2,3-二氯苯氧基乙酸 | 2976-74-1
中文名称
2,3-二氯苯氧基乙酸
中文别名
2,3-二氯苯氧乙酸;2,3-滴酸
英文名称
2,3-dichlorophenoxyacetic acid
英文别名
2-(2,3-dichlorophenoxy)acetic acid
CAS
2976-74-1
化学式
C8H6Cl2O3
mdl
MFCD00004299
分子量
221.04
InChiKey
RBJIGQRZLITQJG-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:172-175 °C
-
沸点:316.96°C (rough estimate)
-
密度:1.4564 (rough estimate)
-
溶解度:可溶于DMSO(少许)、甲醇(少许)
-
稳定性/保质期:
遵照规定使用和储存,则不会分解。
计算性质
-
辛醇/水分配系数(LogP):2.5
-
重原子数:13
-
可旋转键数:3
-
环数:1.0
-
sp3杂化的碳原子比例:0.125
-
拓扑面积:46.5
-
氢给体数:1
-
氢受体数:3
安全信息
-
危险等级:6.1(b)
-
危险品标志:Xi
-
安全说明:S16,S26,S36/37/39,S37/39,S45
-
危险类别码:R36/37/38
-
海关编码:2918990090
-
危险品运输编号:2811
-
包装等级:III
-
危险类别:6.1(b)
-
WGK Germany:3
-
危险标志:GHS07
-
危险性描述:H302,H315,H319,H335
-
危险性防范说明:P301 + P312 + P330,P305 + P351 + P338
-
储存条件:存储于阴凉干燥处。
SDS
| Name: | 2 3-Dichlorophenoxyacetic Acid Material Safety Data Sheet |
| Synonym: | Non |
| CAS: | 2976-74-1 |
Synonym:Non
Section 2 - COMPOSITION, INFORMATION ON INGREDIENTS
| CAS# | Chemical Name | content | EINECS# |
| 2976-74-1 | 2,3-Dichlorophenoxyacetic Acid | ca 100 | 221-022-9 |
Risk Phrases: 36/37/38
Section 3 - HAZARDS IDENTIFICATION
EMERGENCY OVERVIEW
Irritating to eyes, respiratory system and skin.The toxicological properties of this material have not been fully investigated.
Potential Health Effects
Eye:
Causes eye irritation.
Skin:
Causes skin irritation. The toxicological properties of this material have not been fully investigated.
Ingestion:
Causes gastrointestinal irritation with nausea, vomiting and diarrhea. The toxicological properties of this substance have not been fully investigated.
Inhalation:
Causes respiratory tract irritation. The toxicological properties of this substance have not been fully investigated.
Chronic:
No information found.
Section 4 - FIRST AID MEASURES
Eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid.
Skin:
Get medical aid. Flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes. Wash clothing before reuse.
Ingestion:
Never give anything by mouth to an unconscious person. Get medical aid. Do NOT induce vomiting. If conscious and alert, rinse mouth and drink 2-4 cupfuls of milk or water.
Inhalation:
Remove from exposure and move to fresh air immediately. If not breathing, give artificial respiration. If breathing is difficult, give oxygen. Get medical aid.
Notes to Physician:
Treat symptomatically and supportively.
Section 5 - FIRE FIGHTING MEASURES
General Information:
As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear. During a fire, irritating and highly toxic gases may be generated by thermal decomposition or combustion.
Extinguishing Media:
In case of fire, use water, dry chemical, chemical foam, or alcohol-resistant foam. Use agent most appropriate to extinguish fire. Use water spray, dry chemical, carbon dioxide, or appropriate foam.
Section 6 - ACCIDENTAL RELEASE MEASURES
General Information: Use proper personal protective equipment as indicated in Section 8.
Spills/Leaks:
Clean up spills immediately, observing precautions in the Protective Equipment section. Sweep up, then place into a suitable container for disposal. Avoid generating dusty conditions. Provide ventilation.
