1-(3-氨基丙基)-2-吡咯烷酮 | 7663-77-6
中文名称
1-(3-氨基丙基)-2-吡咯烷酮
中文别名
N-(3-氨基丙基)-2-吡咯烷酮;N-(3'-氨丙基)-2-吡咯烷酮;N-(3'-丙胺基)-2-吡咯烷酮;1-(3-氨丙基)-2-吡咯烷酮;N-(3-丙胺基)-2-吡咯烷酮;N-(3-氨丙基)-2-吡咯烷酮
英文名称
1-(3-aminopropyl)-2-pyrrolidone
英文别名
1-(3-AMINOPROPYL)-2-PYRROLIDINONE;1-(3-aminopropyl)pyrrolidin-2-one;N-(3-aminopropyl)-2-pyrrolidinone
CAS
7663-77-6
化学式
C7H14N2O
mdl
MFCD00003201
分子量
142.201
InChiKey
HJORCZCMNWLHMB-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
沸点:120-123 °C1 mm Hg(lit.)
-
密度:1.014 g/mL at 25 °C(lit.)
-
闪点:>230 °F
-
溶解度:可溶于氯仿(少许)、甲醇(少许)
计算性质
-
辛醇/水分配系数(LogP):-0.8
-
重原子数:10
-
可旋转键数:3
-
环数:1.0
-
sp3杂化的碳原子比例:0.857
-
拓扑面积:46.3
-
氢给体数:1
-
氢受体数:2
安全信息
-
危险品标志:Xi,C
-
安全说明:S26,S27,S36/37/39,S45
-
危险类别码:R34
-
WGK Germany:3
-
RTECS号:UY5739500
-
海关编码:2933790090
-
危险标志:GHS05
-
危险品运输编号:UN 3267 8/PG 2
-
危险性描述:H314
-
危险性防范说明:P280,P305 + P351 + P338,P310
-
包装等级:II
-
危险类别:8
-
储存条件:| 冰箱 |
SDS
| Name: | N-(3 -Aminopropyl)-2-Pyrrolidinone 95% (GC) Material Safety Data Sheet |
| Synonym: | None |
| CAS: | 7663-77-6 |
Synonym:None
Section 2 - COMPOSITION, INFORMATION ON INGREDIENTS
| CAS# | Chemical Name | content | EINECS# |
| 7663-77-6 | N-(3'-Aminopropyl)-2-Pyrrolidinone | 95 | 231-632-7 |
Risk Phrases: None Listed.
Section 3 - HAZARDS IDENTIFICATION
EMERGENCY OVERVIEW
The toxicological properties of this material have not been fully investigated.
Potential Health Effects
Eye:
May cause eye irritation. The toxicological properties of this material have not been fully investigated.
Skin:
May cause skin irritation. The toxicological properties of this material have not been fully investigated.
Ingestion:
May cause irritation of the digestive tract. The toxicological properties of this substance have not been fully investigated.
Inhalation:
May cause respiratory tract irritation. The toxicological properties of this substance have not been fully investigated.
Chronic:
No information found.
Section 4 - FIRST AID MEASURES
Eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid.
Skin:
Get medical aid. Flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes. Wash clothing before reuse.
Ingestion:
Never give anything by mouth to an unconscious person. Get medical aid. Do NOT induce vomiting. If conscious and alert, rinse mouth and drink 2-4 cupfuls of milk or water.
Inhalation:
Remove from exposure and move to fresh air immediately. If not breathing, give artificial respiration. If breathing is difficult, give oxygen. Get medical aid.
Notes to Physician:
Section 5 - FIRE FIGHTING MEASURES
General Information:
As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear. During a fire, irritating and highly toxic gases may be generated by thermal decomposition or combustion. Use water spray to keep fire-exposed containers cool. Vapors may be heavier than air. They can spread along the ground and collect in low or confined areas. Containers may explode when heated.
Extinguishing Media:
Use agent most appropriate to extinguish fire. Cool containers with flooding quantities of water until well after fire is out. Use water spray, dry chemical, carbon dioxide, or appropriate foam.
Section 6 - ACCIDENTAL RELEASE MEASURES
General Information: Use proper personal protective equipment as indicated in Section 8.
Spills/Leaks:
Avoid runoff into storm sewers and ditches which lead to waterways.
Clean up spills immediately, observing precautions in the Protective Equipment section. Remove all sources of ignition. Absorb spill using an absorbent, non-combustible material such as earth, sand, or vermiculite. Do not use combustible materials such as sawdust.
