4-庚基苯酚 | 1987-50-4
中文名称
4-庚基苯酚
中文别名
4-正庚基苯酚;4-N-庚基苯酚;对庚基苯酚
英文名称
p-heptylphenol
英文别名
4-heptylphenol;4-n-Heptylphenol;para-n-heptylphenol
CAS
1987-50-4
化学式
C13H20O
mdl
MFCD00041751
分子量
192.301
InChiKey
KNDDEFBFJLKPFE-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:26-28 °C
-
沸点:156 °C (9 mmHg)
-
密度:0.9402 (estimate)
-
闪点:156°C/9mm
-
溶解度:乙腈(微溶)、氯仿(微溶)、甲醇(微溶)
-
LogP:5.130 (est)
-
物理描述:Liquid
-
保留指数:1665.5;1669;1673.8;1678
-
稳定性/保质期:
如果按照规定使用和储存,则不会发生分解,目前没有已知的危险反应,应避免接触氧化剂。
计算性质
-
辛醇/水分配系数(LogP):4.8
-
重原子数:14
-
可旋转键数:6
-
环数:1.0
-
sp3杂化的碳原子比例:0.538
-
拓扑面积:20.2
-
氢给体数:1
-
氢受体数:1
安全信息
-
TSCA:Yes
-
危险等级:8
-
危险品标志:C
-
安全说明:S26,S36/37/39,S45
-
危险类别码:R34
-
海关编码:29071990
-
危险品运输编号:2430
-
包装等级:III
-
危险类别:8
-
储存条件:请将贮藏器密封保存,并将其存放在阴凉、干燥处。同时,确保工作环境具有良好的通风或排气设施。
SDS
| Name: | 4-Heptylphenol 98% (Titr.) Material Safety Data Sheet |
| Synonym: | None known |
| CAS: | 1987-50-4 |
Synonym:None known
Section 2 - COMPOSITION, INFORMATION ON INGREDIENTS
| CAS# | Chemical Name | content | EINECS# |
| 1987-50-4 | Phenol, 4-Heptyl- | 98 | 217-862-0 |
Risk Phrases: 36/37/38
Section 3 - HAZARDS IDENTIFICATION
EMERGENCY OVERVIEW
Irritating to eyes, respiratory system and skin.The toxicological properties of this material have not been fully investigated.
Potential Health Effects
Eye:
Causes eye irritation. May cause chemical conjunctivitis.
Skin:
Causes skin irritation.
Ingestion:
May cause gastrointestinal irritation with nausea, vomiting and diarrhea. The toxicological properties of this substance have not been fully investigated.
Inhalation:
Causes respiratory tract irritation. The toxicological properties of this substance have not been fully investigated. Can produce delayed pulmonary edema.
Chronic:
Effects may be delayed.
Section 4 - FIRST AID MEASURES
Eyes: Immediately flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid.
Skin:
Get medical aid. Flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes. Wash clothing before reuse.
Ingestion:
Get medical aid. Do NOT induce vomiting. If conscious and alert, rinse mouth and drink 2-4 cupfuls of milk or water.
Inhalation:
Remove from exposure and move to fresh air immediately. If not breathing, give artificial respiration. If breathing is difficult, give oxygen. Get medical aid.
Notes to Physician:
Treat symptomatically and supportively.
Section 5 - FIRE FIGHTING MEASURES
General Information:
As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear. During a fire, irritating and highly toxic gases may be generated by thermal decomposition or combustion. Use water spray to keep fire-exposed containers cool. Vapors may be heavier than air. They can spread along the ground and collect in low or confined areas. Containers may explode when heated.
Extinguishing Media:
Use agent most appropriate to extinguish fire. Cool containers with flooding quantities of water until well after fire is out. Use water spray, dry chemical, carbon dioxide, or appropriate foam.
Section 6 - ACCIDENTAL RELEASE MEASURES
General Information: Use proper personal protective equipment as indicated in Section 8.
