2-乙基己酸正丁酯 | 68443-63-0
中文名称
2-乙基己酸正丁酯
中文别名
丁基-2-乙基己酸酯;2-乙基己酸丁酯
英文名称
butyl 2-ethylhexanoate
英文别名
——
CAS
68443-63-0
化学式
C12H24O2
mdl
——
分子量
200.321
InChiKey
USPADFUBVAGYOJ-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
沸点:230 °C
-
密度:0.86
-
LogP:4.705 (est)
-
保留指数:1281
-
稳定性/保质期:
远离氧化物、阳光、热源、明火、高温、火花以及静电电荷和其他点火源。
计算性质
-
辛醇/水分配系数(LogP):4.2
-
重原子数:14
-
可旋转键数:9
-
环数:0.0
-
sp3杂化的碳原子比例:0.92
-
拓扑面积:26.3
-
氢给体数:0
-
氢受体数:2
安全信息
-
海关编码:2915900090
SDS
Butyl 2-Ethylhexanoate
SAFETY DATA SHEET
Section 1. IDENTIFICATION
Product name: Butyl 2-Ethylhexanoate
Section 2. HAZARDS IDENTIFICATION
GHS classification
PHYSICAL HAZARDS Not classified
Not classified
HEALTH HAZARDS
ENVIRONMENTAL HAZARDS Not classified
GHS label elements, including precautionary statements
Pictograms or hazard symbols None
No signal word
Signal word
Hazard statements None
None
Precautionary statements:
Section 3. COMPOSITION/INFORMATION ON INGREDIENTS
Substance/mixture: Substance
Components: Butyl 2-Ethylhexanoate
Percent: >98.0%(GC)
CAS Number: 68443-63-0
Synonyms: 2-Ethylhexanoic Acid Butyl Ester
Chemical Formula: C12H24O2
Section 4. FIRST AID MEASURES
Inhalation: Remove victim to fresh air and keep at rest in a position comfortable for breathing.
Get medical advice/attention if you feel unwell.
Skin contact: Remove/Take off immediately all contaminated clothing. Rinse skin with
water/shower. If skin irritation or rash occurs: Get medical advice/attention.
Eye contact: Rinse cautiously with water for several minutes. Remove contact lenses, if present
and easy to do. Continue rinsing. If eye irritation persists: Get medical
advice/attention.
Ingestion: Get medical advice/attention if you feel unwell. Rinse mouth.
Protection of first-aiders: A rescuer should wear personal protective equipment, such as rubber gloves and air-
tight goggles.
Section 5. FIRE-FIGHTING MEASURES
Suitable extinguishing Dry chemical, foam, carbon dioxide.
media:
Unsuitable extinguishing Water (It may scatter and spread fire.)
media:
Butyl 2-Ethylhexanoate
Section 5. FIRE-FIGHTING MEASURES
Precautions for firefighters: Fire-extinguishing work is done from the windward and the suitable fire-extinguishing
method according to the surrounding situation is used. Uninvolved persons should
evacuate to a safe place. In case of fire in the surroundings: Remove movable
containers if safe to do so.
Special protective When extinguishing fire, be sure to wear personal protective equipment.
equipment for firefighters:
Section 6. ACCIDENTAL RELEASE MEASURES
Use personal protective equipment. Keep people away from and upwind of spill/leak.
Personal precautions,
protective equipment and Ensure adequate ventilation. Entry to non-involved personnel should be controlled
emergency procedures: around the leakage area by roping off, etc.
Environmental precautions: Prevent product from entering drains.
Methods and materials for Absorb spilled material in a suitable absorbent (e.g. rag, dry sand, earth, saw-dust).
containment and cleaning In case of large amount of spillage, contain a spill by bunding. Adhered or collected
up: material should be promptly disposed of, in accordance with appropriate laws and
regulations.
Section 7. HANDLING AND STORAGE
Precautions for safe handling
Technical measures: Handling is performed in a well ventilated place. Wear suitable protective equipment.
Prevent generation of vapour or mist. Wash hands and face thoroughly after
handling.
Use a ventilation, local exhaust if vapour or aerosol will be generated.
