1-溴-4,4,4-三氟丁烷 | 406-81-5
中文名称
1-溴-4,4,4-三氟丁烷
中文别名
4-溴-1,1,1-三氟丁烷
英文名称
1-Bromo-4,4,4-trifluorobutane
英文别名
4,4,4-trifluorobutyl bromide;4-bromo-1,1,1-trifluorobutane
CAS
406-81-5
化学式
C4H6BrF3
mdl
——
分子量
190.991
InChiKey
DBCAQXHNJOFNGC-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
沸点:103 °C
-
密度:1.555
-
闪点:103-105°C
计算性质
-
辛醇/水分配系数(LogP):2.8
-
重原子数:8
-
可旋转键数:2
-
环数:0.0
-
sp3杂化的碳原子比例:1.0
-
拓扑面积:0
-
氢给体数:0
-
氢受体数:3
安全信息
-
危险等级:IRRITANT
-
危险品标志:Xi
-
安全说明:S26,S36
-
危险类别码:R36/37/38
-
危险品运输编号:UN 1993
-
海关编码:2903799090
-
包装等级:II
-
储存条件:室温下,请密封保存。
SDS
| Name: | 1-Bromo-4 4 4-trifluorobutane 99% Material Safety Data Sheet |
| Synonym: | 4,4,4-Trifluorobutyl bromid |
| CAS: | 406-81-5 |
Synonym:4,4,4-Trifluorobutyl bromid
Section 2 - COMPOSITION, INFORMATION ON INGREDIENTS
| CAS# | Chemical Name | content | EINECS# |
| 406-81-5 | 1-Bromo-4,4,4-trifluorobutane | 99% | unlisted |
Risk Phrases: 36/37/38
Section 3 - HAZARDS IDENTIFICATION
EMERGENCY OVERVIEW
Irritating to eyes, respiratory system and skin.
Potential Health Effects
Eye:
Causes eye irritation.
Skin:
Causes skin irritation. May be harmful if absorbed through the skin.
Ingestion:
May cause irritation of the digestive tract. May be harmful if swallowed.
Inhalation:
Causes respiratory tract irritation. May be harmful if inhaled.
Chronic:
Not available.
Section 4 - FIRST AID MEASURES
Eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid.
Skin:
Get medical aid. Flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes.
Ingestion:
Get medical aid. Wash mouth out with water.
Inhalation:
Remove from exposure and move to fresh air immediately. If not breathing, give artificial respiration. If breathing is difficult, give oxygen. Get medical aid.
Notes to Physician:
Treat symptomatically and supportively.
Section 5 - FIRE FIGHTING MEASURES
General Information:
As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear.
Extinguishing Media:
Use water spray, dry chemical, carbon dioxide, or chemical foam.
Section 6 - ACCIDENTAL RELEASE MEASURES
General Information: Use proper personal protective equipment as indicated in Section 8.
Spills/Leaks:
Absorb spill with inert material (e.g. vermiculite, sand or earth), then place in suitable container.
Section 7 - HANDLING and STORAGE
Handling:
Avoid breathing dust, vapor, mist, or gas. Avoid contact with skin and eyes. Use only in a chemical fume hood.
Storage:
Store in a cool, dry place. Store in a tightly closed container.
Section 8 - EXPOSURE CONTROLS, PERSONAL PROTECTION
Engineering Controls:
Facilities storing or utilizing this material should be equipped with an eyewash facility and a safety shower. Use adequate ventilation to keep airborne concentrations low.
Exposure Limits CAS# 406-81-5: Personal Protective Equipment Eyes: Not available.
Skin:
Wear appropriate protective gloves to prevent skin exposure.
Clothing:
Wear appropriate protective clothing to prevent skin exposure.
Respirators:
Follow the OSHA respirator regulations found in 29 CFR 1910.134 or European Standard EN 149. Use a NIOSH/MSHA or European Standard EN 149 approved respirator if exposure limits are exceeded or if irritation or other symptoms are experienced.
Section 9 - PHYSICAL AND CHEMICAL PROPERTIES
Physical State: Liquid
Color: clear to slight yellow
Odor: organohalide-like - characteristic odor
pH: Not available.
Vapor Pressure: Not available.
Viscosity: Not available.
Boiling Point: 103 - 105 deg C @ 760mmHg
Freezing/Melting Point: Not available.
Autoignition Temperature: Not available.