Spills/Leaks:
Absorb spill with inert material (e.g. vermiculite, sand or earth), then place in suitable container. Avoid runoff into storm sewers and ditches which lead to waterways. Clean up spills immediately, observing precautions in the Protective Equipment section. Remove all sources of ignition. Provide ventilation.
Section 7 - HANDLING and STORAGE
Handling:
Wash thoroughly after handling. Remove contaminated clothing and wash before reuse. Use with adequate ventilation. Avoid contact with eyes, skin, and clothing. Keep container tightly closed. Avoid ingestion and inhalation.
Storage:
Store in a tightly closed container. Store in a cool, dry, well-ventilated area away from incompatible substances.
Section 8 - EXPOSURE CONTROLS, PERSONAL PROTECTION
Engineering Controls:
Facilities storing or utilizing this material should be equipped with an eyewash facility and a safety shower. Use adequate ventilation to keep airborne concentrations low.
Exposure Limits CAS# 1987-50-4: Personal Protective Equipment Eyes: Wear appropriate protective eyeglasses or chemical safety goggles as described by OSHA's eye and face protection regulations in 29 CFR 1910.133 or European Standard EN166.
Skin:
Wear appropriate protective gloves to prevent skin exposure.
Clothing:
Wear appropriate protective clothing to prevent skin exposure.
Respirators:
A respiratory protection program that meets OSHA's 29 CFR 1910.134 and ANSI Z88.2 requirements or European Standard EN 149 must be followed whenever workplace conditions warrant respirator use.
Section 9 - PHYSICAL AND CHEMICAL PROPERTIES
Physical State: Liquid
Color: colorless
Odor: phenol-like
pH: Not available.
Vapor Pressure: Not available.
Viscosity: Not available.
Boiling Point: 156 deg C @ 9.00mm Hg
Freezing/Melting Point: Not available.
Autoignition Temperature: Not available.
Flash Point: > 110 deg C (> 230.00 deg F)
Explosion Limits, lower: Not available.
Explosion Limits, upper: Not available.
Decomposition Temperature:
Solubility in water:
Specific Gravity/Density:
Molecular Formula: C13H20O
Molecular Weight: 192.30
Section 10 - STABILITY AND REACTIVITY
Chemical Stability:
Stable at room temperature in closed containers under normal storage and handling conditions.
Conditions to Avoid:
Incompatible materials, excess heat, strong oxidants.
Incompatibilities with Other Materials:
Oxidizing agents.
Hazardous Decomposition Products:
Carbon monoxide, irritating and toxic fumes and gases, carbon dioxide.
Hazardous Polymerization: Has not been reported
Section 11 - TOXICOLOGICAL INFORMATION
RTECS#:
CAS# 1987-50-4 unlisted.
LD50/LC50:
Not available.
Carcinogenicity:
Phenol, 4-Heptyl- - Not listed by ACGIH, IARC, or NTP.
Section 12 - ECOLOGICAL INFORMATION
Section 13 - DISPOSAL CONSIDERATIONS
Dispose of in a manner consistent with federal, state, and local regulations.
Section 14 - TRANSPORT INFORMATION
IATA
Not regulated as a hazardous material.
IMO
Not regulated as a hazardous material.
RID/ADR
Not regulated as a hazardous material.
Section 15 - REGULATORY INFORMATION
European/International Regulations
European Labeling in Accordance with EC Directives
Hazard Symbols: XI
Risk Phrases:
R 36/37/38 Irritating to eyes, respiratory system
and skin.
Safety Phrases:
S 26 In case of contact with eyes, rinse immediately
with plenty of water and seek medical advice.
S 28A After contact with skin, wash immediately with
plenty of water.
S 37 Wear suitable gloves.
S 37/39 Wear suitable gloves and eye/face
protection.
S 45 In case of accident or if you feel unwell, seek
medical advice immediately (show the label where
possible).
WGK (Water Danger/Protection)
CAS# 1987-50-4: No information available.
Canada
CAS# 1987-50-4 is listed on Canada's DSL List.
CAS# 1987-50-4 is not listed on Canada's Ingredient Disclosure List.
US FEDERAL
TSCA
CAS# 1987-50-4 is listed on the TSCA inventory.