Section 7 - HANDLING and STORAGE
Handling:
Wash thoroughly after handling. Remove contaminated clothing and wash before reuse. Avoid contact with eyes, skin, and clothing. Keep container tightly closed. Avoid ingestion and inhalation. Use with adequate ventilation. Wash clothing before reuse.
Storage:
Keep container closed when not in use. Store in a tightly closed container. Store in a cool, dry, well-ventilated area away from incompatible substances.
Section 8 - EXPOSURE CONTROLS, PERSONAL PROTECTION
Engineering Controls:
Facilities storing or utilizing this material should be equipped with an eyewash facility and a safety shower. Use adequate ventilation to keep airborne concentrations low.
Exposure Limits CAS# 2976-74-1: Personal Protective Equipment Eyes: Wear appropriate protective eyeglasses or chemical safety goggles as described by OSHA's eye and face protection regulations in 29 CFR 1910.133 or European Standard EN166.
Skin:
Wear appropriate protective gloves to prevent skin exposure.
Clothing:
Wear appropriate protective clothing to prevent skin exposure.
Respirators:
Follow the OSHA respirator regulations found in 29 CFR 1910.134 or European Standard EN 149. Use a NIOSH/MSHA or European Standard EN 149 approved respirator if exposure limits are exceeded or if irritation or other symptoms are experienced.
Section 9 - PHYSICAL AND CHEMICAL PROPERTIES
Physical State: Powder
Color: white
Odor: None reported.
pH: Not available.
Vapor Pressure: Not available.
Viscosity: Not available.
Boiling Point: Not available.
Freezing/Melting Point: 173.00 - 175.00 deg C
Autoignition Temperature: Not available.
Flash Point: Not available.
Explosion Limits, lower: Not available.
Explosion Limits, upper: Not available.
Decomposition Temperature:
Solubility in water:
Specific Gravity/Density:
Molecular Formula: C8H6Cl2O3
Molecular Weight: 221.04
Section 10 - STABILITY AND REACTIVITY
Chemical Stability:
Stable at room temperature in closed containers under normal storage and handling conditions.
Conditions to Avoid:
Incompatible materials, excess heat, strong oxidants.
Incompatibilities with Other Materials:
Oxidizing agents.
Hazardous Decomposition Products:
Hydrogen chloride, carbon monoxide, irritating and toxic fumes and gases, carbon dioxide.
Hazardous Polymerization: Has not been reported.
Section 11 - TOXICOLOGICAL INFORMATION
RTECS#:
CAS# 2976-74-1 unlisted.
LD50/LC50:
Not available.
Carcinogenicity:
2,3-Dichlorophenoxyacetic Acid - Not listed by ACGIH, IARC, or NTP.
Section 12 - ECOLOGICAL INFORMATION
Section 13 - DISPOSAL CONSIDERATIONS
Dispose of in a manner consistent with federal, state, and local regulations.
Section 14 - TRANSPORT INFORMATION
IATA
Not regulated as a hazardous material.
IMO
Not regulated as a hazardous material.
RID/ADR
Not regulated as a hazardous material.
Section 15 - REGULATORY INFORMATION
European/International Regulations
European Labeling in Accordance with EC Directives
Hazard Symbols: XI
Risk Phrases:
R 36/37/38 Irritating to eyes, respiratory system
and skin.
Safety Phrases:
S 24/25 Avoid contact with skin and eyes.
S 28A After contact with skin, wash immediately with
plenty of water.
S 37 Wear suitable gloves.
S 45 In case of accident or if you feel unwell, seek
medical advice immediately (show the label where
possible).
WGK (Water Danger/Protection)
CAS# 2976-74-1: No information available.
Canada
None of the chemicals in this product are listed on the DSL/NDSL list.
CAS# 2976-74-1 is not listed on Canada's Ingredient Disclosure List.
US FEDERAL
TSCA
CAS# 2976-74-1 is not listed on the TSCA inventory.
It is for research and development use only.