Advice on safe handling: Avoid contact with skin, eyes and clothing.
Conditions for safe storage, including any
incompatibilities
Storage conditions: Keep container tightly closed. Store in a cool and dark place.
Store away from incompatible materials such as oxidizing agents.
Packaging material: Comply with laws.
Section 8. EXPOSURE CONTROLS / PERSONAL PROTECTION
Install a closed system or local exhaust as possible so that workers should not be
Engineering controls:
exposed directly. Also install safety shower and eye bath.
Personal protective equipment
Respiratory protection: Vapor respirator. Follow local and national regulations.
Hand protection: Protective gloves.
Eye protection: Safety glasses. A face-shield, if the situation requires.
Skin and body protection: Protective clothing. Protective boots, if the situation requires.
Section 9. PHYSICAL AND CHEMICAL PROPERTIES
Physical state (20°C): Liquid
Form: Clear
Colorless - Almost colorless
Colour:
Odour: No data available
pH: No data available
Melting point/freezing point:No data available
230°C
Boiling point/range:
Flash point: No data available
Flammability or explosive
limits:
No data available
Lower:
Upper: No data available
0.86
Relative density:
Solubility(ies):
No data available
[Water]
[Other solvents] No data available
Butyl 2-Ethylhexanoate
Section 10. STABILITY AND REACTIVITY
Chemical stability: Stable under proper conditions.
Possibility of hazardous No special reactivity has been reported.
reactions:
Incompatible materials: Oxidizing agents
Hazardous decomposition Carbon monoxide, Carbon dioxide
products:
Section 11. TOXICOLOGICAL INFORMATION
Acute Toxicity: No data available
Skin corrosion/irritation: No data available
Serious eye No data available
damage/irritation:
Germ cell mutagenicity: No data available
Carcinogenicity:
IARC = No data available
No data available
NTP =
Reproductive toxicity: No data available
Section 12. ECOLOGICAL INFORMATION
Ecotoxicity:
Fish: No data available
Crustacea: No data available
Algae: No data available
Persistence / degradability: No data available
Bioaccumulative No data available
potential(BCF):
Mobility in soil
No data available
Log Pow:
Soil adsorption (Koc): No data available
Henry's Law No data available
constant(PaM3/mol):
Section 13. DISPOSAL CONSIDERATIONS
Recycle to process, if possible. Consult your local regional authorities. You may be able to burn in a chemical
incinerator equipped with an afterburner and scrubber system. Observe all federal, state and local regulations when
disposing of the substance.
Section 14. TRANSPORT INFORMATION
Does not correspond to the classification standard of the United Nations
Hazards Class:
UN-No: Not listed
Section 15. REGULATORY INFORMATION
Safe management ordinance of dangerous chemical product (State Council announces on January 26, 2002
and revised on February 16,2011): Safe use and production, the storage of a dangerous chemical, transport,
loading and unloading were prescribed.
Butyl 2-Ethylhexanoate
SECTION 16 - ADDITIONAL INFORMATION
N/A
SAFETY DATA SHEET
Section 1. IDENTIFICATION
Product name: Butyl 2-Ethylhexanoate
Section 2. HAZARDS IDENTIFICATION
GHS classification
PHYSICAL HAZARDS Not classified
Not classified
HEALTH HAZARDS
ENVIRONMENTAL HAZARDS Not classified
GHS label elements, including precautionary statements
Pictograms or hazard symbols None
No signal word
Signal word
Hazard statements None
None
Precautionary statements:
Section 3. COMPOSITION/INFORMATION ON INGREDIENTS
Substance/mixture: Substance
Components: Butyl 2-Ethylhexanoate
Percent: >98.0%(GC)
CAS Number: 68443-63-0
Synonyms: 2-Ethylhexanoic Acid Butyl Ester
Chemical Formula: C12H24O2
Section 4. FIRST AID MEASURES
Inhalation: Remove victim to fresh air and keep at rest in a position comfortable for breathing.
Get medical advice/attention if you feel unwell.
Skin contact: Remove/Take off immediately all contaminated clothing. Rinse skin with
water/shower. If skin irritation or rash occurs: Get medical advice/attention.
