2,5-二甲基噻唑 | 4175-66-0
中文名称
2,5-二甲基噻唑
中文别名
——
英文名称
2,5-dimethyl-1,3-thiazole
英文别名
2,5-Dimethyl-thiazol;2,5-dimethylthiazole
CAS
4175-66-0
化学式
C5H7NS
mdl
——
分子量
113.183
InChiKey
WVUHHPQQQLBMOE-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
沸点:152°C (estimate)
-
密度:1.075 (estimate)
-
LogP:1.56
-
物理描述:Yellow liquid; Cooked, brown and roasted nutty aroma
-
溶解度:Soluble in non-polar solvents; slightly soluble in water
-
折光率:1.523-1.527
-
保留指数:922;887;922;868;899
计算性质
-
辛醇/水分配系数(LogP):1.8
-
重原子数:7
-
可旋转键数:0
-
环数:1.0
-
sp3杂化的碳原子比例:0.4
-
拓扑面积:41.1
-
氢给体数:0
-
氢受体数:2
安全信息
-
海关编码:2934100090
-
危险性防范说明:P261,P305+P351+P338
-
危险性描述:H302,H315,H319,H335
SDS
Material Safety Data Sheet
Section 1. Identification of the substance
Product Name: 2,5-Dimethylthiazole
Synonyms:
Section 2. Hazards identification
Harmful by inhalation, in contact with skin, and if swallowed.
Section 3. Composition/information on ingredients.
Ingredient name: 2,5-Dimethylthiazole
CAS number: 4175-66-0
Section 4. First aid measures
Skin contact: Immediately wash skin with copious amounts of water for at least 15 minutes while removing
contaminated clothing and shoes. If irritation persists, seek medical attention.
Eye contact: Immediately wash skin with copious amounts of water for at least 15 minutes. Assure adequate
flushing of the eyes by separating the eyelids with fingers. If irritation persists, seek medical
attention.
Inhalation: Remove to fresh air. In severe cases or if symptoms persist, seek medical attention.
Ingestion: Wash out mouth with copious amounts of water for at least 15 minutes. Seek medical attention.
Section 5. Fire fighting measures
In the event of a fire involving this material, alone or in combination with other materials, use dry
powder or carbon dioxide extinguishers. Protective clothing and self-contained breathing apparatus
should be worn.
Section 6. Accidental release measures
Personal precautions: Wear suitable personal protective equipment which performs satisfactorily and meets local/state/national
standards.
Respiratory precaution: Wear approved mask/respirator
Hand precaution: Wear suitable gloves/gauntlets
Skin protection: Wear suitable protective clothing
Eye protection: Wear suitable eye protection
Methods for cleaning up: Mix with sand or similar inert absorbent material, sweep up and keep in a tightly closed container
for disposal. See section 12.
Environmental precautions: Do not allow material to enter drains or water courses.
Section 7. Handling and storage
Handling: This product should be handled only by, or under the close supervision of, those properly qualified
in the handling and use of potentially hazardous chemicals, who should take into account the fire,
health and chemical hazard data given on this sheet.
Store in closed vessels, refrigerated.
Storage:
Section 8. Exposure Controls / Personal protection
Engineering Controls: Use only in a chemical fume hood.
Personal protective equipment: Wear laboratory clothing, chemical-resistant gloves and safety goggles.
General hydiene measures: Wash thoroughly after handling. Wash contaminated clothing before reuse.
Section 9. Physical and chemical properties
Appearance: Not specified
Boiling point: No data
No data
Melting point:
Flash point: No data
Density: No data
Molecular formula: C5H7NS
Molecular weight: 113.2
Section 10. Stability and reactivity
Conditions to avoid: Heat, flames and sparks.
Materials to avoid: Oxidizing agents.
Possible hazardous combustion products: Carbon monoxide, nitrogen oxides, sulfur oxides.
Section 11. Toxicological information
No data.
Section 12. Ecological information
No data.
Section 13. Disposal consideration
Arrange disposal as special waste, by licensed disposal company, in consultation with local waste
disposal authority, in accordance with national and regional regulations.
Section 14. Transportation information
Non-harzardous for air and ground transportation.
Section 15. Regulatory information
No chemicals in this material are subject to the reporting requirements of SARA Title III, Section
302, or have known CAS numbers that exceed the threshold reporting levels established by SARA
Title III, Section 313.
