5-nitrofurfuraldoxime | 555-15-7
中文名称
——
中文别名
——
英文名称
5-nitrofurfuraldoxime
英文别名
Nifuroxime;N-[(5-nitrofuran-2-yl)methylidene]hydroxylamine
CAS
555-15-7
化学式
C5H4N2O4
mdl
——
分子量
156.098
InChiKey
PTBKFATYSVLSSD-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:160 °C
-
沸点:280.29°C (rough estimate)
-
密度:1.6814 (rough estimate)
-
溶解度:可溶于DMSO(少许)、甲醇(少许)
-
保留指数:2380
计算性质
-
辛醇/水分配系数(LogP):0.3
-
重原子数:11
-
可旋转键数:1
-
环数:1.0
-
sp3杂化的碳原子比例:0.0
-
拓扑面积:91.6
-
氢给体数:1
-
氢受体数:5
安全信息
-
危险等级:6.1
-
安全说明:S22,S24/25,S36/37
-
危险类别码:R22
-
海关编码:2932190090
-
包装等级:III
-
危险类别:6.1
-
危险品运输编号:UN 2811
-
储存条件:库房应保持通风、低温和干燥,并需与其他氧化剂、碱类及食品添加剂分开存放。
SDS
| Name: | Nifuroxime Material Safety Data Sheet |
| Synonym: | anti-5-Nitro-2-furaldoxime; 5-nitro-2-furaldoxime |
| CAS: | 555-15-7 |
Synonym:anti-5-Nitro-2-furaldoxime; 5-nitro-2-furaldoxime
Section 2 - COMPOSITION, INFORMATION ON INGREDIENTS
| CAS# | Chemical Name | content | EINECS# |
| 555-15-7 | Nifuroxime | ca. 100 | 209-082-4 |
Risk Phrases: None Listed.
Section 3 - HAZARDS IDENTIFICATION
EMERGENCY OVERVIEW
Light sensitive.
Potential Health Effects
Eye:
May cause eye irritation.
Skin:
May cause skin irritation.
Ingestion:
May cause irritation of the digestive tract.
Inhalation:
May cause respiratory tract irritation.
Chronic:
Not available.
Section 4 - FIRST AID MEASURES
Eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid.
Skin:
Flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes. Get medical aid if irritation develops or persists. Wash clothing before reuse.
Ingestion:
If victim is conscious and alert, give 2-4 cupfuls of milk or water.
Never give anything by mouth to an unconscious person. Get medical aid.
Inhalation:
Remove from exposure and move to fresh air immediately. If not breathing, give artificial respiration. If breathing is difficult, give oxygen. Get medical aid if cough or other symptoms appear.
Notes to Physician:
Antidote: None reported.
Section 5 - FIRE FIGHTING MEASURES
General Information:
As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear. During a fire, irritating and highly toxic gases may be generated by thermal decomposition or combustion.
Extinguishing Media:
Use water spray, dry chemical, carbon dioxide, or chemical foam.
Section 6 - ACCIDENTAL RELEASE MEASURES
General Information: Use proper personal protective equipment as indicated in Section 8.
Spills/Leaks:
Vacuum or sweep up material and place into a suitable disposal container. Clean up spills immediately, observing precautions in the Protective Equipment section. Avoid generating dusty conditions.
Provide ventilation.
Section 7 - HANDLING and STORAGE
Handling:
Wash thoroughly after handling. Minimize dust generation and accumulation. Avoid breathing dust, vapor, mist, or gas. Avoid contact with eyes, skin, and clothing. Keep container tightly closed. Avoid ingestion and inhalation. Use with adequate ventilation.
Storage:
Do not store in direct sunlight. Store in a tightly closed container. Store in a cool, dry, well-ventilated area away from incompatible substances.
Section 8 - EXPOSURE CONTROLS, PERSONAL PROTECTION
Engineering Controls:
Facilities storing or utilizing this material should be equipped with an eyewash facility and a safety shower. Use adequate ventilation to keep airborne concentrations low.