SECTION 16 - ADDITIONAL INFORMATION
N/A
制备方法与用途
用途:用于制作液晶原料及中间体。
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 对壬基酚 4-Nonylphenol 104-40-5 C15H24O 220.355 —— 1-heptyl-4-methoxybenzene 92368-72-4 C14H22O 206.328 —— 1-(4-hydroxy-phenyl)-heptan-1-ol 106379-86-6 C13H20O2 208.301 4'-羟基苯庚酮 4-heptanoylphenol 14392-72-4 C13H18O2 206.285 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 庚基苯 heptylbenzene 1078-71-3 C13H20 176.302 —— 1-(allyloxy)-4-n-heptylbenzene 1448537-47-0 C16H24O 232.366 —— 2-chloro-4-heptylphenol 18980-06-8 C13H19ClO 226.746 —— 2-bromo 4-n-heptylphenol 88134-01-4 C13H19BrO 271.197 —— (S)-3-(4-n-heptylphenoxy)-propane-1,2-diol 1448537-54-9 C16H26O3 266.381 —— p-heptylbenzenethiol 4527-47-3 C13H20S 208.368 5-庚基-2-羟基苯甲腈 2-cyano 4-n-heptylphenol 52899-66-8 C14H19NO 217.311 —— 4-Heptyl-2,6-diiodophenol 113372-82-0 C13H18I2O 444.09
反应信息
-
作为反应物:参考文献:名称:使用离子液体在温和条件下将苯酚选择性加氢脱氧成芳烃的负载型Pt催化剂的表面改性。摘要:利用木质素作为可再生芳香族化学品的来源,从木质素衍生的酚中选择性和有效地去除含氧基团是一项严峻的挑战。该报告描述了使用离子液体(ILs)对沸石负载的Pt催化剂进行的表面改性如何在使用大气压H2的温和反应条件下显着提高苯酚加氢脱氧(HDO)生成芳烃的选择性。未经修饰的Pt / H-ZSM-5将苯酚与少量的芳烃(选择性为10%)一起转化为主要产物的脂肪族物质。相反,用IL 1-氟-3-甲基咪唑鎓三氟甲磺酸酯改性的催化剂即使在苯酚几乎完全转化的情况下,仍能保持高达76%的芳烃选择性。DOI:10.1002/chem.201902668
-
作为产物:描述:参考文献:名称:锂-乙二胺-THF-介导的芳烃α-位裂解的范围和局限性:在温和条件下芳基甲基醚和苄基醚的脱保护摘要:考察了锂-乙二胺-THF介导的芳烃α位还原键断裂的范围和局限性。即使在低至 -10 °C 的条件下,无氧 THF 中的锂金属(5 equiv)和乙二胺(7 equiv)等非常温和的条件对芳族醚的去甲基化也非常有效。烯丙基苄基醚也在这些条件下脱保护,烯丙醇部分几乎没有变化。通过这项研究,开发了 2,6-二甲基苄基 (m-xylylmethyl, MXM) 基团作为苄基的替代物,在上述还原条件下易于裂解。DOI:10.1055/s-2004-815977
-
作为试剂:描述:参考文献:名称:PROCESS FOR THE PREPARATION OF N-SUBSTITUTED CARBAMIC ACID ESTER AND PROCESS FOR THE PREPARATION OF ISOCYANATE USING THE N-SUBSTITUTED CARBAMIC ACID ESTER摘要:公开号:EP2322504B9
文献信息
-
Certain benzopyran and benzothiopyran derivatives申请人:Ciba-Geigy Corporation公开号:US05155130A1公开(公告)日:1992-10-13The invention relates to the compounds of the formula ##STR1## wherein each R independently represents hydrogen, lower alkyl, halogen, trifluoromethyl, lower alkoxy, carbocyclic or heterocyclic aryl, carbocyclic or heterocyclic aryloxy, carbocyclic or heterocyclic aryl-lower alkyloxy, carbocyclic or heterocyclic aryl-lower alkyl, C.sub.3 -C.sub.7 -cycloalkyl-lower alkyloxy, or C.sub.3 -C.sub.7 -cycloalkyloxy; R.sub.1 represents hydrogen, lower alkyl or carbocyclic aryl; R.sub.2 represents hydrogen or lower alkyl; Y represents oxygen (O), sulfur (S), sulfinyl or sulfonyl; n represents 1, 2 or 3; A represents a direct bond or lower alkylene; Z represents ##STR2## wherein R.sub.3 represents hydrogen or acyl; R.sub.4 