SECTION 16 - ADDITIONAL INFORMATION
N/A
Section 1. Identification of the substance
Product Name: 2,5-Dimethylthiazole
Synonyms:
Section 2. Hazards identification
Harmful by inhalation, in contact with skin, and if swallowed.
Section 3. Composition/information on ingredients.
Ingredient name: 2,5-Dimethylthiazole
CAS number: 4175-66-0
Section 4. First aid measures
Skin contact: Immediately wash skin with copious amounts of water for at least 15 minutes while removing
contaminated clothing and shoes. If irritation persists, seek medical attention.
Eye contact: Immediately wash skin with copious amounts of water for at least 15 minutes. Assure adequate
flushing of the eyes by separating the eyelids with fingers. If irritation persists, seek medical
attention.
Inhalation: Remove to fresh air. In severe cases or if symptoms persist, seek medical attention.
Ingestion: Wash out mouth with copious amounts of water for at least 15 minutes. Seek medical attention.
Section 5. Fire fighting measures
In the event of a fire involving this material, alone or in combination with other materials, use dry
powder or carbon dioxide extinguishers. Protective clothing and self-contained breathing apparatus
should be worn.
Section 6. Accidental release measures
Personal precautions: Wear suitable personal protective equipment which performs satisfactorily and meets local/state/national
standards.
Respiratory precaution: Wear approved mask/respirator
Hand precaution: Wear suitable gloves/gauntlets
Skin protection: Wear suitable protective clothing
Eye protection: Wear suitable eye protection
Methods for cleaning up: Mix with sand or similar inert absorbent material, sweep up and keep in a tightly closed container
for disposal. See section 12.
Environmental precautions: Do not allow material to enter drains or water courses.
Section 7. Handling and storage
Handling: This product should be handled only by, or under the close supervision of, those properly qualified
in the handling and use of potentially hazardous chemicals, who should take into account the fire,
health and chemical hazard data given on this sheet.
Store in closed vessels, refrigerated.
Storage:
Section 8. Exposure Controls / Personal protection
Engineering Controls: Use only in a chemical fume hood.
Personal protective equipment: Wear laboratory clothing, chemical-resistant gloves and safety goggles.
General hydiene measures: Wash thoroughly after handling. Wash contaminated clothing before reuse.
Section 9. Physical and chemical properties
Appearance: Not specified
Boiling point: No data
No data
Melting point:
Flash point: No data
Density: No data
Molecular formula: C5H7NS
Molecular weight: 113.2
Section 10. Stability and reactivity
Conditions to avoid: Heat, flames and sparks.
Materials to avoid: Oxidizing agents.
Possible hazardous combustion products: Carbon monoxide, nitrogen oxides, sulfur oxides.
Section 11. Toxicological information
No data.
Section 12. Ecological information
No data.
Section 13. Disposal consideration
Arrange disposal as special waste, by licensed disposal company, in consultation with local waste
disposal authority, in accordance with national and regional regulations.
Section 14. Transportation information
Non-harzardous for air and ground transportation.
Section 15. Regulatory information
No chemicals in this material are subject to the reporting requirements of SARA Title III, Section
302, or have known CAS numbers that exceed the threshold reporting levels established by SARA
Title III, Section 313.