Flash Point: Not available.
Explosion Limits, lower: Not available.
Explosion Limits, upper: Not available.
Decomposition Temperature:
Solubility in water:
Specific Gravity/Density: 1.3817
Molecular Formula: C4H6BrF3
Molecular Weight: 190.99
Section 10 - STABILITY AND REACTIVITY
Chemical Stability:
Not available.
Conditions to Avoid:
Incompatible materials.
Incompatibilities with Other Materials:
Strong oxidizing agents, strong reducing agents, strong acids, strong bases.
Hazardous Decomposition Products:
Carbon monoxide, carbon dioxide, hydrogen fluoride gas, hydrogen bromide.
Hazardous Polymerization: Has not been reported.
Section 11 - TOXICOLOGICAL INFORMATION
RTECS#:
CAS# 406-81-5 unlisted.
LD50/LC50:
Not available.
Carcinogenicity:
1-Bromo-4,4,4-trifluorobutane - Not listed by ACGIH, IARC, or NTP.
Section 12 - ECOLOGICAL INFORMATION
Section 13 - DISPOSAL CONSIDERATIONS
Dispose of in a manner consistent with federal, state, and local regulations.
Section 14 - TRANSPORT INFORMATION
IATA
Not regulated as a hazardous material.
IMO
Not regulated as a hazardous material.
RID/ADR
Not regulated as a hazardous material.
Section 15 - REGULATORY INFORMATION
European/International Regulations
European Labeling in Accordance with EC Directives
Hazard Symbols: XI
Risk Phrases:
R 36/37/38 Irritating to eyes, respiratory system
and skin.
Safety Phrases:
S 26 In case of contact with eyes, rinse immediately
with plenty of water and seek medical advice.
S 37/39 Wear suitable gloves and eye/face
protection.
WGK (Water Danger/Protection)
CAS# 406-81-5: No information available.
Canada
None of the chemicals in this product are listed on the DSL/NDSL list.
CAS# 406-81-5 is not listed on Canada's Ingredient Disclosure List.
US FEDERAL
TSCA
CAS# 406-81-5 is not listed on the TSCA inventory.
It is for research and development use only.
SECTION 16 - ADDITIONAL INFORMATION
N/A
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 4,4,4-三氟丁醇 4,4,4-trifluorobutanol 461-18-7 C4H7F3O 128.094 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 4,4,4-三氟-1-碘丁烷 1,1,1-trifluoro-4-iodo-butane 461-17-6 C4H6F3I 237.991
反应信息
-
作为反应物:描述:参考文献:名称:WO2006/122200摘要:公开号:
-
作为产物:描述:参考文献:名称:什么时候三氟甲基比甲基更具亲脂性?脂族醇和三氟醇的分配系数和选定的化学位移。摘要:确定了12种三氟甲基化脂肪醇及其非氟化对应物的辛醇-水分配系数。后一个值是在建立适当的相关方程后,使用苯甲醇-水溶剂系统的测量值得出的。附带地,发现了经验方程,其允许在给定分子式和沸点的情况下估计未取代的醇的分配系数。仅当三氟甲基位于α-位时,三氟强烈增强亲脂性。对于β-和γ-(三氟甲基)醇而言,这种增强几乎无法测量,而δ-和ε-(三氟甲基)化合物比其母体化合物具有更大的亲水性。化学位移的比较表明,相对亲脂性的变化主要受三氟甲基对羟基酸碱性的诱导作用控制。提出了用于获得一些醇的新的合成方法。DOI:10.1002/jps.2600751016
-
作为试剂:描述:(S)-3-(6-hydroxybenzo[d]isoxazol-3-yl)-5-(methoxymethyl)oxazolidin-2-one 、 1-溴-4,4,4-三氟丁烷 在 1-溴-4,4,4-三氟丁烷 作用下, 以76的产率得到(5S)-5-(甲氧基甲基)-3-[6-(4,4,4-三氟丁氧基)-1,2-苯并恶唑-3-基]-1,3-恶唑烷-2-酮参考文献:名称:Journal of labelled compounds and radiopharmaceuticals 2018, 61, 252 - 262摘要:DOI:
文献信息
-
2,4-diamino pyrimidine compounds having anti-cell proliferative activity申请人:AstraZeneca AB公开号:US06593326B1公开(公告)日:2003-07-15A pyrimidine derivative of formula (I): wherein: R1 is an optional substituent as defined within; Rx is selected from halo, hydroxy, nitro, amino, cyano, mercapto, carboxy, sulphamoyl, formamido, ureido or carbamoyl or a group of formula (Ib): A—B—C— as defined within; Q1 and Q2 are independently selected from aryl, a 5- or 6-membered monocyclic moiety; and a 9- or 10-membered bicyclic heterocyclic moiety; and one or both of Q1 and Q2 bears on any available carbon atom one substituent of formula (Ia) as defined within; and Q1 and Q2 are optionally further substituted; or a pharmaceutically acceptable salt or in vivo hydrolysable ester thereof; are useful as anti-cancer agents; and processes for their manufacture and pharmaceutical compositions containing them are described.