Eye contact: Rinse cautiously with water for several minutes. Remove contact lenses, if present
and easy to do. Continue rinsing. If eye irritation persists: Get medical
advice/attention.
Ingestion: Get medical advice/attention if you feel unwell. Rinse mouth.
Protection of first-aiders: A rescuer should wear personal protective equipment, such as rubber gloves and air-
tight goggles.
Section 5. FIRE-FIGHTING MEASURES
Suitable extinguishing Dry chemical, foam, carbon dioxide.
media:
Unsuitable extinguishing Water (It may scatter and spread fire.)
media:
Butyl 2-Ethylhexanoate
Section 5. FIRE-FIGHTING MEASURES
Precautions for firefighters: Fire-extinguishing work is done from the windward and the suitable fire-extinguishing
method according to the surrounding situation is used. Uninvolved persons should
evacuate to a safe place. In case of fire in the surroundings: Remove movable
containers if safe to do so.
Special protective When extinguishing fire, be sure to wear personal protective equipment.
equipment for firefighters:
Section 6. ACCIDENTAL RELEASE MEASURES
Use personal protective equipment. Keep people away from and upwind of spill/leak.
Personal precautions,
protective equipment and Ensure adequate ventilation. Entry to non-involved personnel should be controlled
emergency procedures: around the leakage area by roping off, etc.
Environmental precautions: Prevent product from entering drains.
Methods and materials for Absorb spilled material in a suitable absorbent (e.g. rag, dry sand, earth, saw-dust).
containment and cleaning In case of large amount of spillage, contain a spill by bunding. Adhered or collected
up: material should be promptly disposed of, in accordance with appropriate laws and
regulations.
Section 7. HANDLING AND STORAGE
Precautions for safe handling
Technical measures: Handling is performed in a well ventilated place. Wear suitable protective equipment.
Prevent generation of vapour or mist. Wash hands and face thoroughly after
handling.
Use a ventilation, local exhaust if vapour or aerosol will be generated.
Advice on safe handling: Avoid contact with skin, eyes and clothing.
Conditions for safe storage, including any
incompatibilities
Storage conditions: Keep container tightly closed. Store in a cool and dark place.
Store away from incompatible materials such as oxidizing agents.
Packaging material: Comply with laws.
Section 8. EXPOSURE CONTROLS / PERSONAL PROTECTION
Install a closed system or local exhaust as possible so that workers should not be
Engineering controls:
exposed directly. Also install safety shower and eye bath.
Personal protective equipment
Respiratory protection: Vapor respirator. Follow local and national regulations.
Hand protection: Protective gloves.
Eye protection: Safety glasses. A face-shield, if the situation requires.
Skin and body protection: Protective clothing. Protective boots, if the situation requires.
Section 9. PHYSICAL AND CHEMICAL PROPERTIES
Physical state (20°C): Liquid
Form: Clear
Colorless - Almost colorless
Colour:
Odour: No data available
pH: No data available
Melting point/freezing point:No data available
230°C
Boiling point/range:
Flash point: No data available
Flammability or explosive
limits:
No data available
Lower:
Upper: No data available
0.86
Relative density:
Solubility(ies):
No data available
[Water]
[Other solvents] No data available
Butyl 2-Ethylhexanoate
Section 10. STABILITY AND REACTIVITY
Chemical stability: Stable under proper conditions.
Possibility of hazardous No special reactivity has been reported.
reactions:
Incompatible materials: Oxidizing agents
Hazardous decomposition Carbon monoxide, Carbon dioxide
products:
Section 11. TOXICOLOGICAL INFORMATION
Acute Toxicity: No data available
Skin corrosion/irritation: No data available
Serious eye No data available
damage/irritation:
Germ cell mutagenicity: No data available
Carcinogenicity:
IARC = No data available
No data available
NTP =
Reproductive toxicity: No data available
Section 12. ECOLOGICAL INFORMATION
Ecotoxicity:
Fish: No data available
Crustacea: No data available
Algae: No data available
Persistence / degradability: No data available
Bioaccumulative No data available
potential(BCF):
Mobility in soil
No data available
Log Pow:
Soil adsorption (Koc): No data available
Henry's Law No data available
constant(PaM3/mol):
Section 13. DISPOSAL CONSIDERATIONS
Recycle to process, if possible. Consult your local regional authorities. You may be able to burn in a chemical
incinerator equipped with an afterburner and scrubber system. Observe all federal, state and local regulations when
disposing of the substance.