SECTION 16 - ADDITIONAL INFORMATION
N/A
制备方法与用途
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 2-甲基噻唑 2-methylthiazole 3581-87-1 C4H5NS 99.1564 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 —— 2-(bromomethyl)-5-methylthiazole 221020-96-8 C5H6BrNS 192.079 5-(溴甲基)-2-甲基噻唑 5-(bromomethyl)-2-methylthiazole 838892-95-8 C5H6BrNS 192.079
反应信息
-
作为反应物:描述:2,5-二甲基噻唑 在 盐酸 、 2,4,6-三甲基苯磺酰羟胺 、 sodium hydroxide 、 sodium nitrite 作用下, 以 二氯甲烷 、 水 为溶剂, 反应 10.75h, 生成 2,6-dimethylpyrazolo[5,1-b][1,3]thiazole-7-amine参考文献:名称:PYRAZOLOTHIAZOLE COMPOUND摘要:化合物的分子式为(I),或其药理学上可接受的盐,表现出优异的CRF受体拮抗作用,其中X是氮原子或CH;R1是-A11-A12;A11是一个单键或C1-6烷基链;A12是氢原子、C1-6烷基或C3-6环烷基等;R2是-A21-A22;A21是一个单键或C1-6烷基链;A22是氢原子、C1-6烷基、C3-6环烷基、非芳香杂环基或杂芳基等;R3是C1-6烷基、C3-6环烷基、C1-6烷氧基、C3-6环烷氧基C1-6烷基、二C1-6烷基氨基、卤素原子、氰基、甲酰基或羧基等;R4是氢原子或C1-6烷氧基;R5是卤素原子、C1-6烷基或C1-6烷氧基;R6是氢原子、C1-6烷基、C1-6烷氧基、C1-6烷基硫基或C1-6烷基亚砜基等;R7是C1-6烷基、C1-6烷氧基或C1-6烷基硫基。公开号:US20110086882A1
-
作为产物:描述:参考文献:名称:Jur'ew; Shukowa, Zhurnal Obshchei Khimii, 1958, vol. 28, p. 7,10; engl. Ausg. S. 5, 7摘要:DOI:
文献信息
-
A Carbonylation Approach Toward Activation of C<sub>sp2</sub>-H and C<sub>sp3</sub>-H Bonds: Cu-Catalyzed Regioselective Cross Coupling of Imidazo[1,2-<i>a</i>]pyridines with Methyl Hetarenes作者:Sai Lei、Yingying Mai、Caijuan Yan、Jianwen Mao、Hua CaoDOI:10.1021/acs.orglett.6b01588日期:2016.8.5An efficient copper-catalyzed selective cross coupling of imidazo[1,2-a]pyridines with methyl hetarenes has been reported. This transformation opened a new route to synthesize the C-3 carbonyl imidazo[1,2-a]pyridine derivative, which is a common structural motif in natural products and pharmaceuticals. 18O-labeling experiments indicated that the oxygen source of products originated from O2.
-
[EN] INDANE DERIVATES AS MUSCARINIC RECEPTOR AGONISTS<br/>[FR] DERIVES D'INDANE UTILISES COMME AGONISTES DU RECEPTEUR MUSCARINIQUE申请人:LILLY CO ELI公开号:WO2005009941A1公开(公告)日:2005-02-03The present invention relates to compounds of Formula I: I which are agonists of the M-1 muscarinic receptor.这项发明涉及到Formula I的化合物,这些化合物是M-1肌氨酸受体的激动剂。
-
Über die Kondensationsfähigkeit der 2-ständigen Methylgruppe in Thiazolverbindungen作者:H. Erlenmeyer、O. Weber、P. Schmidt、G. Küng、Chr. Zinsstag、B. PrijsDOI:10.1002/hlca.19480310421日期:——1. Die Kondensationsfähigkeit von 2-Methylthiazolverbindungen gegenüber Benzaldehyd wurde an zahlreichen Beispielen, so auch am 2-Methylthiazol, nachgeprüft und die Struktur der so erhaltenen 2-Styrylthiazolverbindungen durch direkte Synthese aus Zimtsäurethioamid und Halogencarbonylverbindungen belegt.
