3,4-diphenyl-1,2,4-oxadiazol-5(4H)-one | 14562-01-7
中文名称
——
中文别名
——
英文名称
3,4-diphenyl-1,2,4-oxadiazol-5(4H)-one
英文别名
4,5-Dihydro-3,4-diphenyl-1,2,4-oxadiazolon-(5);3,4-diphenyl-1,2,4-oxadiazol-5-one
CAS
14562-01-7
化学式
C14H10N2O2
mdl
——
分子量
238.246
InChiKey
YOWASPMXEXKPDM-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:170 °C
-
沸点:342.6±25.0 °C(Predicted)
-
密度:1.24±0.1 g/cm3(Predicted)
计算性质
-
辛醇/水分配系数(LogP):3.2
-
重原子数:18
-
可旋转键数:2
-
环数:3.0
-
sp3杂化的碳原子比例:0.0
-
拓扑面积:41.9
-
氢给体数:0
-
氢受体数:3
反应信息
-
作为反应物:描述:3,4-diphenyl-1,2,4-oxadiazol-5(4H)-one 在 palladium 10% on activated carbon 、 氢气 、 溶剂黄146 作用下, 以 乙酸乙酯 为溶剂, 20.0 ℃ 、111.46 kPa 条件下, 反应 20.0h, 以94%的产率得到N-苯基苄脒参考文献:名称:通过环化捕获原位生成的氰胺离子和腈氧化物合成恶二唑-5-亚胺。摘要:氨酰胺阴离子和几种腈氧化物的前所未有的分子间环化捕获能够合成恶二唑-5-亚胺。DOI:10.1021/acs.orglett.6b00203
-
作为产物:描述:benzohydroximoyl chloride 在 盐酸 、 水 、 cesium fluoride 作用下, 以 甲醇 、 水 、 乙腈 为溶剂, 反应 9.0h, 生成 3,4-diphenyl-1,2,4-oxadiazol-5(4H)-one参考文献:名称:通过环化捕获原位生成的氰胺离子和腈氧化物合成恶二唑-5-亚胺。摘要:氨酰胺阴离子和几种腈氧化物的前所未有的分子间环化捕获能够合成恶二唑-5-亚胺。DOI:10.1021/acs.orglett.6b00203
文献信息
-
13C NMR substituent-induced chemical shifts in 4-(substituted phenyl)-3-phenyl-1,2,4-oxadiazol-5(4H)-ones (thiones)作者:Yesim Saniye KaraDOI:10.1016/j.saa.2015.04.081日期:2015.104-(substituted phenyl)-3-phenyl-1,2,4-oxadiazol-5(4H)-ones and ten 4-(substituted phenyl)-3-phenyl-1,2,4-oxadiazol-5(4H)-thiones were synthesized. These oxadiazole derivatives were characterized by IR, (1)H NMR, (13)C NMR and elemental analyses. Their (13)C NMR spectra were measured in Deuterochloroform (CDCl3). The correlation analysis for the substituent-induced chemical shift (SCS) with Hammett substituent目前,主要研究新颖的十个4-(取代的苯基)-3-苯基-1,2,4-恶二唑-5(4H)-和十个4-(取代的苯基)-3-苯基-1,2,合成了4-恶二唑-5(4H)-硫酮。这些恶二唑衍生物通过IR,(1)H NMR,(13)C NMR和元素分析来表征。在氘代氯仿(CDCl3)中测量了它们的(13)C NMR光谱。取代基引起的化学位移(SCS)与哈米特取代基常数(σ),冈本布朗取代基常数(σ(+),σ(-)),感应取代基常数(σI)和不同的共振取代基常数之间的相关性分析(使用SSP(单取代基参数),DSP(双取代基参数)和DSP-NLR(双取代基参数-非线性共振)方法以及单次和多次回归分析执行σR,σR(o)。发现所有相关性均为负ρ值(反向取代基效应)。所有统计分析的结果,恶二唑环的CN,CO和CS碳的(13)C NMR化学位移均显示出令人满意的相关性。