Exposure Limits CAS# 555-15-7: Personal Protective Equipment Eyes: Wear appropriate protective eyeglasses or chemical safety goggles as described by OSHA's eye and face protection regulations in 29 CFR 1910.133 or European Standard EN166.
Skin:
Wear appropriate protective gloves to prevent skin exposure.
Clothing:
Wear appropriate protective clothing to prevent skin exposure.
Respirators:
Follow the OSHA respirator regulations found in 29 CFR 1910.134 or European Standard EN 149. Use a NIOSH/MSHA or European Standard EN 149 approved respirator if exposure limits are exceeded or if irritation or other symptoms are experienced.
Section 9 - PHYSICAL AND CHEMICAL PROPERTIES
Physical State: Crystalline solid
Color: yellow
Odor: Not available.
pH: Not available.
Vapor Pressure: Not available.
Viscosity: Not available.
Boiling Point: Not available.
Freezing/Melting Point: 160 deg C dec
Autoignition Temperature: Not available.
Flash Point: Not available.
Explosion Limits, lower: N/A
Explosion Limits, upper: N/A
Decomposition Temperature:
Solubility in water:
Specific Gravity/Density:
Molecular Formula: C5H4N2O4
Molecular Weight: 156.10
Section 10 - STABILITY AND REACTIVITY
Chemical Stability:
Stable under normal temperatures and pressures. May discolor on exposure to light.
Conditions to Avoid:
Incompatible materials, light.
Incompatibilities with Other Materials:
Strong oxidizing agents.
Hazardous Decomposition Products:
Carbon monoxide, oxides of nitrogen, carbon dioxide.
Hazardous Polymerization: Has not been reported.
Section 11 - TOXICOLOGICAL INFORMATION
RTECS#:
CAS# 555-15-7: LT7525000 LD50/LC50:
Not available.
Carcinogenicity:
Nifuroxime - Not listed by ACGIH, IARC, or NTP.
Other:
See actual entry in RTECS for complete information.
Section 12 - ECOLOGICAL INFORMATION
Other No information available.
Section 13 - DISPOSAL CONSIDERATIONS
Dispose of in a manner consistent with federal, state, and local regulations.
Section 14 - TRANSPORT INFORMATION
IATA
Not regulated as a hazardous material.
IMO
Not regulated as a hazardous material.
RID/ADR
Not regulated as a hazardous material.
Section 15 - REGULATORY INFORMATION
European/International Regulations
European Labeling in Accordance with EC Directives
Hazard Symbols: Not available.
Risk Phrases:
Safety Phrases:
S 24/25 Avoid contact with skin and eyes.
WGK (Water Danger/Protection)
CAS# 555-15-7: No information available.
Canada
None of the chemicals in this product are listed on the DSL/NDSL list.
CAS# 555-15-7 is not listed on Canada's Ingredient Disclosure List.
US FEDERAL
TSCA
CAS# 555-15-7 is not listed on the TSCA inventory.
It is for research and development use only.