Provide ventilation.
Section 7 - HANDLING and STORAGE
Handling:
Wash thoroughly after handling. Remove contaminated clothing and wash before reuse. Use with adequate ventilation. Avoid contact with eyes, skin, and clothing. Keep container tightly closed. Avoid ingestion and inhalation.
Storage:
Store in a tightly closed container. Store in a cool, dry, well-ventilated area away from incompatible substances.
Section 8 - EXPOSURE CONTROLS, PERSONAL PROTECTION
Engineering Controls:
Use adequate ventilation to keep airborne concentrations low.
Exposure Limits CAS# 7663-77-6: Personal Protective Equipment Eyes: Wear appropriate protective eyeglasses or chemical safety goggles as described by OSHA's eye and face protection regulations in 29 CFR 1910.133 or European Standard EN166.
Skin:
Wear appropriate protective gloves to prevent skin exposure.
Clothing:
Wear appropriate protective clothing to prevent skin exposure.
Respirators:
Follow the OSHA respirator regulations found in 29 CFR 1910.134 or European Standard EN 149. Use a NIOSH/MSHA or European Standard EN 149 approved respirator if exposure limits are exceeded or if irritation or other symptoms are experienced.
Section 9 - PHYSICAL AND CHEMICAL PROPERTIES
Physical State: Liquid
Color: clear, colorless
Odor: Not available.
pH: Not available.
Vapor Pressure: Not available.
Viscosity: Not available.
Boiling Point: 120.0 - 123.0 deg C @ 1.00mm
Freezing/Melting Point: Not available.
Autoignition Temperature: Not available.
Flash Point: > 112 deg C (> 233.60 deg F)
Explosion Limits, lower: Not available.
Explosion Limits, upper: Not available.
Decomposition Temperature:
Solubility in water:
Specific Gravity/Density: 1.0140g/cm3
Molecular Formula: C7H14N2O
Molecular Weight: 142.20
Section 10 - STABILITY AND REACTIVITY
Chemical Stability:
Stable under normal temperatures and pressures.
Conditions to Avoid:
Incompatible materials, excess heat, strong oxidants.
Incompatibilities with Other Materials:
Oxidizing agents.
Hazardous Decomposition Products:
Nitrogen oxides, carbon monoxide, irritating and toxic fumes and gases, carbon dioxide.
Hazardous Polymerization: Has not been reported
Section 11 - TOXICOLOGICAL INFORMATION
RTECS#:
CAS# 7663-77-6: UY5739500 LD50/LC50:
Not available.
Carcinogenicity:
N-(3'-Aminopropyl)-2-Pyrrolidinone - Not listed by ACGIH, IARC, or NTP.
Other:
See actual entry in RTECS for complete information.
Section 12 - ECOLOGICAL INFORMATION
Section 13 - DISPOSAL CONSIDERATIONS
Dispose of in a manner consistent with federal, state, and local regulations.
Section 14 - TRANSPORT INFORMATION
IATA
Not regulated as a hazardous material.
IMO
Not regulated as a hazardous material.
RID/ADR
Not regulated as a hazardous material.
Section 15 - REGULATORY INFORMATION
European/International Regulations
European Labeling in Accordance with EC Directives
Hazard Symbols: Not available.
Risk Phrases:
Safety Phrases:
S 24/25 Avoid contact with skin and eyes.
S 28A After contact with skin, wash immediately with
plenty of water.
S 37 Wear suitable gloves.
S 45 In case of accident or if you feel unwell, seek
medical advice immediately (show the label where
possible).
WGK (Water Danger/Protection)
CAS# 7663-77-6: No information available.
Canada
None of the chemicals in this product are listed on the DSL/NDSL list.
CAS# 7663-77-6 is not listed on Canada's Ingredient Disclosure List.
US FEDERAL
TSCA
CAS# 7663-77-6 is not listed on the TSCA inventory.
It is for research and development use only.