represents lower alkyl, C.sub.3 -C.sub.7 -cycloalkyl, carbocyclic or heterocyclic aryl, carbocyclic or heterocyclic aryl-lower alkyl, or C.sub.3 -C.sub.7 -cycloalkyl-lower alkyl; or Z represents ##STR3## wherein X represents oxygen or sulfur, R.sub.3 represents hydrogen, acyl, lower alkoxycarbonyl, aminocarbonyl, mono- or di-lower alkylaminocarbonyl, lower alkenylaminocarbonyl, lower alkynylaminocarbonyl, carbocyclic or heterocyclic aryl-lower alkylaminocarbonyl, carbocyclic or heterocyclic arylamino-carbonyl; C.sub.3 -C.sub.7 -cycloalkylaminocarbonyl, or C.sub.3 -C.sub.7 -cycloalkyl-lower alkylaminocarbonyl; R.sub.5 represents lower alkyl, lower alkoxycarbonyl-lower alkyl, C.sub.3 -C.sub.7 -cycloalkyl, carbocyclic or heterocyclic aryl, carbocyclic or heterocyclic aryl-lower alkyl, C.sub.3 -C.sub.7 -cycloalkyl-lower alkyl, amino, mono- or di-lower alkylamino, lower alkenylamino, lower alkynylamino, carbocyclic or heterocyclic aryl-lower alkylamino, carbocyclic or heterocyclic arylamino, C.sub.3 -C.sub.7 -cycloalkylamino, C.sub.3 -C.sub.7 -cycloalkyl-lower alkylamino, lower alkoxycarbonyl-lower alkylamino, or lower alkoxy; R.sub.6 and R.sub.7 independently represent hydrogen or lower alkyl; and pharmaceutically acceptable salts thereof; which are useful as 5-lipoxygenase inhibitors.该发明涉及以下式的化合物##STR1##其中每个R独立地代表氢、较低的烷基、卤素、三氟甲基、较低的烷氧基、碳环或杂环芳基、碳环或杂环芳氧基、碳环或杂环芳基-较低烷氧基、碳环或杂环芳基-较低烷基、C.sub.3-C.sub.7-环烷基-较低烷氧基、或C.sub.3-C.sub.7-环烷氧基;R.sub.1代表氢、较低烷基或碳环芳基;R.sub.2代表氢或较低烷基;Y代表氧(O)、硫(S)、亚砜或磺酰基;n代表1、2或3;A代表直链键或较低烷基;Z代表##STR2##其中R.sub.3代表氢或酰基;R.sub.4代表较低烷基、C.sub.3-C.sub.7-环烷基、碳环或杂环芳基、碳环或杂环芳基-较低烷基、或C.sub.3-C.sub.7-环烷基-较低烷基;或Z代表##STR3##其中X代表氧或硫,R.sub.3代表氢、酰基、较低烷氧羰基、氨基羰基、单-或双-较低烷基氨基羰基、较低烯基氨基羰基、较低炔基氨基羰基、碳环或杂环芳基-较低烷基氨基羰基、碳环或杂环芳胺基羰基;C.sub.3-C.sub.7-环烷基氨基羰基,或C.sub.3-C.sub.7-环烷基-较低烷基氨基羰基;R.sub.5代表较低烷基、较低烷氧羰基-较低烷基、C.sub.3-C.sub.7-环烷基、碳环或杂环芳基、碳环或杂环芳基-较低烷基、C.sub.3-C.sub.7-环烷基-较低烷基、氨基、单-或双-较低烷基氨基、较低烯基氨基、较低炔基氨基、碳环或杂环芳基-较低烷基氨基、碳环或杂环芳胺基、C.sub.3-C.sub.7-环烷基氨基、C.sub.3-C.sub.7-环烷基-较低烷基氨基、较低烷氧羰基-较低烷基氨基,或较低烷氧基;R.sub.6和R.sub.7独立地代表氢或较低烷基;及其药学上可接受的盐;这些化合物可用作5-脂氧酶抑制剂。
-
Palladium catalyzed cross-coupling reaction of Grignard reagents with halobenzoic acids, halophenols and haloanilines作者:Nikolai A. Bumagin、Elena V. LuzikovaDOI:10.1016/s0022-328x(96)06794-0日期:1997.4Convenient syntheses of substituted benzoic acids, phenols and anilines have been achieved by using pall cross-coupling reactions between Grignard reagents and aryl halides containing carboxy, hydroxy and amino groups without a protection-deprotection sequence.