SECTION 16 - ADDITIONAL INFORMATION
N/A
制备方法与用途
上下游信息
反应信息
-
作为反应物:描述:5-nitrofurfuraldoxime 在 吡啶 、 盐酸 、 sodium hypochlorite 、 aluminum amalgam 、 sodium methylate 作用下, 以 四氢呋喃 、 1,4-二氧六环 、 N,N-二甲基甲酰胺 为溶剂, 生成 1-[5-(4-Amino-isoxazolo[5,4-d]pyrimidin-3-yl)-furan-2-yl]-3-(3-trifluoromethyl-phenyl)-urea参考文献:名称:Isothiazolopyrimidines and isoxazolopyrimidines as novel multi-targeted inhibitors of receptor tyrosine kinases摘要:A series of isothiazolopyrimidines and isoxazolopyrimidines were synthesized and identified as potent KDR inhibitors. SAR studies led to isothiazolopyrimidine urea analogs that potently inhibit VEGFR tyrosine kinases (KDR enzymatic and cellular IC50 values below 10 nM) as well as cKIT and TIE2. The selected compounds 8 and 13 display 56% and 48% oral bioavailability in mice, respectively. (c) 2006 Elsevier Ltd. All rights reserved.DOI:10.1016/j.bmcl.2006.05.057
-
作为产物:描述:(hydroxyamino)(5-nitrofuran-2-yl)methanol 在 potassium chloride 作用下, 以 水 为溶剂, 生成 5-nitrofurfuraldoxime参考文献:名称:芳香醛与羟基和非羟基胺反应的自发脱水机理摘要:糠醛和 5-硝基糠醛与羟胺、N-甲基羟胺和 O-甲基羟胺反应的脱水步骤的速率常数与 pH 值的关系图显示了对应于氧鎓离子催化和自发脱水的两个区域。糠醛与上述羟胺反应的氧鎓离子催化脱水区域表现出具有优异布朗斯台德相关性的一般酸催化(布朗斯台德系数:0.76 (r = 0.986)、0.68 (r = 0.987) 和 0.67 (r = 0.993))分别)。然而,这些羟胺的自发脱水的速率常数(其中水被认为是一般酸催化剂)与布朗斯台德线有很大的正偏差。其他作者在非羟基胺与不同芳香醛的反应中没有观察到这一事实,因此支持这些反应的自发脱水步骤是通过分子内催化进行的。分子内催化的机理可能是逐步的。首先,形成两性离子中间体。然后它可以在第二步中通过失水进化,或遵循协调的途径,质子通过五元环转移(一般分子内酸催化)。在非羟基胺的情况下,数据表明分子内质子通过一个或两个水分子从胺的氮转移到离开的氢氧根离子的机制的可能性。©DOI:10.1002/kin.10095
文献信息
-
Design, synthesis, antileishmanial, and antifungal biological evaluation of novel 3,5‐disubstituted isoxazole compounds based on 5‐nitrofuran scaffolds作者:Ozildéia S. Trefzger、Natália V. Barbosa、Renata L. Scapolatempo、Amarith R. Neves、Maria L. F. S. Ortale、Diego B. Carvalho、Antônio M. Honorato、Mariana R. Fragoso、Cristiane Y. K. Shuiguemoto、Renata T. Perdomo、Maria F. C. Matos、Marilene R. Chang、Carla C. P. Arruda、Adriano