SECTION 16 - ADDITIONAL INFORMATION
N/A
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 2-氧代-1-吡咯烷PROPIONITRILE 3-(2-oxopyrrolidin-1-yl)propanenitrile 7663-76-5 C7H10N2O 138.169 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 N-[3-(2-氧代吡咯烷-1-基)丙基]乙酰胺 N-(3-acetamidopropyl)pyrrolidin-2-one 106692-36-8 C9H16N2O2 184.238 —— 1-(3-azidopropyl)pyrrolidin-2-one 674303-36-7 C7H12N4O 168.198 —— N-(3-isothiocyanatopropyl)-γ-butyrolactam 53485-01-1 C8H12N2OS 184.262 —— 3-(2-Oxopyrrolidin-1-yl)propylcarbamic acid 1333203-81-8 C8H14N2O3 186.211 —— N-Carbomethoxymethyl-N-(3-(2-oxopyrrolidin-1-yl)prop-1-yl)amine 280752-70-7 C10H18N2O3 214.265 —— 1-[3-(2-oxo-pyrrolidin-1-yl)-propyl]-piperazine-2,6-dione 1421691-43-1 C11H17N3O3 239.274
反应信息
-
作为反应物:参考文献:名称:Highly selective 2,4-diaminopyrimidine-5-carboxamide inhibitors of Sky kinase摘要:We report the SAR around a series of 2,4-diaminopyrimidine-5-carboxamide inhibitors of Sky kinase. 2-Aminophenethyl analogs demonstrate excellent potency but moderate kinase selectivity, while 2-aminobenzyl analogs that fill the Ala571 subpocket exhibit good inhibition activity and excellent kinase selectivity. (C) 2012 Elsevier Ltd. All rights reserved.DOI:10.1016/j.bmcl.2012.12.013
-
作为产物:描述:3-(烯丙基氨基)丙腈 在 dirhodium tetraacetate 、 lithium aluminium tetrahydride 、 三丁基膦 、 氢气 作用下, 以 苯 为溶剂, 80.0 ℃ 、2.76 MPa 条件下, 反应 20.0h, 生成 1-(3-氨基丙基)-2-吡咯烷酮参考文献:名称:Rhodium-catalysed hydroformylation and carbonylation of N-alkenyl-1,3-diaminopropanes摘要:The rhodium catalysed reactions of N-alkenyl-1,3-diaminopropanes with H-2/CO usually give mixtures of diazabicycloalkanes and aminopropyl lactams. The chemo-and regioselectivity of these reactions are influenced by the choice of ligand and by the ratio of H-2 and CO in the gas mixture and in some cases formation of a single compound can be achieved. (C) 1997 Elsevier Science Ltd.DOI:10.1016/s0040-4020(97)10193-4
-
作为试剂:描述:硫酸 、 水 、 uranyl(VI) acetate dihydrate 在 1-(3-氨基丙基)-2-吡咯烷酮 作用下, 反应 24.0h, 生成 uranium sulfate tetrahydrate参考文献:名称:四价金属硫酸盐四水合物的热化学研究:A 4+(SO 4)2(H 2 O)4(A 4+ = Zr,Ce,U)摘要:四种四价金属硫酸盐四水合物(A 4+(SO 4)2(H 2 O)4(A 4+ = Zr,Ce,U):α-Zr(SO)的形成焓的合成,表征和量热测量4)2(H 2 O)4,α-Ce(SO 4)2(H 2 O)4,β-Ce(SO 4)2(H 2 O)4和β-U(SO 4)2( H 2 O)4)报告。直接量热法测量表明,这些化合物从相应氧化物形成的焓与四价阳离子的八倍配位半径相关。在α-(Fddd)和β-(Pnma)形式之间观察到能量斜率变化,这与先前报道的研究一致。DOI:10.1016/j.jssc.2019.04.030
文献信息
-
Cell adhesion-inhibiting antiinflammatory and immune-suppressive compounds申请人:Abbott Laboratories公开号:US20040116518A1公开(公告)日:2004-06-17The present invention relates to novel cinnamide compounds that are useful for treating inflammatory and immune diseases and cerebral vasospasm, to pharmaceutical compositions containing these compounds, and to methods of inhibiting inflammation or suppressing immune response in a mammal.本发明涉及新型肉桂酰胺化合物,用于治疗炎症和免疫性疾病以及脑血管痉挛,以及含有这些化合物的药物组合物,以及在哺乳动物中抑制炎症或抑制免疫反应的方法。
-
LTA4H modulators and uses thereof申请人:Barchuk William T.公开号:US20080194630A1公开(公告)日:2008-08-14Leukotriene A4 hydrolase (LTA4H) inhibitors, compositions containing them, and methods of use for the inhibition of LTA4H enzyme activity and the treatment, prevention or inhibition of inflammation and/or conditions associated with inflammation.