SECTION 16 - ADDITIONAL INFORMATION
N/A
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 一缩二乙二醇一(2-乙基已基)醚 2,3-Dichlorphenoxy-essigsaeure-methylester 1928-56-9 C9H8Cl2O3 235.067 —— (dichloro-2,3 phenoxy)acetate d'ethyle 37536-92-8 C10H10Cl2O3 249.094 (2,3-二氯-苯氧基)-乙酸酰胺 (2,3-dichloro-phenoxy)-acetic acid amide 67526-49-2 C8H7Cl2NO2 220.055 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 —— (2,3-dichlorophenoxy)acetyl chloride 85630-84-8 C8H5Cl3O2 239.485 2-(2,3-二氯苯氧基)-N-(2-羟乙基)乙酰胺 2-(2,3-dichlorophenoxy)-N-(2-hydroxyethyl)acetamide 87762-15-0 C10H11Cl2NO3 264.108
反应信息
-
作为反应物:描述:参考文献:名称:N-(5-(3,5-甲氧基苯基)-(噻唑-2-基))苯氧乙酰胺衍生物的设计、合成和除草活性摘要:噻唑和苯氧乙酸是许多天然和合成生物活性剂中的关键部分。设计合成了一系列 N-(5-(3,5-甲氧基苯基)-(噻唑-2-基))苯氧基乙酰胺衍生物 6an–6bd,并通过 NMR 和 HRMS 证实了它们的结构。通过培养皿生物测定,大多数衍生物对 Echinochloa crusgalli (E.c.) 和 Lactuca sativa (L.s.) 种子萌发表现出优异的抑制作用。事实上,除草剂生物测定表明,6an(2-(2,4-二氯苯氧基)-N-(5-(3,5-二甲氧基苯基)-1,3,4-噻二唑-2-基)乙酰胺)对L.s.的抑制作用最好(IC50 = 42.7 g/ha,田间实验为 375 g/ha)。6an 在 2 至 4 倍的田间使用量对 Zea mays 也没有有害影响。此外,转录组学和代谢组学分析显示,6an 显著影响细胞代谢,包括半乳糖代谢以及抗坏血酸和积石酸盐代谢。这些发现突出表明DOI:10.1021/acs.jafc.4c01824
-
作为产物:描述:二氯-苯酚 在 potassium carbonate 、 lithium hydroxide 作用下, 以 四氢呋喃 、 甲醇 、 水 、 N,N-二甲基甲酰胺 为溶剂, 生成 2,3-二氯苯氧基乙酸参考文献:名称:一系列 N-(1-(1,1-二氧化四氢噻吩-3-基)-3-甲基-1H-吡唑-5-基)乙酰胺醚作为新型 GIRK1/2 钾通道激活剂的发现、合成和生物学表征摘要:本研究描述了一系列N -(1-(1,1-二氧化四氢噻吩-3-基)-3-甲基-1 H-吡唑-5-基)乙酰胺醚作为 G 蛋白内门控的发现和表征-整流钾(GIRK)通道激活剂。从我们之前的先导化合物优化工作中,我们已经确定了一种新的基于醚的支架,并将其与一种新型的基于砜的头基配对,以确定一种有效且选择性的 GIRK1/2 激活剂。此外,我们在第 1 层 DMPK 测定中评估了这些化合物,并确定了作为 GIRK1/2 激活剂显示纳摩尔效力的化合物,与典型的基于尿素的化合物相比,其代谢稳定性得到了改善。DOI:10.1039/d1md00129a
文献信息
-
Camptothecin derivatives申请人:California Pacific Medical Center公开号:US06350756B1公开(公告)日:2002-02-26(20S) esters of camptothecin analogs are provided. The compounds are (20S) esters of an oxyalkanoic acid and camptothecin, which is optionally substituted at the 7, 9, 10, 11, and 12 positions of the camptothecin ring. The compounds are useful for treating cancer.