C. M. BaroniDOI:10.1002/ardp.201900241日期:2020.2Nineteen 3,5‐disubstituted‐isoxazole analogs were synthesized based on nitrofuran scaffolds, by a [3 + 2] cycloaddition reaction between terminal acetylenes and 5‐nitrofuran chloro‐oxime. The compounds were obtained in moderate to very good yields (45–91%). The antileishmanial activity was assayed against the promastigote and amastigote forms of Leishmania (Leishmania) amazonensis. Alkylchlorinated基于硝基呋喃支架,通过末端乙炔和 5-硝基呋喃氯肟之间的 [3 + 2] 环加成反应合成了 19 种 3,5-二取代的异恶唑类似物。以中等至非常好的产率 (45–91%) 获得这些化合物。针对前鞭毛体和无鞭毛体形式的利什曼原虫 (Leishmania) amazonensis 测定了抗利什曼原虫活性。烷基氯化化合物 14p–r 对前鞭毛体和无鞭毛体形式均有活性,重点是化合物 14p,它对无鞭毛体形式表现出很强的活性(IC50 = 0.6 μM 和选择性指数 [SI] = 5.2)。在烷基系列中,化合物 14o 以无鞭毛体形式的 IC50 = 8.5 μM 和 SI = 8.0 脱颖而出。在芳香族系列中,最活跃的化合物是那些含有电子给体基团的化合物,如三甲氧基异恶唑 14g (IC50 = 1. 2 μM 和 SI = 20.2);化合物 14 小时,IC50 = 7.0 μM,SI = 6
-
Synthesis and SAR Study of Simple Aryl Oximes and Nitrofuranyl Derivatives with Potent Activity Against Mycobacterium tuberculosis作者:Cristiane França da Costa、Marcus Vinicius Nora de Souza、Maria Cristina da Silva Lourenço、Elaine Soares Coimbra、Guilherme da Silva Lourenço Carvalho、James Wardell、Stephane Lima Calixto、Juliana da Trindade GranatoDOI:10.2174/1570180816666181227115738日期:2019.12.31Background: Oximes and nitrofuranyl derivatives are particularly important compounds in medicinal chemistry. Thus, many researchers have been reported to possess antibacterial, antiparasitic, insecticidal and fungicidal activities. Methods: In this work, we report the synthesis and the biological activity against Mycobacterium tuberculosis H37RV of a series of fifty aryl oximes, ArCH=N-OH, I, and eight背景:肟和硝基呋喃基衍生物是药物化学中特别重要的化合物。因此,据报道,许多研究人员具有抗菌、抗寄生虫、杀虫和杀真菌活性。方法:在这项工作中,我们报告了一系列五十种芳基肟 ArCH=N-OH, I 和八种硝基呋喃基化合物 2-硝基呋喃基-X, II 的合成和抗结核分枝杆菌 H37RV 的生物活性。结果:在肟中,I:Ar = 2-OH-4-OH,42 和 I:Ar = 5-硝基呋喃基,46,分别在 3.74 和 32.0 µM 处具有最佳活性。还有,46,硝基呋喃化合物,II;X = MeO,55 和 II:X = NHCH2Ph,58,(分别为 14.6 和 12.6 µM),表现出优异的生物活性并且无细胞毒性。结论:化合物55显示出9.85的选择性指数。使用化合物 55 进行了进一步的抗菌测试,该化合物对粪肠球菌、肺炎克雷伯菌、铜绿假单胞菌、金黄色葡萄球菌、鼠伤寒沙门氏菌和福氏志贺氏菌无活
