4-甲烷磺酰氨基苯腈 | 36268-67-4
中文名称
4-甲烷磺酰氨基苯腈
中文别名
4-甲磺酰氨基苯腈;4-甲磺酰胺基苯腈
英文名称
4-((methylsulfonyl)amino)benzonitrile
英文别名
N-(4-cyanobenzyl) methanesulfonamide;N-(4-cyanophenyl)methanesulfonamide;4-cyano methanesulphonanilide;4-mesylaminobenzonitrile;4-methanesulfonamino-1-benzonitrile;4-(Methanesulfonylamino)benzonitrile
CAS
36268-67-4
化学式
C8H8N2O2S
mdl
MFCD00204724
分子量
196.23
InChiKey
BRDHOCVMWSXEHI-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:191-194 °C
-
沸点:365.5±44.0 °C(Predicted)
-
密度:1.36±0.1 g/cm3(Predicted)
-
稳定性/保质期:
常规情况下不会分解,没有危险反应。
计算性质
-
辛醇/水分配系数(LogP):0.9
-
重原子数:13
-
可旋转键数:2
-
环数:1.0
-
sp3杂化的碳原子比例:0.125
-
拓扑面积:78.3
-
氢给体数:1
-
氢受体数:4
安全信息
-
危险等级:6.1
-
危险品标志:Xn
-
危险类别码:R20/21/22
-
危险品运输编号:UN 3276
-
海关编码:2935009090
-
包装等级:III
-
危险类别:6.1
-
安全说明:S36/37
-
危险性防范说明:P261,P280,P311
-
危险性描述:H302,H312,H331
-
储存条件:密封、阴凉、干燥、通风保存
SDS
| Name: | 4-(Methanesulfonylamino)benzonitrile Material Safety Data Sheet |
| Synonym: | None Known |
| CAS: | 36268-67-4 |
Synonym:None Known
Section 2 - COMPOSITION, INFORMATION ON INGREDIENTS
| CAS# | Chemical Name | content | EINECS# |
| 36268-67-4 | 4-(Methanesulfonylamino)benzonitrile | 98% | unlisted |
Risk Phrases: 20/21/22
Section 3 - HAZARDS IDENTIFICATION
EMERGENCY OVERVIEW
Harmful by inhalation, in contact with skin and if swallowed.The toxicological properties of this material have not been fully investigated.
Potential Health Effects
Eye:
May cause eye irritation.
Skin:
May cause skin irritation. Harmful if absorbed through the skin. May be metabolized to cyanide which in turn acts by inhibiting cytochrome oxidase impairing cellular respiration.
Ingestion:
Harmful if swallowed. May cause irritation of the digestive tract.
The toxicological properties of this substance have not been fully investigated. Metabolism may release cyanide, which may result in headache, dizziness, weakness, collapse, unconsciousness and possible death.
Inhalation:
Harmful if inhaled. May cause respiratory tract irritation. The toxicological properties of this substance have not been fully investigated. May be metabolized to cyanide which in turns act by inhibiting cytochrome oxidase impairing cellular respiration.
Chronic:
Chronic exposure to cyanide solutions may lead to the development of a "cyanide" rash, characterized by itching, and by macular, papular, and vesicular eruptions, and may be accompanied by secondary infections. Exposure to small amounts of cyanide compounds over long periods of time is reported to cause loss of appetite, headache, weakness, nausea, dizziness, and symptoms of irritation of the upper respiratory tract and eyes.
Section 4 - FIRST AID MEASURES
Eyes: In case of contact, immediately flush eyes with plenty of water for at least 15 minutes. Get medical aid.
Skin:
In case of contact, flush skin with plenty of water. Remove contaminated clothing and shoes. Get medical aid if irritation develops and persists. Wash clothing before reuse.
Ingestion:
If swallowed, do not induce vomiting unless directed to do so by medical personnel. Never give anything by mouth to an unconscious person. Get medical aid.
Inhalation:
If inhaled, remove to fresh air. If not breathing, give artificial respiration. If breathing is difficult, give oxygen. Get medical aid.