-
Tyrosine phosphatase inhibitors申请人:——公开号:US20030144338A1公开(公告)日:2003-07-31A compound of the formula (I): 1 wherein X 1 and X 2 are the same or different and each is a bond or a spacer having 1 to 20 atom(s) in the main chain; one of R 1 and R 2 is a cycle group having substituent(s) selected from 1) an optionally substituted carboxy-C 1-6 alkoxy group and 2) an optionally substituted carboxy-C 1-6 aliphatic hydrocarbon group, wherein the cycle group optionally has additional substituent(s), and the other is an optionally substituted cycle group or a hydrogen atom; and R 3 , R 4 and R 5 are the same or different and each is a hydrogen atom or a substituent, or R 4 may link together with R 3 or R 5 to form an optionally substituted ring; provided that when R 3 is a hydrogen atom, R 4 is a hydrogen atom and R 5 is methyl, X 2 —R 2 is not 4-cyclohexylphenyl; when R 3 is 4-methoxyphenyl, R 4 is a hydrogen atom and R 5 is methyl, X 2 —R 2 is not 4-methoxyphenyl; and when R 1 or R 2 is a hydrogen atom, the adjacent X 1 or X 2 is not a C 1-7 alkylene; or a salt thereof exhibits a protein tyrosine phosphatase inhibitory action and is useful as a prophylactic or therapeutic agent for diabetes or the like.式(I)的化合物: 其中X1和X2相同或不同,每个都是主链中具有1至20个原子的键或间隔物; R1和R2中的一个是具有取代基的环基,所述取代基选自1)可选择地取代的羧基-C1-6烷氧基和2)可选择地取代的羧基-C1-6脂肪烃基,其中所述环基可选择地具有额外的取代基,另一个是可选择地取代的环基或氢原子;以及 R3、R4和R5相同或不同,每个是氢原子或取代基,或R4可以与R3或R5结合形成可选择地取代的环; 但是当R3是氢原子时,R4是氢原子且R5是甲基时,X2—R2不是4-环己基苯基;当R3是4-甲氧基苯基,R4是氢原子且R5是甲基时,X2—R2不是4-甲氧基苯基;当R1或R2是氢原子时,相邻的X1或X2不是C1-7烷基; 或其盐具有蛋白酪氨酸磷酸酶抑制作用,并且可用作糖尿病等疾病的预防或治疗剂。
-
ISOCYANATE PRODUCTION PROCESS申请人:Shinohata Masaaki公开号:US20110092731A1公开(公告)日:2011-04-21An object of the present invention is to provide a process that enables isocyanate to be produced stably over a long period of time and at high yield without encountering problems of the prior art during production of isocyanate without using phosgene. The present invention discloses a process for producing an isocyanate by subjecting a carbamic acid ester to a thermal decomposition reaction, including the steps of: recovering a low boiling point component in a form of a gaseous phase component from a thermal decomposition reaction vessel in which the thermal decomposition reaction is carried out; recovering a liquid phase component containing a carbamic acid ester from a bottom of the thermal decomposition reaction vessel; and supplying all or a portion of the liquid phase component to an upper portion of the thermal decomposition reaction vessel.