-
Discovery of new chemical entities as potential leads against Mycobacterium tuberculosis作者:Xiaoyun Lu、Jian Tang、Zhiyong Liu、Minke Li、Tianyu Zhang、Xiantao Zhang、Ke DingDOI:10.1016/j.bmcl.2016.11.003日期:2016.12A series of biheterocyclic (1H-indole, benzofuran, pyrazolo[1,5-a]pyrimidine, pyrazolo[1,5-a]pyrimidin-5(4H)-one, imidazo[2,1-b]thiazole and pyrazolo[5,1-b]thiazole) derivatives were synthesized and evaluated for their anti-tubercular activities. The imidazo[2,1-b]thiazoles 9a-c and pyrazolo[5,1-b]thiazoles 10a-c exhibited promising anti-tubercular activity in varying degrees. Especially, the 2,6-dimethylpyrazolo[5一系列双杂环(1H-吲哚,苯并呋喃,吡唑并[1,5-a]嘧啶,吡唑并[1,5-a]嘧啶-5(4H)-one,咪唑并[2,1-b]噻唑和吡唑并[合成了5,1-b]噻唑)衍生物并评估了它们的抗结核活性。咪唑并[2,1-b]噻唑9a-c和吡唑并[5,1-b]噻唑10a-c在不同程度上表现出有希望的抗结核活性。特别地,2,6-二甲基吡唑并[5,1-b]噻唑10a具有对H37Ra菌株的强抑制作用,MIC值为0.03μg/ mL。化合物10a还显示出良好的药代动力学特征,口服生物利用度(F)为41.7%,半衰期为13.4h。此外,10a显着降低了自发光H37Ra感染的小鼠模型中的细菌负担,表明其开发抗结核药物的潜力很大。
-
Quaternary Salt Formation of Substituted Oxazoles and Thiazoles<sup>1</sup>作者:Vernon N. Kerr、Donald G. Ott、F. Newton HayesDOI:10.1021/ja01486a042日期:1960.1The course of quaternization of a number of oxazoles and thiazoles and various dimethylaminophenyl, pyridyl, and quinolyl derivatives was investigated. The salts were screened for hypotensive activity. (auth)
表征谱图
-
氢谱1HNMR
-
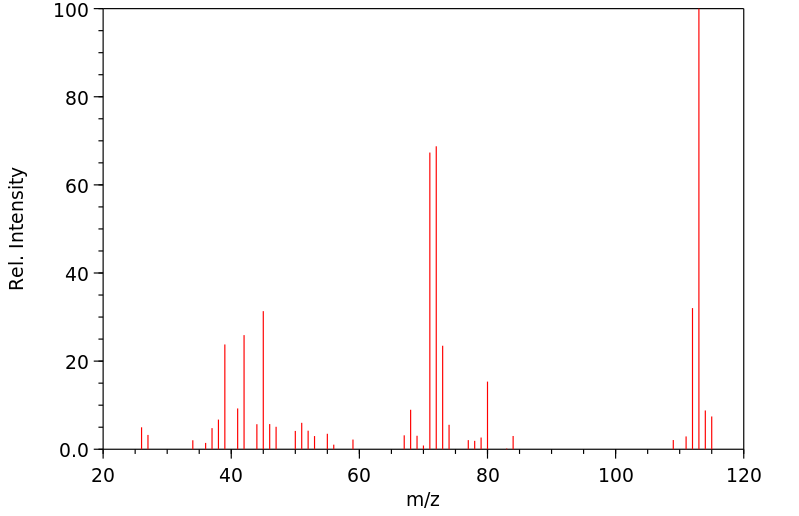
质谱MS
-
碳谱13CNMR
-
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(SP-4-1)-二氯双(1-苯基-1H-咪唑-κN3)-钯
(5aS,6R,9S,9aR)-5a,6,7,8,9,9a-六氢-6,11,11-三甲基-2-(2,3,4,5,6-五氟苯基)-6,9-甲基-4H-[1,2,4]三唑[3,4-c][1,4]苯并恶嗪四氟硼酸酯
(5-氨基-1,3,4-噻二唑-2-基)甲醇
齐墩果-2,12-二烯[2,3-d]异恶唑-28-酸
黄曲霉毒素H1
高效液相卡套柱
非昔硝唑
非布索坦杂质Z19
非布索坦杂质T
非布索坦杂质K
非布索坦杂质E
非布索坦杂质D
非布索坦杂质67
非布索坦杂质65
非布索坦杂质64
非布索坦杂质61
非布索坦代谢物67M-4
非布索坦代谢物67M-2
非布索坦代谢物 67M-1
非布索坦-D9
非布索坦
非唑拉明
雷非那酮-d7
雷西那德杂质2
雷西纳德杂质L
雷西纳德杂质H
雷西纳德杂质B
雷西纳德
雷西奈德杂质
阿西司特
阿莫奈韦
阿考替胺杂质9
阿米苯唑
阿米特罗13C2,15N2
阿瑞匹坦杂质
阿格列扎
阿扎司特
阿尔吡登
阿塔鲁伦中间体
阿培利司N-1
阿哌沙班杂质26
阿哌沙班杂质15
阿可替尼
阿作莫兰
阿佐塞米
镁(2+)(Z)-4'-羟基-3'-甲氧基肉桂酸酯
锌1,2-二甲基咪唑二氯化物
锌(II)(苯甲醇)(四苯基卟啉)
锌(II)(正丁醇)(四苯基卟啉)
锌(II)(异丁醇)(四苯基卟啉)