Notes to Physician:
Treat symptomatically and supportively.
Section 5 - FIRE FIGHTING MEASURES
General Information:
As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear. During a fire, irritating and highly toxic gases may be generated by thermal decomposition or combustion.
Extinguishing Media:
Use foam, dry chemical, or carbon dioxide.
Section 6 - ACCIDENTAL RELEASE MEASURES
General Information: Use proper personal protective equipment as indicated in Section 8.
Spills/Leaks:
Vacuum or sweep up material and place into a suitable disposal container. Clean up spills immediately, observing precautions in the Protective Equipment section. Avoid generating dusty conditions.
Provide ventilation.
Section 7 - HANDLING and STORAGE
Handling:
Wash thoroughly after handling. Minimize dust generation and accumulation. Avoid breathing dust, vapor, mist, or gas. Avoid contact with eyes, skin, and clothing. Avoid contact with skin and eyes. Keep container tightly closed. Avoid ingestion and inhalation.
Use only in a chemical fume hood.
Storage:
Store in a cool, dry place. Store in a tightly closed container.
Section 8 - EXPOSURE CONTROLS, PERSONAL PROTECTION
Engineering Controls:
Facilities storing or utilizing this material should be equipped with an eyewash facility and a safety shower. Use adequate ventilation to keep airborne concentrations low.
Exposure Limits CAS# 36268-67-4: Personal Protective Equipment Eyes: Wear appropriate protective eyeglasses or chemical safety goggles as described by OSHA's eye and face protection regulations in 29 CFR 1910.133 or European Standard EN166.
Skin:
Wear appropriate protective gloves to prevent skin exposure.
Clothing:
Wear appropriate protective clothing to prevent skin exposure.
Respirators:
Follow the OSHA respirator regulations found in 29 CFR 1910.134 or European Standard EN 149. Use a NIOSH/MSHA or European Standard EN 149 approved respirator if exposure limits are exceeded or if irritation or other symptoms are experienced.
Section 9 - PHYSICAL AND CHEMICAL PROPERTIES
Physical State: Crystalline powder
Color: beige - fine
Odor: Not available.
pH: Not available.
Vapor Pressure: Not available.
Viscosity: Not available.
Boiling Point: Not available.
Freezing/Melting Point: 191 - 194 deg C
Autoignition Temperature: Not available.
Flash Point: Not available.
Explosion Limits, lower: Not available.
Explosion Limits, upper: Not available.
Decomposition Temperature:
Solubility in water:
Specific Gravity/Density:
Molecular Formula: C8H8N2O2S
Molecular Weight: 196.1282
Section 10 - STABILITY AND REACTIVITY
Chemical Stability:
Not currently available.
Conditions to Avoid:
Incompatible materials, dust generation, excess heat.
Incompatibilities with Other Materials:
Strong oxidizing agents, strong reducing agents, strong acids, strong bases.
Hazardous Decomposition Products:
Nitrogen oxides, carbon monoxide, oxides of sulfur, carbon dioxide.
Hazardous Polymerization: Has not been reported
Section 11 - TOXICOLOGICAL INFORMATION
RTECS#:
CAS# 36268-67-4 unlisted.
LD50/LC50:
Not available.
Carcinogenicity:
4-(Methanesulfonylamino)benzonitrile - Not listed by ACGIH, IARC, or NTP.
Section 12 - ECOLOGICAL INFORMATION
Section 13 - DISPOSAL CONSIDERATIONS
Dispose of in a manner consistent with federal, state, and local regulations.
Section 14 - TRANSPORT INFORMATION
IATA
Shipping Name: TOXIC SOLID, ORGANIC, N.O.S.
Hazard Class: 6.1
UN Number: 2811
Packing Group: III
IMO
Shipping Name: TOXIC SOLID, ORGANIC, N.O.S.
Hazard Class: 6.1
UN Number: 2811
Packing Group: III
RID/ADR
Shipping Name: TOXIC SOLID, ORGANIC, N.O.S.*
Hazard Class: 6.1
UN Number: 2811
Packing group: III
Section 15 - REGULATORY INFORMATION
European/International Regulations
European Labeling in Accordance with EC Directives
Hazard Symbols: XN
Risk Phrases:
R 20/21/22 Harmful by inhalation, in contact with
skin and if swallowed.
