ethyl (2S,3R)-2,3-epoxybutanoate | 58917-48-9
中文名称
——
中文别名
——
英文名称
ethyl (2S,3R)-2,3-epoxybutanoate
英文别名
erthyl (2S,3R)-3-methyloxirane-2-carboxylate;Ethyl trans-3-methyl-2-oxiranecarboxylate;ethyl (2S,3R)-3-methyloxirane-2-carboxylate
CAS
58917-48-9
化学式
C6H10O3
mdl
——
分子量
130.144
InChiKey
VYXHEFOZRVPJRK-UHNVWZDZSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
计算性质
-
辛醇/水分配系数(LogP):0.6
-
重原子数:9
-
可旋转键数:3
-
环数:1.0
-
sp3杂化的碳原子比例:0.83
-
拓扑面积:38.8
-
氢给体数:0
-
氢受体数:3
反应信息
-
作为反应物:描述:参考文献:名称:研究天然产物吲哚霉素和合成改进的抗分枝杆菌药物类似物摘要:吲哚霉素 (IND) 是一种微生物天然产物,可选择性抑制细菌色氨酸-tRNA 合成酶 (TrpRS)。色氨酸生物合成途径最近被证明是开发针对结核分枝杆菌(Mtb) 的新型抗菌剂的重要目标。我们研究了 IND 对几种分枝杆菌模型菌株的抗菌活性。开发了一种 TrpRS 生化试验来分析合成 IND 类似物的文库。4″-甲基化 IND 化合物 Y-13 显示出改善的抗 Mtb 活性,最小抑制浓度 (MIC) 为 1.88 μM (~0.5 μg/mL)。当在基因工程替代牛分枝杆菌中诱导 TrpRS 过表达时,MIC 显着增加卡介苗。Mtb TrpRS 与 IND 和 ATP 复合的共晶结构表明氨基酸袋处于 apo 蛋白的开放形式和与反应中间体的封闭复合物之间的状态。在基于全细胞的实验中,我们研究了 Y-13 与不同抗菌剂的组合效果。我们通过基因组测序评估了杀伤动力学、对 IND 的抗性频率以及 INDDOI:10.1021/acschembio.1c00394
-
作为产物:描述:丁酸,2-氯-3-羟基-,乙基酯,(2R,3R)- 在 sodium ethanolate 作用下, 以 乙醇 为溶剂, 反应 0.5h, 以65%的产率得到ethyl (2S,3R)-2,3-epoxybutanoate参考文献:名称:A new enantioselective synthesis of glycidates via dynamic kinetic resolution of racemic 2-chloro-3-keto esters using chiral Ru (II) complexes摘要:2-chloro-3-keto esters were quantitatively hydrogenated to syn and anti 2-chloro-3-hydroxyester by asymmetric hydrogenation with chiral ruthenium (Ii) catalysts prepared in-situ from (COD)Ru(2-Methylallyl)(2) in presence of atropisomeric ligands such as MeO-Biphep and Binap, giving enantioselectivity up to 99%. 2-chloro-3-hydroxy esters were treated with different bases to give (E)- and (Z)-2,3-epoxyalkanoates in 65-90% yields with 84-97% ee.DOI:10.1016/0040-4039(95)00182-c
文献信息
-
Development of a Stereoselective Practical Synthetic Route to Indolmycin, a Candidate Anti-H. pylori Agent.作者:Atsushi HASUOKA、Yutaka NAKAYAMA、Mari ADACHI、Hidenori KAMIGUCHI、Keiji KAMIYAMADOI:10.1248/cpb.49.1604日期:——A stereoselective practical synthetic route to indolmycin is described. The route is composed of the regioselective coupling of indolyl magnesium halide with a trans-epoxy ester, diastereoselective oxazolone ring formation with guanidine and amine exchange reaction with methylamine. In the coupling step, use of dichloromethane as co-solvent and conversion of the resulting hydroxy ester to the hydroxy
-
Process for producing indolmycins申请人:Takeda Chemical Industries, Ltd.公开号:US06265586B1公开(公告)日:2001-07-24A process for producing indolmycin or a salt thereof which comprises reacting a compound of the formula: or a salt thereof with methylamine or a salt thereof, can be produced in an optically active form in a high yield and high quality, and is advantageous from the industrial point of view.一种生产吲哚霉素或其盐的方法,包括将式的化合物或其盐与甲基胺或其盐反应,可以高产高质地制备出其光学活性形式,从工业角度来看是有优势的。
-
Reaction of caesium 4-chlorophenate and chlorohydrins from threonines: synthesis of clofibrate analogues作者:Maria Grazia Perrone、Ernesto Santandrea、Leonardo Di Nunno、Antonio Scilimati、Vincenzo Tortorella、Francesco Capitelli、Valerio BertolasiDOI:10.1016/j.tetasy.2005.01.006日期:2005.2Clofibrate is a well-known peroxisome prolifierator-activated receptor-alpha (PPARalpha) agonist, used in the treatment of hyperlipaemias and atherosclerosis and to prevent heart failure. Herein, the preparation of the four enantiomerically pure stereoisomers of ethyl 2-(4-chlorophenoxy)-3-hydroxybutanoate as clofibrate analogues is described. Biological evaluation of these new compounds was performed by a transactivation assay in a transiently transfected monkey kidney fibroblast cell line. All four diastereomers were inactive even at 300 muM, where clofibrate showed an evident activity, suggesting that the designed clofibrate molecular structural modifications in the analogues caused the loss of peroxisome proliferator-activated receptor-alpha (PPARalpha) activity. (C) 2005 Elsevier Ltd. All rights reserved.
