1-(9-溴菲-3-基)乙酮 | 6328-08-1
中文名称
1-(9-溴菲-3-基)乙酮
中文别名
——
英文名称
3-Acetyl-9-brom-phenanthren
英文别名
3-Acetyl-9-bromophenanthrene;1-(9-bromophenanthren-3-yl)ethanone
CAS
6328-08-1
化学式
C16H11BrO
mdl
——
分子量
299.167
InChiKey
PODNEAAXQLNICQ-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
计算性质
-
辛醇/水分配系数(LogP):4.8
-
重原子数:18
-
可旋转键数:1
-
环数:3.0
-
sp3杂化的碳原子比例:0.06
-
拓扑面积:17.1
-
氢给体数:0
-
氢受体数:1
安全信息
-
海关编码:2914700090
SDS
上下游信息
反应信息
-
作为反应物:描述:1-(9-溴菲-3-基)乙酮 在 溴 、 sodium hydroxide 、 盐酸 作用下, 以 1,4-二氧六环 、 水 为溶剂, 反应 1.0h, 以85%的产率得到9-bromophenanthrene-3-carboxylic acid参考文献:名称:Piperazine-2,3-dicarboxylic Acid Derivatives as Dual Antagonists of NMDA and GluK1-Containing Kainate Receptors摘要:Competitive N-methyl-D-aspartate receptor (NMDAR) antagonists bind to the GluN2 subunit, of which there are four types (GluN2A-D). We report that some N-1-substituted derivatives of cis-piperazine-2,3-dicarboxylic acid display improved relative affinity for GluN2C and GluN2D versus GluN2A and GluN2B. These derivatives also display subtype selectivity among the more distantly related kainate receptor family. Compounds 18i and (-)-4 were the most potent kainate receptor antagonists, and 18i was selective for GluK1 versus GluK2, GluK3 and AMPA receptors. Modeling studies revealed structural features required for activity at GluK1 subunits and suggested that S674 was vital for antagonist activity. Consistent with this hypothesis, replacing the equivalent residue in GluK3 (alanine) with a serine imparts 18i antagonist activity. Antagonists with dual GluN2D and GluK1 antagonist activity may have beneficial effects in various neurological disorders. Consistent with this idea, antagonist 18i (30 mg/kg ip) showed antinociceptive effects in an animal model of mild nerve injury.DOI:10.1021/jm201230z
-
作为产物:描述:参考文献:名称:Piperazine-2,3-dicarboxylic Acid Derivatives as Dual Antagonists of NMDA and GluK1-Containing Kainate Receptors摘要:Competitive N-methyl-D-aspartate receptor (NMDAR) antagonists bind to the GluN2 subunit, of which there are four types (GluN2A-D). We report that some N-1-substituted derivatives of cis-piperazine-2,3-dicarboxylic acid display improved relative affinity for GluN2C and GluN2D versus GluN2A and GluN2B. These derivatives also display subtype selectivity among the more distantly related kainate receptor family. Compounds 18i and (-)-4 were the most potent kainate receptor antagonists, and 18i was selective for GluK1 versus GluK2, GluK3 and AMPA receptors. Modeling studies revealed structural features required for activity at GluK1 subunits and suggested that S674 was vital for antagonist activity. Consistent with this hypothesis, replacing the equivalent residue in GluK3 (alanine) with a serine imparts 18i antagonist activity. Antagonists with dual GluN2D and GluK1 antagonist activity may have beneficial effects in various neurological disorders. Consistent with this idea, antagonist 18i (30 mg/kg ip) showed antinociceptive effects in an animal model of mild nerve injury.DOI:10.1021/jm201230z
文献信息
-
Synthesis of a Series of Novel 3,9-Disubstituted Phenanthrenes as Analogues of Known N-Methyl-d-aspartate Receptor Allosteric Modulators作者:Mark Irvine、Guangyu Fang、Richard Eaves、Maria Mayo-Martin、Erica Burnell、Blaise Costa、Georgia Culley、Arturas Volianskis、Graham Collingridge、Daniel Monaghan、David JaneDOI:10.1055/s-0034-1380114日期:2015.6
9-Substituted phenanthrene-3-carboxylic acids have been reported to have allosteric modulatory activity at the N-methyl-d-aspartate (NMDA) receptor. This receptor is activated by the excitatory neurotransmitter l-glutamate and has been implicated in a range of neurological disorders such as schizophrenia, epilepsy and chronic pain, and in neurodegenerative disorders such as Alzheimer’s disease. Herein, the convenient synthesis of a wide range of novel 3,9-disubstituted phenanthrene derivatives starting from a few common intermediates is described. These new phenanthrene derivatives will help to clarify the structural requirements for allosteric modulation of the NMDA receptor.