Safety Phrases:
S 36/37 Wear suitable protective clothing and
gloves.
WGK (Water Danger/Protection)
CAS# 36268-67-4: No information available.
Canada
None of the chemicals in this product are listed on the DSL/NDSL list.
CAS# 36268-67-4 is not listed on Canada's Ingredient Disclosure List.
US FEDERAL
TSCA
CAS# 36268-67-4 is not listed on the TSCA inventory.
It is for research and development use only.
SECTION 16 - ADDITIONAL INFORMATION
N/A
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 —— N-(4-cyanophenyl)-N-(methylsulfonyl)methanesulfonamide —— C9H10N2O4S2 274.321 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 N-[4-(氨基甲基)苯基]-甲烷磺酰胺 N-<4-(aminomethyl)phenyl>methanesulfonamide 129872-50-0 C8H12N2O2S 200.261 —— N-(4-cyanophenyl)-N-(methylsulfonyl)methanesulfonamide —— C9H10N2O4S2 274.321 N'-羟基-4-[(甲基磺酰基)氨基]苯甲脒 4-((methylsulfonyl)amino)benzenecarboxamide oxime 148459-01-2 C8H11N3O3S 229.26
反应信息
-
作为反应物:描述:参考文献:名称:Derivatized alkanolamines as cardiovascular agents摘要:以下结构式##STR1##的新型衍生烷醇胺被描述为有用的心血管药物。特别描述了它们作为表现出抗心律失常作用的心血管药物的有用性。所述的抗心律失常作用属于II类/III类结合型。还描述了含有这类化合物的药物配方。公开号:US05051423A1
-
作为产物:描述:参考文献:名称:Sklyarova, I. V.; Garabadzhiu, A. V.; Ginzburg, O. F., Journal of Organic Chemistry USSR (English Translation), 1986, vol. 22, p. 578 - 582摘要:DOI:
文献信息
-
Mild hypervalent iodine mediated oxidative nitration of N-aryl sulfonamides作者:Ulrich Kloeckner、Boris J. NachtsheimDOI:10.1039/c4cc04738a日期:——An oxidative and acid-free method for the nitration of N-aryl sulfonamides has been developed using a combination of sodium nitrite as cheap and easy to handle NO2-source and the hypervalent iodine reagent PIFA as stoichiometric oxidant. Under very mild reaction conditions, the desired mononitrated aryl sulfonamides were isolated in up to 87% yield. This is the first example of an iodane-mediated oxidative nitration.