-
Npy antagonists, preparation and uses申请人:Botez Iuliana公开号:US20090233910A1公开(公告)日:2009-09-17The present invention concerns novel compounds, their preparation and their uses, therapeutic uses in particular. More specifically it concerns derivative compounds having at least two aromatic cycles, their preparation and their uses, in particular in the area of human or animal health. These compounds have an affinity for the biological receptors of neuropeptide Y, NPY, present in the central and peripheral nervous systems. The compounds of the invention are preferably NPY antagonists, and more particularly antagonists of sub-type NPY Y1, and can therefore be used for the therapeutic or prophylactic treatment of any disorder involving NPY. The present invention also concerns pharmaceutical compositions containing said compounds, their preparation and their uses, as well as treatment methods using said compounds.本发明涉及新颖化合物,它们的制备和用途,特别是在治疗方面的用途。更具体地说,它涉及至少具有两个芳香环的衍生化合物,它们的制备和用途,特别是在人类或动物健康领域。这些化合物对存在于中枢和外周神经系统中的神经肽Y(NPY)的生物受体具有亲和力。本发明的化合物优选为NPY拮抗剂,更具体地说是NPY Y1亚型的拮抗剂,因此可用于治疗或预防涉及NPY的任何疾病。本发明还涉及含有所述化合物的药物组合物,其制备和用途,以及使用所述化合物的治疗方法。
-
Chemo- and Regioselective Synthesis of Acyl-Cyclohexenes by a Tandem Acceptorless Dehydrogenation-[1,5]-Hydride Shift Cascade作者:Lewis B. Smith、Roly J. Armstrong、Daniel Matheau-Raven、Timothy J. DonohoeDOI:10.1021/jacs.9b12296日期:2020.2.5acceptorless dehydrogenation of the diol followed by a redox-neutral cascade process, which is independent of the iridium catalyst. Deuterium labeling studies established that the key step of this cascade involves a novel base-mediated [1,5]-hydride shift. The cyclohexenyl ketone products could readily be cleaved under mildly acidic conditions to access a range of valuable substituted cyclohexene derivatives
-
Platinum-Catalyzed Domino Reaction with Benziodoxole Reagents for Accessing Benzene-Alkynylated Indoles作者:Yifan Li、Jerome WaserDOI:10.1002/anie.201412321日期:2015.4.27platinum‐catalyzed cyclization/alkynylation domino process to selectively obtain C5‐ or C6‐functionalized indoles starting from easily available pyrroles. The work combines, for the first time, a platinum catalyst with ethynylbenziodoxole hypervalent iodine reagents in a domino process for the synthesis of polyfunctionalized arene rings and gives access to important building blocks for the synthesis of bioactive
-
GPR40 AGONISTS申请人:Kuo Gee-Hong公开号:US20080176912A1公开(公告)日:2008-07-24The invention is directed to compounds of Formula (I) useful as GPR40 agonists. Pharmaceutical compositions and methods of treating one or more conditions including, but not limited to, insulin resistance, hyperglycemia, obesity, diabetes such as NIDDM, and other disorders related to lipid metabolism, energy homeostasis, and complications thereof, using compounds of the invention are also described.