-
A SET OF GELDANAMYCIN DERIVATIVES AND THEIR PREPARATION METHODS申请人:Institute Of Medicinal Biotechnology, Chinese Academy of Medical Sciences公开号:EP2253621A1公开(公告)日:2010-11-24A set of geldanamycin derivatives and their preparation methods. Pharmaceutical compositions comprising the said compounds as an active ingredient which are used as antivirus and antitumor agents. The said derivatives are used in the manufacture of heat shock protein 90(Hsp 90) inhibiting agents which have the utility as antivirus and antitumor agents.
-
Benzene Sulfonamide Thiazole and Oxazole Compounds申请人:Adams Jerry Leroy公开号:US20090298815A1公开(公告)日:2009-12-03The present invention provides thiazole sulfonamide and oxazole sulfonamide compounds, compositions containing the same, as well as processes for the preparation and methods for their use as pharmaceutical agents.
-
[EN] MACROCYCLIC INDOLE DERIVATIVES<br/>[FR] DÉRIVÉS D'INDOLE MACROCYCLIQUES申请人:BROAD INST INC公开号:WO2019096911A1公开(公告)日:2019-05-23The present invention relates to macrocyclic indole derivatives of general formula (I), in which R1, R2, R3, R4, R5, R6, A and L are as defined herein, methods of preparing said compounds, intermediate compounds useful for preparing said compounds, pharmaceutical compositions and combinations comprising said compounds, and the use of said compounds for manufacturing pharmaceutical compositions for the treatment or prophylaxis of diseases, in particular of hyperproliferative disorders, as a sole agent or in combination with other active ingredients.