-
Sulfonation and sulfation in the reactions of the chloro- and dichlorophenols, 3-fluorophenol and (2,3-, 2,4- and 3,4-dichlorophenoxy)acetic acid with concentrated aqueous sulfuric acid and sulfur trioxide作者:Peter de Wit、Hans CerfontainDOI:10.1002/recl.19881070303日期:——The chloro- and dichlorophenols have been sulfonated with sulfuric acid and with SO3 in aprotic solvents. In the sulfonations with concentrated aqueous sulfuric acid, the sulfonic acid isomer distribution is determined mainly by the ortho- and para-directing and activating effect of the hydroxy substituent; this is also the determining factor in the aprotic sulfonations using up to 1.0 equiv of SO3
-
Synthesis and Biological Activity of 1-(Substituted phenoxyacetoxy)-1-(pyridin-2-yl or thien-2-yl)methylphosphonates作者:Tao Wang、Wei Wang、Hao Peng、Hongwu HeDOI:10.1002/jhet.1944日期:2015.1A series of novel O,O‐dimethyl 1‐(substituted phenoxyacetoxy)‐1‐(pyridin‐2‐yl or thien‐2‐yl)methylphosphonates 6a, 6b, 6c, 6d, 6e, 6f, 6g, 6h, 6i, 6j, 6k, 6l, 6m, 6n and 7a, 7b, 7c, 7d were synthesized. Their structures were confirmed by IR, 1H NMR, mass spectroscopy, and elemental analyses. The results of preliminary bioassays show that some of the title compounds exhibit moderate to good herbicidal一系列新颖的O,O-二甲基1-(取代的苯氧基乙酰氧基)-1-(吡啶-2-基或噻吩-2-基)甲基膦酸酯6a,6b,6c,6d,6e,6f,6g,6h,6i,合成了6j,6k,6l,6m,6n和7a,7b,7c,7d。其结构已通过IR确认11 H NMR,质谱和元素分析。初步生物测定的结果表明,某些标题化合物具有中等至良好的除草和杀真菌活性。例如,标题化合物6a,6c,6l,6m和7d在1500 g ai / ha的剂量下对大多数受试植物具有90-100%的抑制作用,而标题化合物6b,6g,6h和6n具有抑制作用针对92-100%抑制尖镰孢,Phyricularia菌,葡萄孢属cinereapers,玉蜀黍赤霉,浓度为50 mg / L的核盘菌菌核病菌(Sclerotinia sclerotiorum)和锥尾核孢菌(Cercospora beticola)。
-
An Efficient One-Pot Synthesis of 2-(Aryloxyacetyl)cyclohexane-1,3-diones as Herbicidal 4-Hydroxyphenylpyruvate Dioxygenase Inhibitors作者:Da-Wei Wang、Hong-Yan Lin、Bo He、Feng-Xu Wu、Tao Chen、Qiong Chen、Wen-Chao Yang、Guang-Fu YangDOI:10.1021/acs.jafc.6b04110日期:2016.11.304-Hydroxyphenylpyruvate dioxygenase (EC 1.13.11.27, HPPD) is an important target for new bleaching herbicides discovery. As a continuous work to discover novel crop selective HPPD inhibitor, a series of 2-(aryloxyacetyl)cyclohexane-1,3-diones were rationally designed and synthesized by an efficient one-pot procedure using N,N′-carbonyldiimidazole (CDI), triethylamine, and acetone cyanohydrin in CH2Cl24-羟基苯基丙酮酸双加氧酶(EC 1.13.11.27,HPPD)是发现新的漂白除草剂的重要目标。为发现新型农作物选择性HPPD抑制剂而进行的持续工作,使用N,N'-羰基二咪唑(CDI),通过高效的一锅法合理设计和合成了一系列2-(芳氧基乙酰基)环己烷-1,3-二酮,三乙胺和丙酮氰醇在CH 2 Cl 2中。总共以良好至优异的产率合成了58种三酮化合物。一些三酮类化合物显示出有效的体外拟南芥HPPD(At HPPD)抑制活性。2-(2-(((1-溴萘-2-基)氧基)乙酰基)-3-羟基环己基-2-烯-1-酮,II-13,在37.5–150 g ai / ha的剂量下表现出高,广谱和芽后除草活性,几乎与甲基磺草酮对某些杂草一样有效。此外,II-13在150 g ai / ha的速率下对玉米和低芥酸菜籽显示出良好的农作物安全性,这表明II-13可能作为除草剂用于控制玉米和低芥酸菜子田中的杂草。II
-
Process for the Preparation of Ethacrynic Acid申请人:Strides Shasun Limited公开号:US20180111890A1公开(公告)日:2018-04-26The invention provides an improved process for preparing Ethacrynic acid of formula I, including the steps of: (a) reacting 4-butyryl-2,3-dichloro-phenoxy acetic acid of formula II with dimethylamine or its salt to obtain [2,3-dichloro-4-[2-dimethylaminomethyl butyryl phenoxy acetic acid of formula III or its salt; (b) hydrolysing [2,3-dichloro-4-[2-dimethylaminomethyl butyryl phenoxy acetic acid hydrochloride of formula III obtained in step a) with t-butyl amine to obtain t-butyl amine salt of Ethacrynic acid; (c) acidifying the t-butyl amine salt of Ethacrynic acid formed in step b) to obtain Ethacrynic acid of formula I; and(d) optionally purifying the obtained Ethacrynic acid with a solvent mixture of alkyl acetate and hydrocarbon solvent. The invention also provides crystalline t-butylamine salt of Ethacrynic acid and process thereof. Also provide compound Ethacrynic acid having a purity of greater than or equal to 99% and a composition including the compound.
表征谱图
-
氢谱1HNMR
-
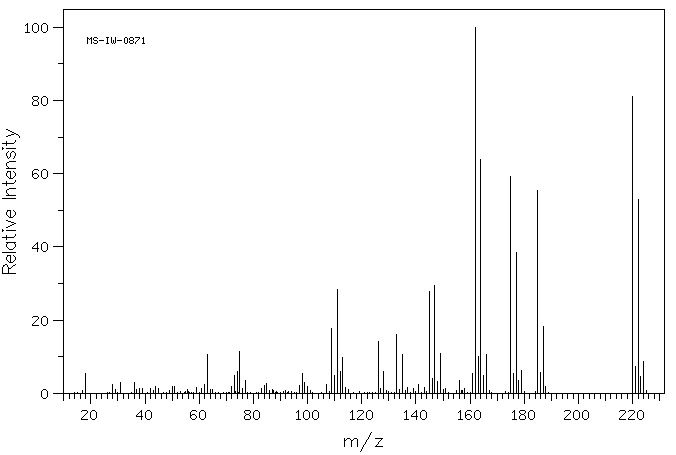
质谱MS
-
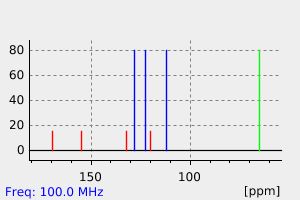