-
Synthesis and serotonergic activity of 5-(oxadiazolyl)tryptamines: potent agonists for 5-HT1D receptors作者:Leslie J. Street、Raymond Baker、Jose L. Castro、Mark S. Chambers、Alexander R. Guiblin、Sarah C. Hobbs、Victor G. Matassa、Austin J. Reeve、Margaret S. BeerDOI:10.1021/jm00063a003日期:1993.5The selectivity of these compounds for 5-HT1D receptors over other serotonergic receptors is discussed. Sulfonamide 20t shows > or = 1000-fold selectivity for 5-HT1D over 5-HT2, 5-HT1C, and 5-HT3 receptors and 10-fold selectivity with respect to 5-HT1A receptors. The functional activity of this series of compounds is studied and demonstrates high 5-HT1D receptor potency and efficacy comparable to that描述了一系列新的5-(恶二唑基)色胺的合成和5-HT1D受体活性。研究了恶二唑3-取代基的修饰,连接链的长度(n)和胺取代基,并揭示了5-HT1D受体域中的大结合口袋。容纳恶二唑取代基如苄基而不会损失激动剂的效力或功效。在苯基或苄基间隔基团上引入极性官能团会导致亲和力和功能效能提高10倍。当杂环与吲哚偶联时,观察到最佳的5-HT1D活性,苄基磺酰胺20t和20u代表了一些已知的最有效的5-HT1D激动剂。杂环中的S取代O导致效能进一步提高。删除恶二唑N-2不会降低活性,建议在此位置仅需要一个H键受体。讨论了这些化合物对5-HT1D受体相对于其他血清素能受体的选择性。磺酰胺20t对5-HT1D的选择性比5-HT2、5-HT1C和5-HT3受体高> 1000倍,对5-HT1A受体的选择性高10倍。研究了该系列化合物的功能活性,并证明了与5-HT相当的5-HT1D受体效能和功效。
-
Discovery of carboxyl-containing biaryl ureas as potent RORγt inverse agonists作者:Nannan Sun、Yafei Huang、Mingcheng Yu、Yunpeng Zhao、Ji-An Chen、Chenyu Zhu、Meiqi Song、Huimin Guo、Qiong Xie、Yonghui WangDOI:10.1016/j.ejmech.2020.112536日期:2020.9GSK805 (1) is a potent RORγt inverse agonist, but a drawback of 1 is its low solubility, leading to a limited absorption in high doses. We have explored detailed structure-activity relationship on the amide linker, biaryl and arylsulfonyl moieties of 1 trying to improve solubility while maintaining RORγt activity. As a result, a novel series of carboxyl-containing biaryl urea derivatives was discoveredGSK805(1)是一种强效RORγt反向激动剂,但缺点1是它的低溶解度,从而导致在高剂量有限的吸收。