Section 14. TRANSPORT INFORMATION
Does not correspond to the classification standard of the United Nations
Hazards Class:
UN-No: Not listed
Section 15. REGULATORY INFORMATION
Safe management ordinance of dangerous chemical product (State Council announces on January 26, 2002
and revised on February 16,2011): Safe use and production, the storage of a dangerous chemical, transport,
loading and unloading were prescribed.
Butyl 2-Ethylhexanoate
SECTION 16 - ADDITIONAL INFORMATION
N/A
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 2-乙基-己酸稀土盐 2-Ethylhexanoic acid 149-57-5 C8H16O2 144.214
反应信息
-
作为反应物:描述:2-乙基己酸正丁酯 在 sodium hypochlorite 、 sodium chlorite 、 9-borabicyclo[3.3.1]nonane dimer 、 正丁基锂 、 2,2,6,6-四甲基哌啶氧化物 、 dichloro(norbornadiene)palladium(II) 、 苯甲酸酐 、 二异丙胺 、 4,5-双二苯基膦-9,9-二甲基氧杂蒽 作用下, 以 四氢呋喃 、 N-甲基吡咯烷酮 、 aq. phosphate buffer 、 正己烷 、 乙腈 为溶剂, 反应 23.34h, 生成 butyl 2-ethyl-2-vinylhexanoate参考文献:名称:钯催化的脱羰脱水用于合成α-乙烯基羰基化合物和(-)-Aspewentins A,B和C的全合成摘要:羰基化合物的直接α-乙烯基化以形成季立体中心是一项具有挑战性的转变。发现δ-氧代羧酸可作为掩蔽的乙烯基化合物,并通过钯催化的脱羰基脱水作用而公开。通过对映选择性丙烯酸酯加成或不对称烯丙基烷基化可容易地获得羧酸。以高收率获得了多种α-乙烯基季羰基化合物,并证明了其在(-)-aspewentins A,B和C的首次对映选择性全合成中的应用。DOI:10.1002/anie.201505161
-
作为产物:描述:参考文献:名称:Kenyon; Platt, Journal of the Chemical Society, 1939, p. 637摘要:DOI:
文献信息
-
A general, high yield synthesis of α-oxoketene dithioacetals and O-(1-alkoxy-2,2-dialkyl)vinyl S-alkyl dithiocarbonates from carboxylic esters作者:Syed Mashhood Ali、Shigeo TanimotoDOI:10.1039/c39890000684日期:——α-Oxoketene dithioacetals (5) and O-(1-alkoxy-2,2-dialkyl)vinyl S-alkyl dithiocarbonates (4) were prepared in high yields from carboxylic esters by the reaction of their lithium enolates (2) with carbon disulphide at –78 °C followed by alkylation.
-
Cross-Dehydrogenating Coupling of Aldehydes with Amines/R-OTBS Ethers by Visible-Light Photoredox Catalysis: Synthesis of Amides, Esters, and Ureas作者:Ganesh Pandey、Suvajit Koley、Ranadeep Talukdar、Pramod Kumar SahaniDOI:10.1021/acs.orglett.8b02537日期:2018.9.21A straightforward synthesis of amides, ureas, and esters is reported by visible-light cross-dehydrogenating coupling (CDC) of aldehydes (or amine carbaldehydes) and amines/R-OTBS ethers by photoredox catalysis. The reaction is found to be general and high yielding. A plausible mechanistic pathway has been proposed for these transformations and is supported by appropriate controlled experiments.