-
PROCESS FOR PRODUCING ISOCYANATES USING DIARYL CARBONATE申请人:Shinohata Masaaki公开号:US20110054211A1公开(公告)日:2011-03-03An object of the present invention is to provide a process that enables isocyanate to be produced stably over a long period of time and at high yield without encountering problems of the prior art during production of isocyanate without using phosgene. The present invention provides an isocyanate production process including the steps of: obtaining a reaction mixture containing an aryl carbamate having an aryl group originating in a diaryl carbonate, an aromatic hydroxy compound originating in a diaryl carbonate, and a diaryl carbonate, by reacting a diaryl carbonate and an amine compound in the presence of a reaction solvent in the form of an aromatic hydroxy compound; transferring the reaction mixture to a thermal decomposition reaction vessel; and obtaining isocyanate by applying the aryl carbamate to a thermal decomposition reaction, wherein the reaction vessel in which the reaction between the diaryl carbonate and the amine compound is carried out and the thermal decomposition reaction vessel for the aryl carbamate are different.
表征谱图
-
氢谱1HNMR
-
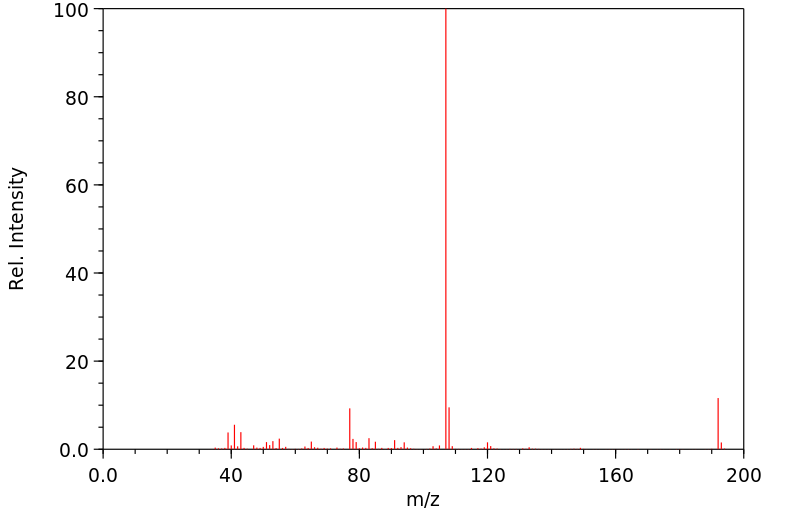
质谱MS
-
碳谱13CNMR
-
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(2-氯-6-羟基苯基)硼酸
黄柄曲菌素
高香草酸-d3
高香草酸-13C6
高香草酸
高香兰酸乙酯
高辣椒素II
高二氢辣椒素I
香草醛醛肟
香草醛苯腙
香草醛-甲氧基-13C
香草醛-(N-对甲苯基肟)
香草醛
香草酸肼
香草壬酰胺
香草基扁桃酸乙酯
香草吗啉
香草二乙胺
香兰素胺硬脂酸盐
香兰素胺硬脂酸盐
香兰素胺盐酸盐
香兰素丙二醇缩醛
香兰素13C6
香兰素-D3
香兰基乙基醚
香兰基丁醚
顺式-5-正十五碳-8'-烯基间苯二酚
顺式-1-(2-羟基-5-甲基苯基)-2-丁烯-1-酮
顺式-1-(2-羟基-4-甲氧基苯基)-2-丁烯-1-酮
顺-3-氯二氢-5-苯基呋喃-2(3H)-酮
雌二醇杂质1
降二氢辣椒碱
阿诺洛尔
阿瓦醇
阿普斯特杂质
间苯二酚双(二苯基磷酸酯)
间苯二酚-烯丙醇聚合物
间苯二酚-D6
间苯二酚
间苯三酚甲醛
间苯三酚二水合物
间苯三酚
间羟基苯乙基溴
间硝基苯酚
间甲酚紫钠盐
间甲酚与对甲酚和苯酚甲醛树脂的聚合物
间甲酚-D7
间甲酚-D3
间甲酚
间溴苯酚