我们已经探索了1的酰胺连接基,联芳基和芳基磺酰基部分的详细结构-活性关系,试图在保持RORγt活性的同时提高溶解度。结果,发现了一系列新颖的含羧基联芳基脲衍生物,作为具有改进的类药物性质的有效RORγt反向激动剂。化合物3i显示了强大的RORγt抑制活性和亚型选择性,在RORγFRET分析中的IC 50为63.8 nM,在基于细胞的RORγ-GAL4启动子报告子分析中为85 nM。合理的3i抑制活性在小鼠Th17细胞分化试验中也获得了抑制(在0.3μM时抑制76%)。此外,与1相比,3i在pH 7.4时具有大大改善的水溶解度,表现出体面的小鼠PK曲线,并在咪喹莫特诱导的牛皮癣小鼠模型中表现出一定的体内功效。
-
Meta Selective C–H Borylation of Sterically Biased and Unbiased Substrates Directed by Electrostatic Interaction作者:Jagriti Chaturvedi、Chabush Haldar、Ranjana Bisht、Gajanan Pandey、Buddhadeb ChattopadhyayDOI:10.1021/jacs.1c01770日期:2021.5.26An electrostatically directed meta borylation of sterically biased and unbiased substrates is described. The borylation follows an electrostatic interaction between the partially positive and negative charges between the ligand and substrate. With this strategy, it has been demonstrated that a wide number of challenging substrates, especially 4-substituted substrates, can selectively be borylated at
-
[EN] CHEMICAL COMPOUNDS<br/>[FR] COMPOSES CHIMIQUES申请人:ASTRAZENECA AB公开号:WO2004011410A1公开(公告)日:2004-02-05Compounds of formula (I):wherein variable groups are as defined within; for use in the inhibition of 11βHSD1 are described.式(I)的化合物:其中变量基团如定义内;用于抑制11βHSD1。
表征谱图
-
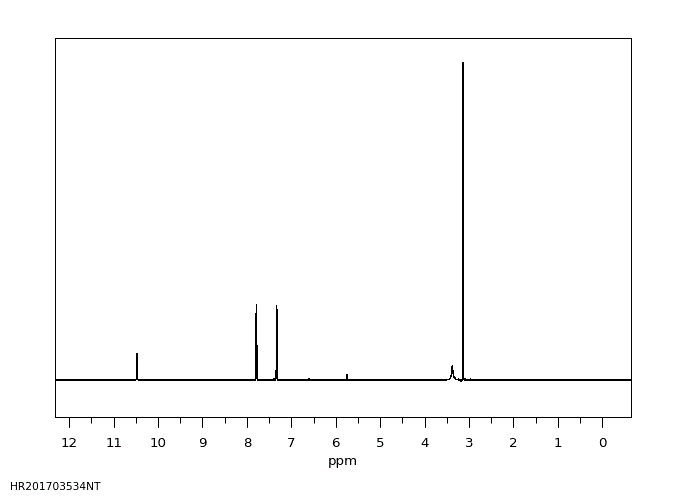
氢谱1HNMR
-
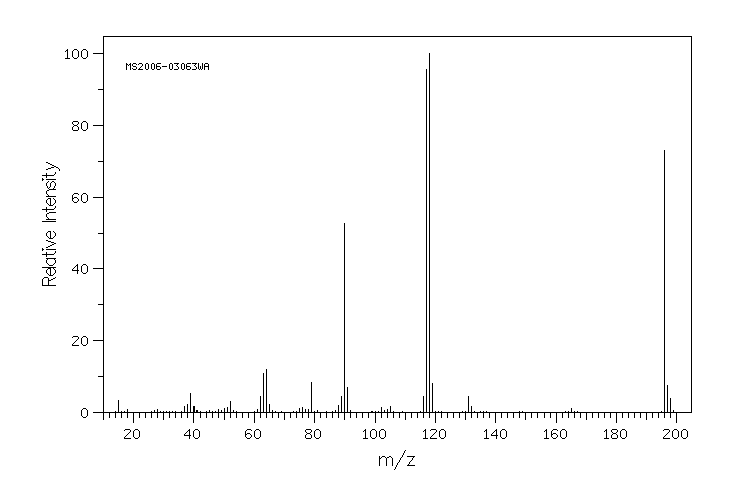
质谱MS
-
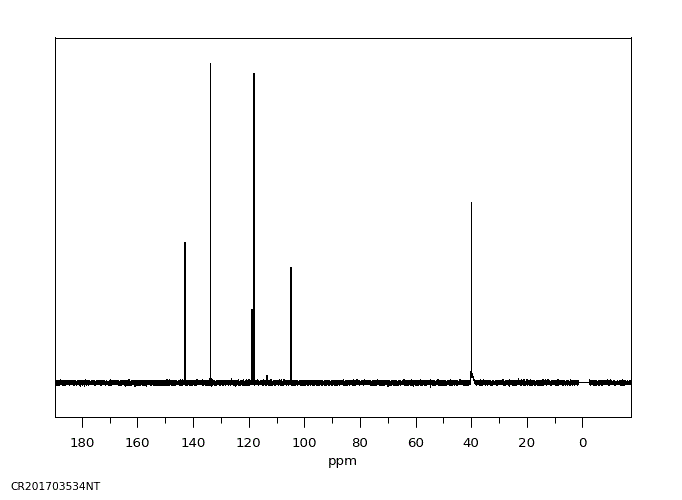
碳谱13CNMR
-
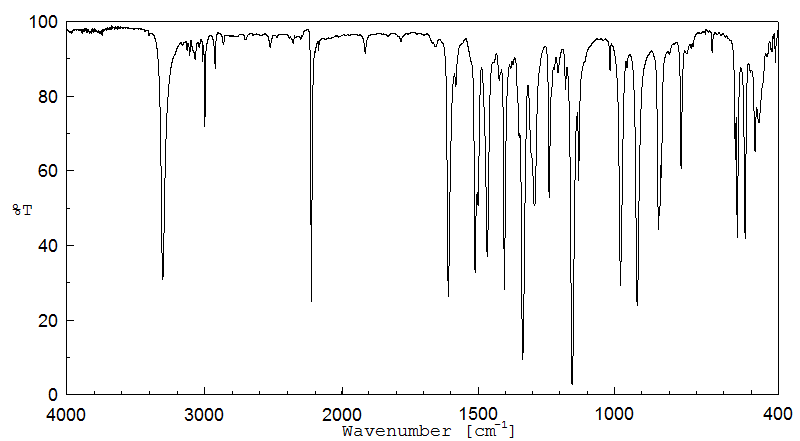
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(βS)-β-氨基-4-(4-羟基苯氧基)-3,5-二碘苯甲丙醇
(S,S)-邻甲苯基-DIPAMP
(S)-(-)-7'-〔4(S)-(苄基)恶唑-2-基]-7-二(3,5-二-叔丁基苯基)膦基-2,2',3,3'-四氢-1,1-螺二氢茚