-
Synthesis and Biological Activity of Nitro Heterocycles Analogous to Megazol, a Trypanocidal Lead作者:Gérard Chauvière、Bernard Bouteille、Bertin Enanga、Cristina de Albuquerque、Simon L. Croft、Michel Dumas、Jacques PériéDOI:10.1021/jm021030a日期:2003.1.1the two rings of the basic nucleus, replacement of the thiadiazole by an oxadiazole, replacement of the nitroimidazole part by a nitrofurane or a nitrothiophene, and substitutions on the exocyclic nitrogen atom for evaluation of an improved import by the glucose or the purine transporters. Assays of the series of compounds on the protozoan parasites Trypanosoma brucei, Trypanosoma cruzi, and Leishmania作为我们开发旨在治疗寄生虫感染的新化合物的努力的一部分,我们合成并测定了主要化合物megazol 5-(1-甲基-5-硝基-1H-2-咪唑基)-1,3的类似物, 4-噻二唑-2-胺,CAS号 19622-55-0),体外。我们首先开发了一条合成巨z的新途径。随后引入了一些结构变化,包括在碱性原子核的两个环上进行取代,用二恶唑取代噻二唑,用硝基呋喃或硝基噻吩取代硝基咪唑部分以及在环外氮原子上进行取代,以评估其改进之处。由葡萄糖或嘌呤转运蛋白进口。对原生动物寄生虫布鲁氏锥虫,克鲁斯锥虫和多形利什曼原虫的一系列化合物的分析,不论是细胞外细胞还是被感染的巨噬细胞,都表明兆唑比衍生物更具活性。然后,对感染了布氏锥虫锥虫的灵长类动物(包括晚期中枢神经系统感染和苏拉明)进行了Megazol评估。在研究中,五只猴子观察到完全恢复,在两年的随访中没有寄生虫病复发。由于缺乏有效的治疗非洲昏睡病和南美南美锥虫病
-
Montmorillonite clay Cu(II) catalyzed domino one-pot multicomponent synthesis of 3,5-disubstituted isoxazoles作者:Sandip B. Bharate、Anil K. Padala、Bashir A. Dar、Rammohan R. Yadav、Baldev Singh、Ram A. VishwakarmaDOI:10.1016/j.tetlet.2013.04.102日期:2013.7corresponding aldehydes and terminal alkynes using recyclable montmorillonite clay supported Cu(II)/NaN3 catalytic system under aqueous conditions have been developed. The ‘domino’ one-pot MCR approach involves hydroxyamination of aldehydes followed by chlorination and then generation of reactive ‘nitrile oxide’ which undergoes 1,3-dipolar cycloaddition with alkynes to produce 3,5-disubstituted isoxazoles