-
Tuning chemoselectivity in <i>O</i>-/<i>N</i>-arylation of 3-aryl-1,2,4-oxadiazolones with <i>ortho</i>-(trimethylsilyl)phenyl triflates <i>via</i> aryne insertion作者:Lijing Zhou、Hongji Li、Wenge Zhang、Lei WangDOI:10.1039/c8cc00124c日期:——
A finely tunable chemoselectivity in arylation of 3-aryl-1,2,4-oxadiazolones with
ortho -(trimethylsilyl)phenyl triflates is reported, including silver-catalysedO -arylation and metal-freeN -arylation. -
Copper‐Catalyzed Selective N‐Arylation of Oxadiazolones by Diaryliodonium Salts作者:Natalia S. Soldatova、Artem V. Semenov、Kirill K. Geyl、Sergey V. Baykov、Anton A. Shetnev、Anna S. Konstantinova、Mikhail M. Korsakov、Mekhman S. Yusubov、Pavel S. PostnikovDOI:10.1002/adsc.202100426日期:2021.7.20Here, we report the method for copper-catalyzed N-arylation of diverse oxadiazolones by diaryliodonium salts under mild conditions in high yields (up to 92%) using available CuI as a catalyst. The developed method allows utilizing both symmetric and unsymmetric diaryliodonium salts bearing auxiliary groups such as 2,4,6-trimethoxyphenyl (TMP). We found that the steric effects in aryl moieties determined在这里,我们报告了使用可用的 CuI 作为催化剂在温和条件下以高产率(高达 92%)通过二芳基碘盐对不同恶二唑酮进行铜催化 N-芳基化的方法。所开发的方法允许使用带有辅助基团(例如 2,4,6-三甲氧基苯基 (TMP))的对称和不对称二芳基碘盐。我们发现芳基部分的空间效应决定了 1,2,4-oxadiazol-5(4 H )-ones的 N-和 O-芳基化的化学选择性。甲基取代的二芳基碘盐显示出作为选择性芳基化试剂的巨大潜力。结构研究表明 1,2,4-oxadiazol-5(4 H)-对空间位阻二芳基碘鎓盐的芳基化的影响。还证明了所提出方法的合成应用对 1,3,4-恶二唑-2(3 H )-酮和 1,2,4-恶二唑-5-硫醇的选择性芳基化。
-