表征谱图
-
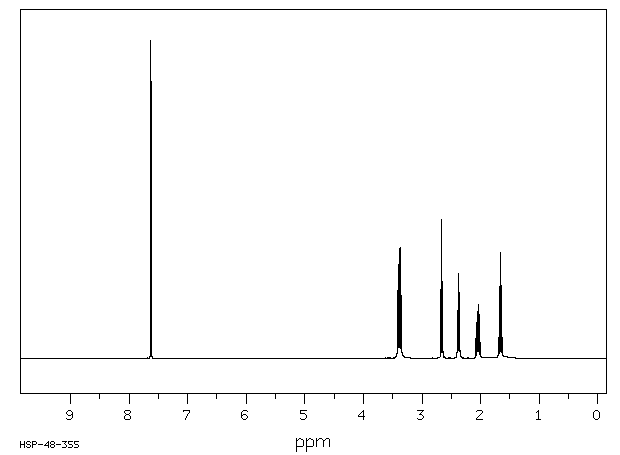
氢谱1HNMR
-
质谱MS
-
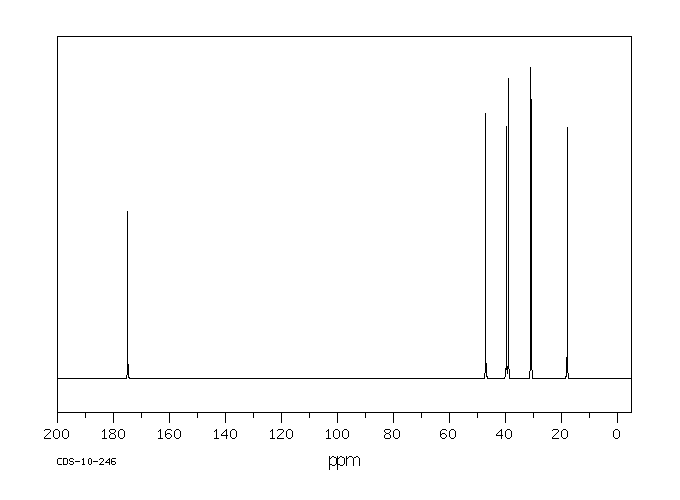
碳谱13CNMR
-
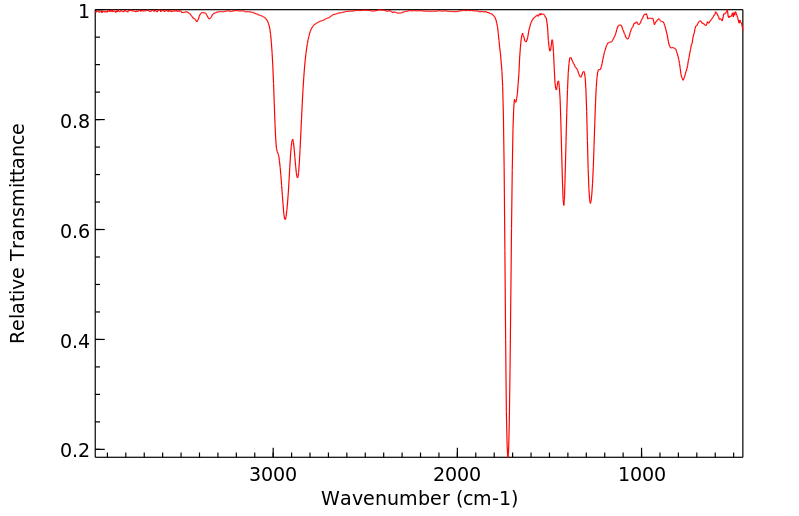
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(5R,Z)-3-(羟基((1R,2S,6S,8aS)-1,3,6-三甲基-2-((E)-prop-1-en-1-yl)-1,2,4a,5,6,7,8,8a-八氢萘-1-基)亚甲基)-5-(羟甲基)-1-甲基吡咯烷-2,4-二酮
(2R,2''R)-(-)-2,2''-联吡咯烷
麦角甾-7,22-二烯-3-基亚油酸酯
马来酰亚胺霉素
马来酰亚胺基酰肼盐酸盐
马来酰亚胺基甲基-3-马来酰亚胺基丙酸酯
马来酰亚胺丙酰基-dPEG4-NHS
马来酰亚胺-酰胺-PEG6-琥珀酰亚胺酯
马来酰亚胺-酰胺-PEG6-丙酸
马来酰亚胺-酰胺-PEG24-丙酸
马来酰亚胺-酰胺-PEG12-丙酸
马来酰亚胺-四聚乙二醇-羧酸
马来酰亚胺-四聚乙二醇-丙酸叔丁酯
马来酰亚胺-四聚乙二醇-丙烯酸琥珀酰亚胺酯
马来酰亚胺-六聚乙二醇-羧酸
马来酰亚胺-六聚乙二醇-丙酸叔丁酯
马来酰亚胺-八聚乙二醇-丙酸叔丁酯
马来酰亚胺-二聚乙二醇-丙酸叔丁酯
马来酰亚胺-三(乙烯乙二醇)-丙酸
马来酰亚胺-一聚乙二醇-羧酸
马来酰亚胺-一聚乙二醇-丙烯酸琥珀酰亚胺酯
马来酰亚胺-PEG3-羟基
马来酰亚胺-PEG2-胺三氟醋酸盐
马来酰亚胺-PEG2-琥珀酰亚胺酯
马来酰亚胺
频哪醇硼酸酯
顺式草酸双(-3,8-二氮杂双环[4.2.0]辛烷-8-羧酸叔丁酯)
顺式4-甲基吡咯烷酮-3-醇盐酸盐
顺式4-氟吡咯烷酮-3-醇盐酸盐
顺式3,4-二羟基吡咯烷盐酸盐
顺式3,4-二氨基吡咯烷-1-羧酸叔丁酯
顺式-二甲基 1-苄基吡咯烷-3,4-二羧酸
顺式-N-[2-(2,6-二甲基-1-哌啶基)乙基]-2-氧代-4-苯基-1-吡咯烷乙酰胺
顺式-N-Boc-吡咯烷-3,4-二羧酸
顺式-5-苄基-2-叔丁氧羰基六氢吡咯并[3,4-c]吡咯
顺式-5-甲基-1H-六氢吡咯并[3,4-b]吡咯二盐酸盐
顺式-5-氧代六氢环戊二烯并[c]吡咯-2(1H)-羧酸叔丁酯
顺式-5-乙氧羰基-1H-六氢吡咯并[3,4-B]吡咯盐酸盐
顺式-5-(碘甲基)-4-苯基-2-吡咯烷酮
顺式-5-(碘甲基)-4-甲基-2-吡咯烷酮
顺式-4-氧代-六氢-吡咯并[3,4-C]吡咯-2-甲酸叔丁酯
顺式-3-氟-4-羟基吡咯烷-1-羧酸叔丁酯
顺式-3-氟-4-甲基吡咯烷盐酸盐
顺式-2-甲基六氢吡咯并[3,4-c]吡咯
顺式-2,5-二甲基吡咯烷
顺式-1-苄基-3,4-吡咯烷二甲酸二乙酯
顺式-1-甲基六氢吡咯并[3,4-b]吡咯
顺式-(9CI)-3,4-二乙烯-1-(三氟乙酰基)-吡咯烷
顺-八氢环戊[c]吡咯-5-酮盐酸盐
非星匹宁