′-thiodiacrylates (I). The stereochemical course of the thiolic nucleophilic additions may be controlled for preparative purposes by changes in solvent polarity. All three stereoisomers of (I) have been prepared.
表征谱图
-
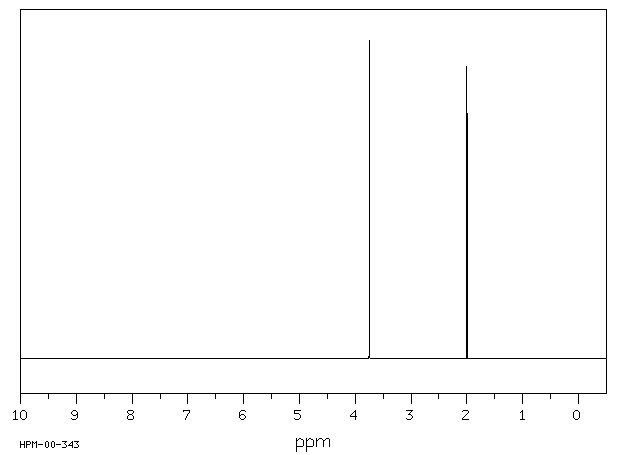
氢谱1HNMR
-
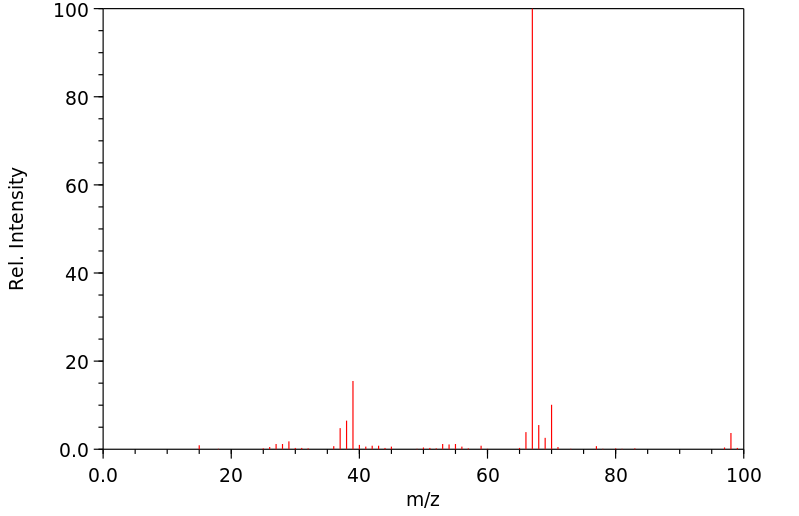
质谱MS
-
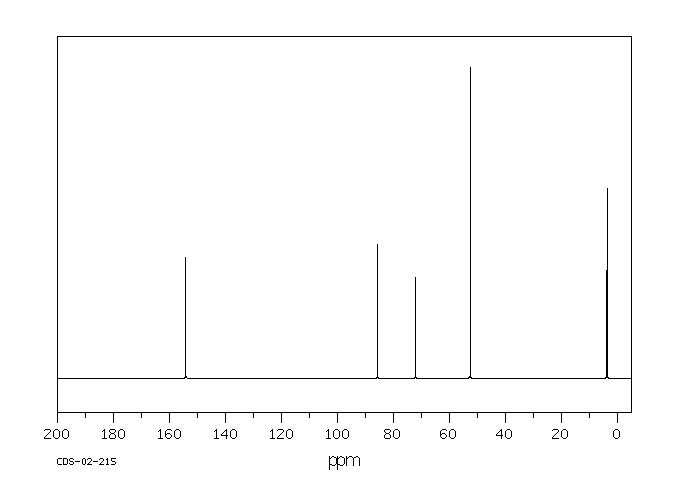
碳谱13CNMR
-
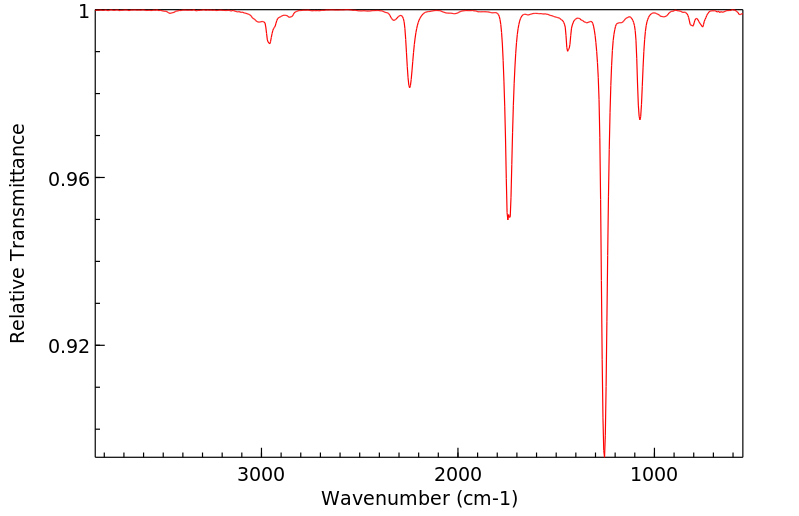
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(±)17,18-二HETE
(±)-辛酰肉碱氯化物
(Z)-5-辛烯甲酯
(Z)-4-辛烯酸
(R)-甲羟戊酸锂盐
(R)-普鲁前列素,游离酸
(R,R)-半乳糖苷
(E)-4-庚烯酸
(E)-4-壬烯酸
(E)-4-十一烯酸
(9Z,12E)-十八烷二烯酸甲酯
(6E)-8-甲基--6-壬烯酸甲基酯-d3
(3R,6S)-rel-8-[2-(3-呋喃基)-1,3-二氧戊环-2-基]-3-羟基-2,6-二甲基-4-辛酮
龙胆二糖
黑曲霉二糖
黄质霉素
麦芽酮糖一水合物
麦芽糖醇
麦芽糖酸
麦芽糖基蔗糖
麦芽糖一水合物
麦芽糖
鳄梨油酸乙酯
鲸蜡醇蓖麻油酸酯
鲸蜡醇油酸酯
鲸蜡硬脂醇硬脂酸酯
鲸蜡烯酸脂
鲸蜡基花生醇
鲫鱼酸
鲁比前列素
鲁比前列素
高级烷基C16-18-醇
高甲羟戊酸
高效氯氰菊酯
高-gamma-亚油酸
马来酸烯丙酯
马来酸氢异丙酯
马来酸氢异丁酯
马来酸氢丙酯
马来酸氢1-[2-(2-羟基乙氧基)乙基]酯
马来酸单乙酯
马来酸单丁酯
马来酸二辛酯
马来酸二癸酯
马来酸二甲酯
马来酸二烯丙酯
马来酸二正丙酯
马来酸二戊基酯
马来酸二异壬酯
马来酸二异丙酯