-
ALIPHATIC CHEMISTRY OF FLUORENE: PART III. SOME DERIVATIVES OF FLUORENE AND PHENANTHRENE作者:P. M. G. BavinDOI:10.1139/v60-128日期:1960.6.1Methyl fluorene-9-carboxylate and 9-methyl-9-acetylfluorene have been nitrated and the products converted to 2-nitrophenanthrene and 2-nitro-9,10-dimethylphenanthrene, respectively.Acetylation of 9,10-dimethylphenanthrene has given only one ketone, probably the 3-isomer. 4-Nitrofluorenone has been synthesized by a convenient route.
-
[EN] COMPOUNDS FOR USE IN MODULATING THE NMDA RECEPTOR<br/>[FR] MODULATEURS POSITIFS ET NÉGATIFS DES RÉCEPTEURS NMDA申请人:UNIV NEBRASKA公开号:WO2012019106A8公开(公告)日:2012-09-27
-
ATTEMPTS TO FIND NEW ANTIMALARIALS. XII. DERIVATIVES OF PHENANTHRENE, IV. 1 (OR 8)-ACETYL-9-HALOPHENANTHRENE作者:J. SCHULTZ、M. A. GOLDBERG、E. P. ORDAS、G. CARSCHDOI:10.1021/jo01174a004日期:1946.7
-
STUDIES IN THE PHENANTHRENE SERIES. II. PHENANTHRENE CARBOXYLIC ACIDS AND 9-BROMOPHENANTHRENE DERIVATIVES<sup>1</sup>作者:Erich Mosettig、Jacob van de KampDOI:10.1021/ja01347a046日期:1932.8
表征谱图
-
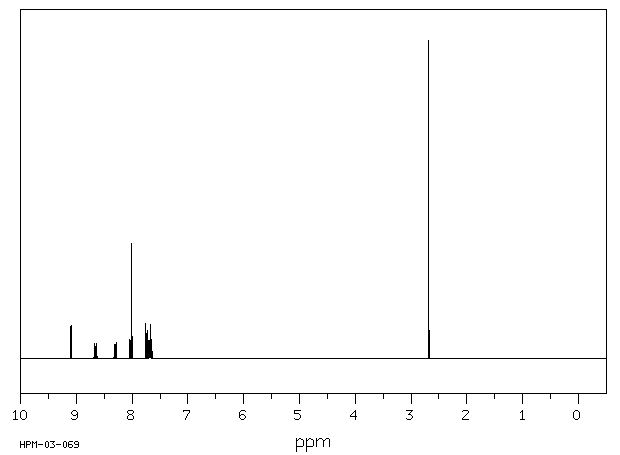
氢谱1HNMR
-
质谱MS
-
碳谱13CNMR
-
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(R)-2,2'',3,3''-四氢-6,6''-二-9-菲基-1,1''-螺双[1H-茚]-7,7''-二醇
(6,6)-苯基-C61己酸甲酯
高雌二醇
马兜铃酸钠
马兜铃酸盐
马兜铃酸C
马兜铃酸B
马兜铃酸(1:1MIXTUREOFARISTOLOCHICACIDIANDARISTOLOCHICACIDII)
马兜铃酸 Ia
马兜铃酸 IVa
马兜铃酸
颜料黑32
颜料红179
颜料红178
颜料红149
颜料红123
顺式-菲-1,2-二醇-3,4-环氧化物
顺式-苯并(a)屈-11,12-二醇-13,14-环氧化物
雷公藤酚A
镁二(1,4,5,6,7,16,17,18,19,19,20,20-十二氯六环[14.2.1.14,7.02,15.03,8.09,14]二十-5,9,11,13,17-五烯-11-磺酸酯)
钩大青酮
钩大青酮
钙(2+)12-羟基十八烷酸酯
酒石酸布托诺啡
那布扶林
还原红32
足球烯
贝那他汀B
贝母兰素
萘并[2,3-b]荧蒽
萘并[2,1-e][1]苯并二硫杂环戊烷
萘并[2,1-C:7,8-C']二菲
萘并[1,2-e][2]苯并呋喃-1,3-二酮
萘并[1,2-b]屈
萘并[1,2-a]蒽
萘并[1,2-B]菲-6-醇
萘二(六氯环戊二烯)加合物
萘,8-溴-1,2,3-三(1,1-二甲基乙基)-6-甲基-
菲醌单缩氨基硫脲
菲醌
菲并[9,10]呋喃
菲并[9,10-e]醋菲烯
菲并[4,5-bcd]噻吩
菲并[4,5-bcd]呋喃-3-醇
菲并[4,3-d]-1,3-二噁唑-5-羧酸,10-羟基-9-甲氧基-6-硝基-
菲并[3,2-b]噻吩
菲并[2,1-d]噻唑
菲并[2'',1'',10'':4,5,6;7'',8'',9'':4',5',6']二异喹啉并[2,1-a:2',1'-a']二萘嵌间二氮杂苯-8,13-二酮
菲并(3,4-b)噻吩
菲并(1,2-b)噻吩