A convenient one-pot synthesis of <i>N</i>-substituted amidoximes and their application toward 1,2,4-oxadiazol-5-ones作者:Wong Phakhodee、Chuthamat Duangkamol、Nitaya Wiriya、Mookda PattarawarapanDOI:10.1039/c8ra08207c日期:——The first direct one-pot approach for the synthesis of N-substituted amidoximes from secondary amides or the intermediate amides has been developed. Through the Ph3P–I2-mediated dehydrative condensation, a variety of N-aryl and N-alkyl amidoximes (R1(CNOH)NHR2, where R1 or R2 = aryl, alkyl, or benzyl) were readily afforded under mild conditions and short reaction times. The synthetic application of
-
Site Selectivity in the Synthesis of <i>O</i>-Methylated Hydroxamic Acids with Diazomethane作者:Antonella Leggio、Angelo Liguori、Anna Napoli、Carlo Siciliano、Giovanni SindonaDOI:10.1021/jo0012391日期:2001.4.1this paper we report the results obtained by treating some selected hydroxamic acids with diazomethane in ethereal media. The multitask reagent diazomethane was used either as a base to induce deprotonation of the chosen hydroxamic acids or as conjugated acid which undergoes one-pot methylation processes of the generated anions. Product distributions clearly showed that a high site selectivity is expressed
表征谱图
-
氢谱1HNMR
-
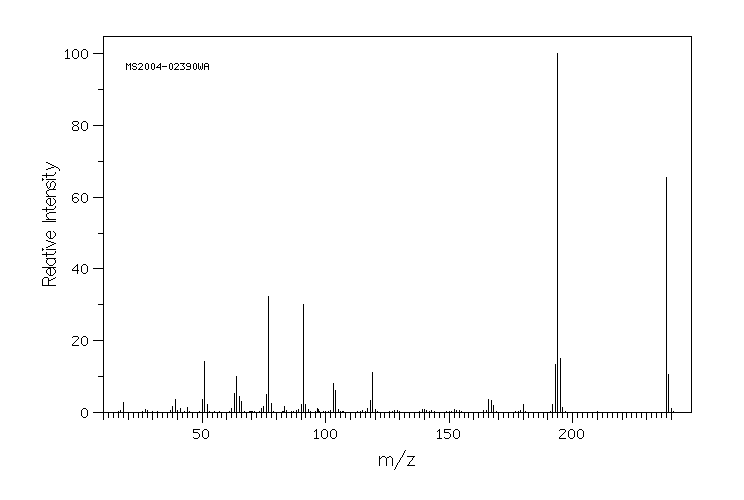
质谱MS
-
碳谱13CNMR
-
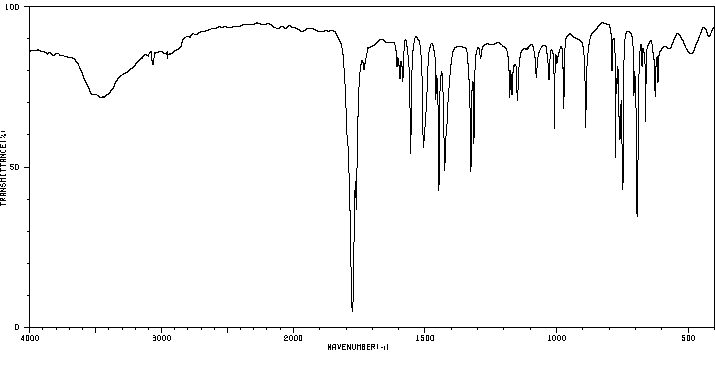
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(SP-4-1)-二氯双(1-苯基-1H-咪唑-κN3)-钯
(5aS,6R,9S,9aR)-5a,6,7,8,9,9a-六氢-6,11,11-三甲基-2-(2,3,4,5,6-五氟苯基)-6,9-甲基-4H-[1,2,4]三唑[3,4-c][1,4]苯并恶嗪四氟硼酸酯
(5-氨基-1,3,4-噻二唑-2-基)甲醇
齐墩果-2,12-二烯[2,3-d]异恶唑-28-酸
黄曲霉毒素H1
高效液相卡套柱
非昔硝唑
非布索坦杂质Z19
非布索坦杂质T
非布索坦杂质K
非布索坦杂质E
非布索坦杂质D
非布索坦杂质67
非布索坦杂质65
非布索坦杂质64
非布索坦杂质61
非布索坦代谢物67M-4
非布索坦代谢物67M-2
非布索坦代谢物 67M-1
非布索坦-D9
非布索坦
非唑拉明
雷非那酮-d7
雷西那德杂质2
雷西纳德杂质L
雷西纳德杂质H
雷西纳德杂质B
雷西纳德
雷西奈德杂质
阿西司特
阿莫奈韦
阿考替胺杂质9
阿米苯唑
阿米特罗13C2,15N2
阿瑞匹坦杂质
阿格列扎
阿扎司特
阿尔吡登
阿塔鲁伦中间体
阿培利司N-1
阿哌沙班杂质26
阿哌沙班杂质15
阿可替尼
阿作莫兰
阿佐塞米
镁(2+)(Z)-4'-羟基-3'-甲氧基肉桂酸酯
锌1,2-二甲基咪唑二氯化物
锌(II)(苯甲醇)(四苯基卟啉)
锌(II)(正丁醇)(四苯基卟啉)
锌(II)(异丁醇)(四苯基卟啉)