-
[EN] PROCESSES FOR PRODUCING CARBOXYLIC ACIDS<br/>[FR] PROCÉDÉS DE PRODUCTION D'ACIDES CARBOXYLIQUES申请人:EASTMAN CHEM CO公开号:WO2020205348A1公开(公告)日:2020-10-08Processes are disclosed for preparing carboxylic acids from organic esters, the processes comprising contacting an ester with water in the presence of an acid catalyst and a homogenizing solvent at conditions effective to form a carboxylic acid. The homogenizing solvent is present in an amount sufficient to form a single-phase reaction mixture comprising the ester, water, and homogenizing solvent. The homogenizing solvent may be selected from acetonitrile, dimethyl sulfoxide, and 1,4-dioxane.
-
METHOD FOR PRODUCING a-SUBSTITUTED CYSTEINE OR SALT THEREOF OR SYNTHETIC INTERMEDIATE OF a-SUBSTITUTED CYSTEINE申请人:API Corporation公开号:US20160083341A1公开(公告)日:2016-03-24According to the present invention, it becomes possible to perform a process for converting into an α-substituted cysteine represented by general formula (1) or a salt thereof at low cost and on an industrial scale by employing a process that is routed through a compound represented by general formula (3) to a compound represented by general formula (6). Particularly, by employing a process that is routed through a compound represented by general formula (7-2), it becomes possible to detach a tert-butyl protection group in a simple manner and to produce the compound represented by general formula (1) with high purity. Furthermore, by employing a process that is routed through tert-butylthiomethanol or a process that is routed through a compound represented by general formula (9), it becomes possible to produce a compound represented by general formula (2) without