(S)-盐酸沙丁胺醇
(S)-3-(叔丁基)-4-(2,6-二甲氧基苯基)-2,3-二氢苯并[d][1,3]氧磷杂环戊二烯
(S)-2,2'-双[双(3,5-三氟甲基苯基)膦基]-4,4',6,6'-四甲氧基联苯
(S)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(R)富马酸托特罗定
(R)-(-)-盐酸尼古地平
(R)-(-)-4,12-双(二苯基膦基)[2.2]对环芳烷(1,5环辛二烯)铑(I)四氟硼酸盐
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[((6-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(4-叔丁基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(3-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-4,7-双(3,5-二-叔丁基苯基)膦基-7“-[(吡啶-2-基甲基)氨基]-2,2”,3,3'-四氢1,1'-螺二茚满
(R)-3-(叔丁基)-4-(2,6-二苯氧基苯基)-2,3-二氢苯并[d][1,3]氧杂磷杂环戊烯
(R)-2-[((二苯基膦基)甲基]吡咯烷
(R)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(N-(4-甲氧基苯基)-N-甲基-3-(1-哌啶基)丙-2-烯酰胺)
(5-溴-2-羟基苯基)-4-氯苯甲酮
(5-溴-2-氯苯基)(4-羟基苯基)甲酮
(5-氧代-3-苯基-2,5-二氢-1,2,3,4-oxatriazol-3-鎓)
(4S,5R)-4-甲基-5-苯基-1,2,3-氧代噻唑烷-2,2-二氧化物-3-羧酸叔丁酯
(4S,4''S)-2,2''-亚环戊基双[4,5-二氢-4-(苯甲基)恶唑]
(4-溴苯基)-[2-氟-4-[6-[甲基(丙-2-烯基)氨基]己氧基]苯基]甲酮
(4-丁氧基苯甲基)三苯基溴化磷
(3aR,8aR)-(-)-4,4,8,8-四(3,5-二甲基苯基)四氢-2,2-二甲基-6-苯基-1,3-二氧戊环[4,5-e]二恶唑磷
(3aR,6aS)-5-氧代六氢环戊基[c]吡咯-2(1H)-羧酸酯
(2Z)-3-[[(4-氯苯基)氨基]-2-氰基丙烯酸乙酯
(2S,3S,5S)-5-(叔丁氧基甲酰氨基)-2-(N-5-噻唑基-甲氧羰基)氨基-1,6-二苯基-3-羟基己烷
(2S,2''S,3S,3''S)-3,3''-二叔丁基-4,4''-双(2,6-二甲氧基苯基)-2,2'',3,3''-四氢-2,2''-联苯并[d][1,3]氧杂磷杂戊环
(2S)-(-)-2-{[[[[3,5-双(氟代甲基)苯基]氨基]硫代甲基]氨基}-N-(二苯基甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[((1S,2S)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[[((1R,2R)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2-硝基苯基)磷酸三酰胺
(2,6-二氯苯基)乙酰氯
(2,3-二甲氧基-5-甲基苯基)硼酸
(1S,2S,3S,5S)-5-叠氮基-3-(苯基甲氧基)-2-[(苯基甲氧基)甲基]环戊醇
(1S,2S,3R,5R)-2-(苄氧基)甲基-6-氧杂双环[3.1.0]己-3-醇
(1-(4-氟苯基)环丙基)甲胺盐酸盐
(1-(3-溴苯基)环丁基)甲胺盐酸盐
(1-(2-氯苯基)环丁基)甲胺盐酸盐
(1-(2-氟苯基)环丙基)甲胺盐酸盐
(1-(2,6-二氟苯基)环丙基)甲胺盐酸盐
(-)-去甲基西布曲明
龙蒿油
龙胆酸钠
龙胆酸叔丁酯
龙胆酸
龙胆紫-d6
龙胆紫