碳谱13CNMR
-
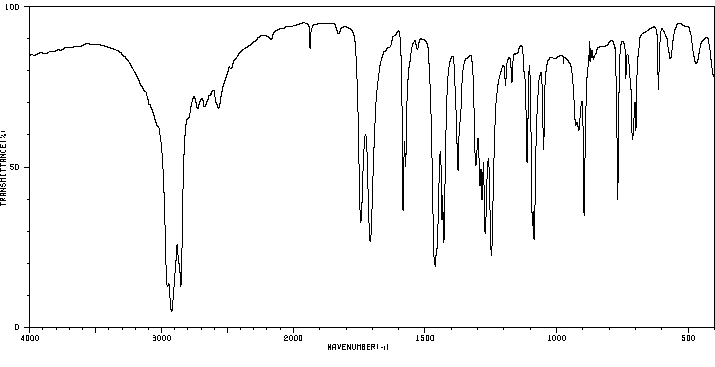
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(βS)-β-氨基-4-(4-羟基苯氧基)-3,5-二碘苯甲丙醇
(S,S)-邻甲苯基-DIPAMP
(S)-(-)-7'-〔4(S)-(苄基)恶唑-2-基]-7-二(3,5-二-叔丁基苯基)膦基-2,2',3,3'-四氢-1,1-螺二氢茚
(S)-盐酸沙丁胺醇
(S)-3-(叔丁基)-4-(2,6-二甲氧基苯基)-2,3-二氢苯并[d][1,3]氧磷杂环戊二烯
(S)-2,2'-双[双(3,5-三氟甲基苯基)膦基]-4,4',6,6'-四甲氧基联苯
(S)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(R)富马酸托特罗定
(R)-(-)-盐酸尼古地平
(R)-(-)-4,12-双(二苯基膦基)[2.2]对环芳烷(1,5环辛二烯)铑(I)四氟硼酸盐
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[((6-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(4-叔丁基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(3-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-4,7-双(3,5-二-叔丁基苯基)膦基-7“-[(吡啶-2-基甲基)氨基]-2,2”,3,3'-四氢1,1'-螺二茚满
(R)-3-(叔丁基)-4-(2,6-二苯氧基苯基)-2,3-二氢苯并[d][1,3]氧杂磷杂环戊烯
(R)-2-[((二苯基膦基)甲基]吡咯烷
(R)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(N-(4-甲氧基苯基)-N-甲基-3-(1-哌啶基)丙-2-烯酰胺)
(5-溴-2-羟基苯基)-4-氯苯甲酮
(5-溴-2-氯苯基)(4-羟基苯基)甲酮
(5-氧代-3-苯基-2,5-二氢-1,2,3,4-oxatriazol-3-鎓)
(4S,5R)-4-甲基-5-苯基-1,2,3-氧代噻唑烷-2,2-二氧化物-3-羧酸叔丁酯
(4S,4''S)-2,2''-亚环戊基双[4,5-二氢-4-(苯甲基)恶唑]
(4-溴苯基)-[2-氟-4-[6-[甲基(丙-2-烯基)氨基]己氧基]苯基]甲酮
(4-丁氧基苯甲基)三苯基溴化磷
(3aR,8aR)-(-)-4,4,8,8-四(3,5-二甲基苯基)四氢-2,2-二甲基-6-苯基-1,3-二氧戊环[4,5-e]二恶唑磷
(3aR,6aS)-5-氧代六氢环戊基[c]吡咯-2(1H)-羧酸酯
(2Z)-3-[[(4-氯苯基)氨基]-2-氰基丙烯酸乙酯
(2S,3S,5S)-5-(叔丁氧基甲酰氨基)-2-(N-5-噻唑基-甲氧羰基)氨基-1,6-二苯基-3-羟基己烷
(2S,2''S,3S,3''S)-3,3''-二叔丁基-4,4''-双(2,6-二甲氧基苯基)-2,2'',3,3''-四氢-2,2''-联苯并[d][1,3]氧杂磷杂戊环
(2S)-(-)-2-{[[[[3,5-双(氟代甲基)苯基]氨基]硫代甲基]氨基}-N-(二苯基甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[((1S,2S)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[[((1R,2R)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2-硝基苯基)磷酸三酰胺
(2,6-二氯苯基)乙酰氯
(2,3-二甲氧基-5-甲基苯基)硼酸
(1S,2S,3S,5S)-5-叠氮基-3-(苯基甲氧基)-2-[(苯基甲氧基)甲基]环戊醇
(1S,2S,3R,5R)-2-(苄氧基)甲基-6-氧杂双环[3.1.0]己-3-醇
(1-(4-氟苯基)环丙基)甲胺盐酸盐
(1-(3-溴苯基)环丁基)甲胺盐酸盐
(1-(2-氯苯基)环丁基)甲胺盐酸盐
(1-(2-氟苯基)环丙基)甲胺盐酸盐
(1-(2,6-二氟苯基)环丙基)甲胺盐酸盐
(-)-去甲基西布曲明
龙蒿油
龙胆酸钠
龙胆酸叔丁酯
龙胆酸
龙胆紫-d6
龙胆紫