表征谱图
-
氢谱1HNMR
-
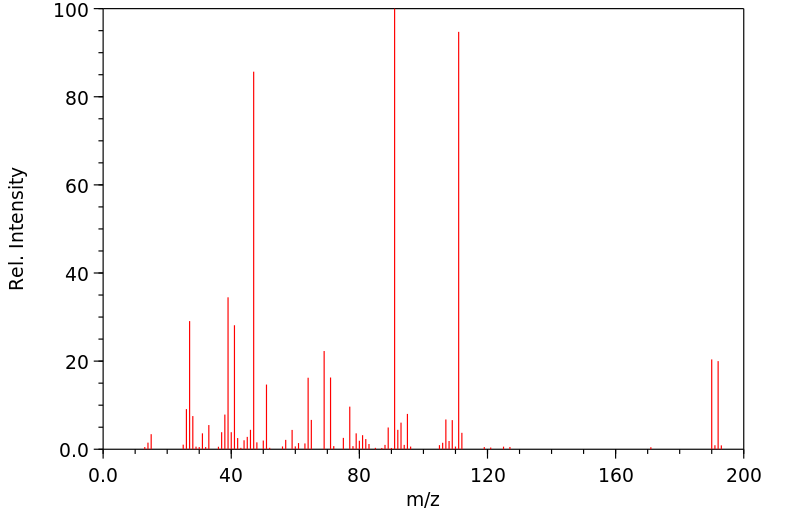
质谱MS
-
碳谱13CNMR
-
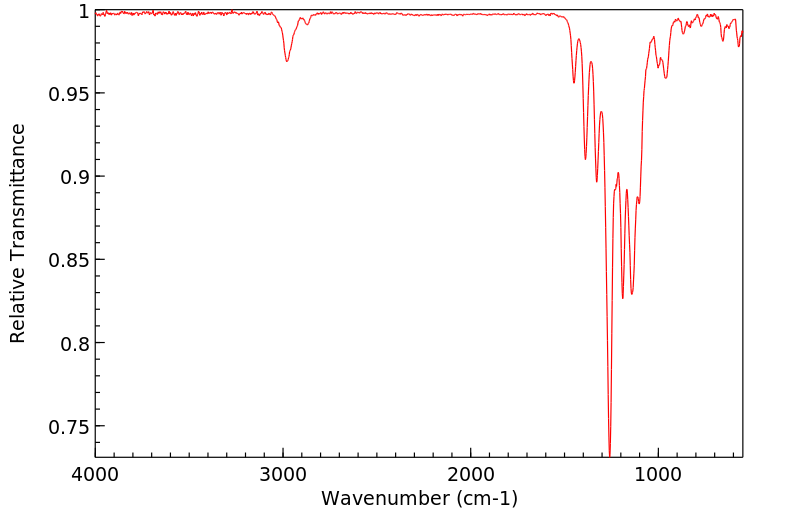
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
顺式-2-氟-环丙胺
顺式-1,1,1,4,4,4-六氟-2-丁烯
顺-1,1,2,2,3,4-六氟环丁烷
酰亚胺基二亚磷酸,甲基-,四(2,2,2-三氟乙基)酯
舒巴坦酸
聚(7-脱氮杂腺嘌呤酸)
癸烷,6-溴-1,1,1,2,2,3,3-七氟-4,4-二(三氟甲基)-
环丙基溴化镁
溴五氟乙烷
氯氟烃-252
氯氟烃-232
氯氟-甲基
氯四氟乙烷
氯二氟乙醛
氯三氟乙烷
氨甲酸,(氟磺酰)-,甲基酯
氢氯氟碳-261
氟甲醇
氟甲基自由基
氟甲基环戊烷
氟甲基环丙烷
氟环辛烷
氟环戊烷
氟环庚烷
氟环十二烷
氟环丁烷
1-溴-1-氯-2,2,2-三氟乙烷
氟氯乙烷
氟化烯丙基
氟化乙亚胺酰基,2-(二氟氨基)-N,2,2-三氟-
氟化丁基
氟乙醛
氟乙烷
氟乙烯醚
正膦胺,N-(2,3,4,5,6-五氯-2,3,4,5,6-五氟亚环己基)-1,1,1,1-四(2,2,3,3-四氟丙氧基)-
桉叶素
替氟烷
恩氟烷
异氟醚
异十八烷酸己酯
己酸,2,5-二氨基-6-羟基-(7CI)
奥替尼啶HCL
壬氟环戊烷
地氟烷
叔丁基氟化物
反式-2-氟环丙胺盐酸盐
反式-2-氟代环戊烷-1-胺盐酸盐
反式-1,2-双(全氟己基)乙烯
反式-1,2-双(全氟-n-丁基)乙烯
反式-1,1,1,2,2,3,3-七氟-4-壬烯