-
An Efficient Synthesis of Nitriles from Aldoximes Using Diethyl Phosphorocyanidate under Mild Conditions作者:Kie-Seung Lee、Hyeon-Seong An、Chan-Yeon HwangDOI:10.5012/bkcs.2012.33.10.3173日期:2012.10.202-chloro-1-methylpyridinium iodide is an efficient and mild reagent for the dehydration of aldoximes to nitriles under mild conditions. In continuation of our interest in developing mild method for the conversion of aldoximes to nitriles, we herein wish to report the first application of DEPC to the efficient synthesis of nitriles from aldoximes under mild conditions (Scheme 1).腈是有机合成中有价值的中间体,不仅因为它们是各种官能团的合适前体,而且因为它们被广泛用作药物、农用化学品和各种 N-杂环化合物的关键中间体。氰基本身也经常存在于许多重要的生物学分子中。因此,已经开发了多种从不同化学前体合成腈的路线。在这些路线中,使用合适的脱水剂从醛肟合成腈是一种通用且清洁的方法。为此,使用了许多试剂,例如氯磺酰异氰酸酯、二-2-吡啶基亚硫酸盐、Burgess 试剂、[RuCl2(对甲基异丙基苯)]2/MS 4A、4d BOP、Pd(OAc)2/PPh3、4f Cu(OAc) 2/超声4g已研制成功。然而,这些试剂 可能在某些方面存在局限性,例如苛刻的反应条件、使用昂贵或不易获得的试剂、产率低和缺乏通用性。因此,仍然需要开发用于这种转换的温和且通用的方法。磷氰酸二乙酯 (DEPC) 最初是作为一种有效的肽偶联剂引入的,并已被用于有用的有机反应。以前,我们报道了 2-chlo
表征谱图
-
氢谱1HNMR
-
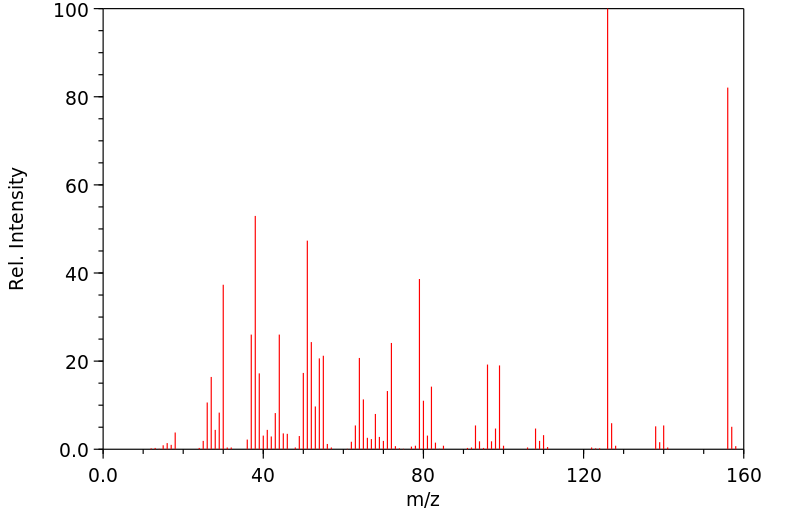
质谱MS
-
碳谱13CNMR
-
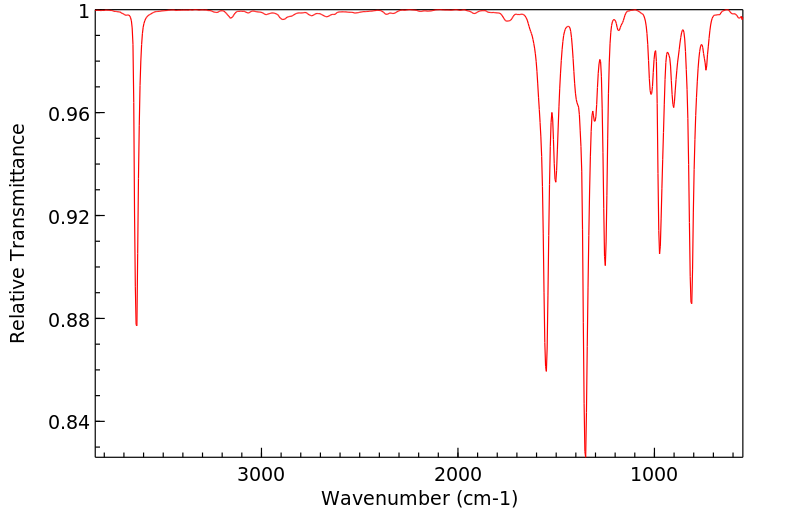
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
除草醚
锡烷,三丁基[(2-呋喃基羰基)氧代]-
醋糠硫胺
醋呋三嗪
酪氨酰-甘氨酰-色氨酰-蛋氨酰-门冬氨酰-苯基丙氨酰-甘氨酸
苯胺,N-[6-乙氧基-2,3-二(4-甲氧苯基)-4H-吡喃-4-亚基]-4-甲基-
糠酸(呋喃甲酸)
糠酸異戊酯
糠酸烯丙酯
碘化溴刚
硫代糠酸甲酯
硝基呋喃杂质
硝呋隆
硝呋醛肟标准品
硝呋达齐
硝呋美隆
硝呋维啶
硝呋立宗
硝呋甲醚
硝呋烯腙盐酸盐
硝呋烯腙
硝呋替莫
硝呋拉定
硝呋拉嗪
硝呋太尔杂质B
硝呋太尔杂质33
硝呋噻唑
硝呋吡醇
硝呋乙宗
盐酸呋喃它酮
盐酸呋喃他酮
疏呋那登
甲基7-[5-乙酰氨基-4-[(2-溴-4,6-二硝基苯基)偶氮]-2-甲氧苯基]-3-羰基-2,4,10-三氧杂-7-氮杂十一烷-11-酸酯
甲基5-溴-3-甲基-2-糠酸酯
甲基5-乙酰氨基-2-糠酸酯
甲基5-{[(氯乙酰基)氨基]甲基}-2-糠酸酯
甲基5-(甲氧基甲基)-2-甲基呋喃-3-羧酸酯
甲基5-(溴甲基)-4-(氯甲基)-2-糠酸酯
甲基5-(乙氧基甲基)-2-甲基-3-糠酸酯
甲基5-({[5-(三氟甲基)-2-吡啶基]硫代}甲基)-2-糠酸
甲基5-(4-甲酰基苯基)-2-糠酸酯
甲基5-(3-甲酰基苯基)-2-糠酸酯
甲基4-甲基-3-糠酸酯
甲基4-溴-5-甲基-2-糠酸酯
甲基4-乙酰基-5-甲基-2-糠酸酯
甲基4,6-二氯-3-(二乙基氨基)呋喃并[3,4-c]吡啶-1-羧酸酯
甲基3-羟基呋喃并[3,2-b]吡啶-2-羧酸酯
甲基3-甲酰基-2-糠酸酯
甲基3-氨基呋喃并[2,3-b]吡啶-2-羧酸酯
甲基3-氨基-5-(2-甲基-2-丙基)-2-糠酸酯