generating bischloromethylether that is an oncogenic substance. In the production of an α-substituted-D-cysteine or a salt thereof, it becomes possible to perform a process for converting the compound represented by general formula (2) into a compound represented by general formula (3S) in one step by allowing an enzyme or the like to act on the compound represented by general formula (2).
-
Highly isoselective catalyst for alkene hydroformylation申请人:Eastman Chemical Company公开号:US10144751B1公开(公告)日:2018-12-04Ligands for use with catalyst compositions used in hydroformylation reactions are described herein. The ligands are used with various octofluorotoluene or hydrocarbon solvents and achieve an increase in isoselectivity with an increase in temperature, an increase in TON with an increase in temperature, and/or will show isoselectivity that is surprisingly high in comparison to the hydroformylation reactions using common solvents.
表征谱图
-
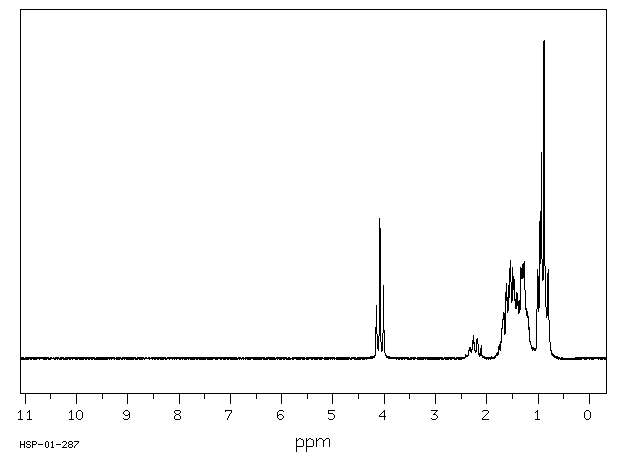
氢谱1HNMR
-
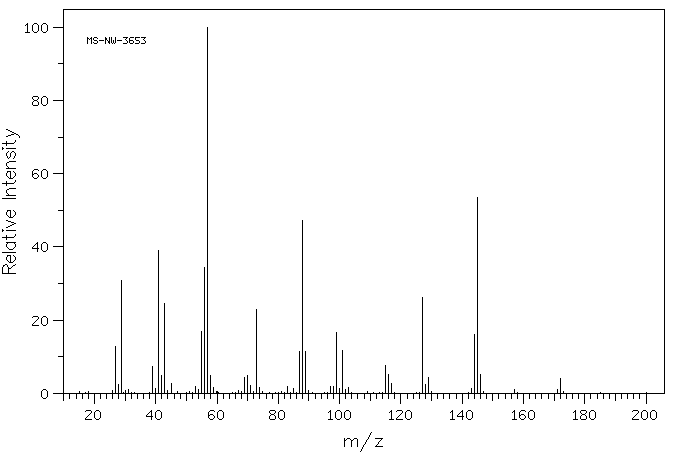
质谱MS
-
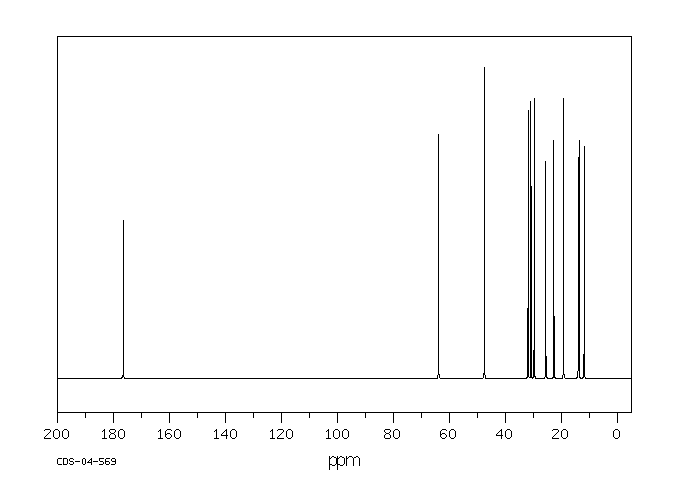
碳谱13CNMR
-
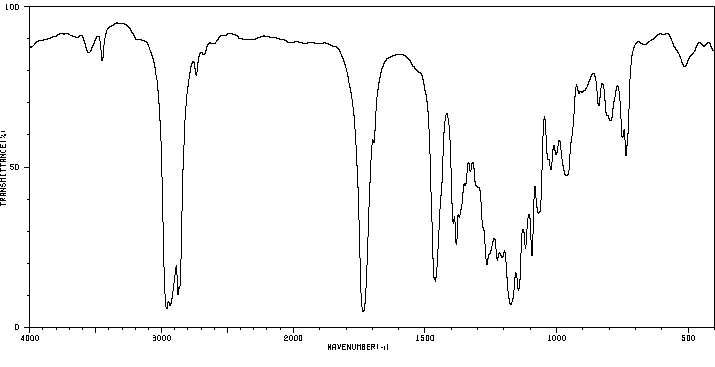
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(±)17,18-二HETE
(±)-辛酰肉碱氯化物
(Z)-5-辛烯甲酯
(Z)-4-辛烯酸
(R)-甲羟戊酸锂盐
(R)-普鲁前列素,游离酸
(R,R)-半乳糖苷
(E)-4-庚烯酸
(E)-4-壬烯酸
(E)-4-十一烯酸
(9Z,12E)-十八烷二烯酸甲酯
(6E)-8-甲基--6-壬烯酸甲基酯-d3
(3R,6S)-rel-8-[2-(3-呋喃基)-1,3-二氧戊环-2-基]-3-羟基-2,6-二甲基-4-辛酮
龙胆二糖
黑曲霉二糖
黄质霉素
麦芽酮糖一水合物
麦芽糖醇
麦芽糖酸
麦芽糖基蔗糖
麦芽糖一水合物
麦芽糖
鳄梨油酸乙酯
鲸蜡醇蓖麻油酸酯
鲸蜡醇油酸酯
鲸蜡硬脂醇硬脂酸酯
鲸蜡烯酸脂
鲸蜡基花生醇
鲫鱼酸
鲁比前列素
鲁比前列素
高级烷基C16-18-醇
高甲羟戊酸
高效氯氰菊酯
高-gamma-亚油酸
马来酸烯丙酯
马来酸氢异丙酯
马来酸氢异丁酯
马来酸氢丙酯
马来酸氢1-[2-(2-羟基乙氧基)乙基]酯
马来酸单乙酯
马来酸单丁酯
马来酸二辛酯
马来酸二癸酯
马来酸二甲酯
马来酸二烯丙酯
马来酸二正丙酯
马来酸二戊基酯
马来酸二异壬酯
马来酸二异丙酯