表征谱图
-
氢谱1HNMR
-
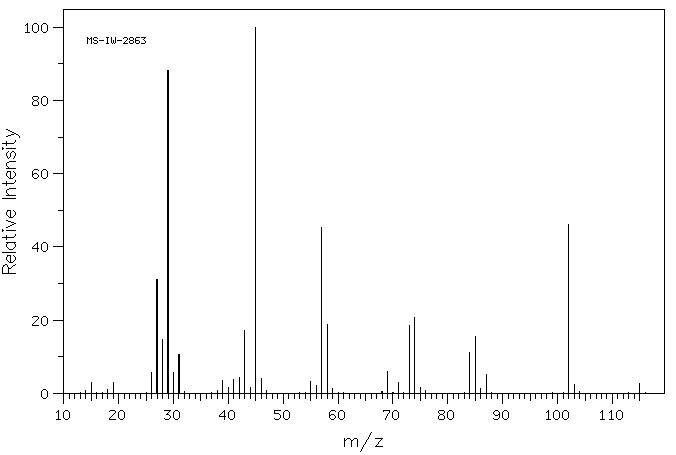
质谱MS
-
碳谱13CNMR
-
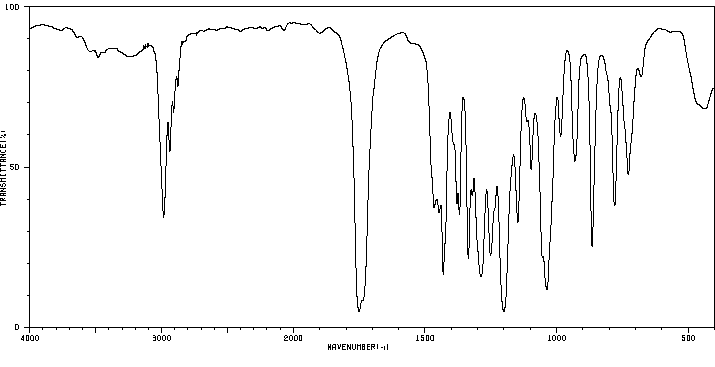
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(S)-4-氯-1,2-环氧丁烷
顺式-环氧琥珀酸氢钾
顺式-1-环己基-2-乙烯基环氧乙烷
顺-(2S,3S)甲基环氧肉桂酸酯
雌舞毒蛾引诱剂
阿洛司他丁
辛基缩水甘油醚
试剂(3S,6S)-(-)-3,6-Diisopropyl-1,4-dioxane-2,5-dione
表氰醇
螺[环氧乙烷-2,2-三环[3.3.1.1~3,7~]癸烷]
蛇根混合碱
benzene oxide
聚碳酸丙烯酯
聚依他丁
羟基乙醛
缩水甘油基异丁基醚
缩水甘油基十六烷基醚
缩水甘油
硬脂基醇聚氧乙烯聚氧丙烯醚
硅烷,三甲基[(3-甲基噁丙环基)乙炔基]-,顺-
盐酸司维拉姆
甲醛与(氯甲基)环氧乙烷,4,4-(1-甲基乙亚基)双酚和2-甲基苯酚的聚合物
甲醛与(氯甲基)环氧乙烷,4,4'-(1-甲基乙亚基)二[苯酚]和4-(1,1,3,3-四甲基丁基)苯酚的聚合物
甲醇环氧乙烷与壬基酚的聚合物
甲胺聚合物与(氯甲基)环氧乙烷
甲硫代环氧丙烷
甲基环氧氯丙烷
甲基环氧巴豆酸酯
甲基环氧乙烷与环氧乙烷和十六烷基或十八烷基醚的聚合物
甲基环氧乙烷与[(2-丙烯基氧基)甲基]环氧乙烷聚合物
甲基环氧丙醇
甲基环氧丙烷
甲基N-丁-3-烯酰甘氨酸酸酯
甲基7-氧杂双环[4.1.0]庚-2,4-二烯-1-羧酸酯
甲基3-环丙基-2-环氧乙烷羧酸酯
甲基1-氧杂螺[2.5]辛烷-2-羧酸酯
甲基(2S,3R)-3-丙基-2-环氧乙烷羧酸酯
甲基(2R,3S)-3-丙基-2-环氧乙烷羧酸酯
甲基(2R,3R)-3-环丙基-2-环氧乙烷羧酸酯
环氧溴丙烷
环氧氯丙烷与双酚A、4-(1,1-二甲乙基)苯酚的聚合物
环氧氯丙烷-d5
环氧氯丙烷-D1
环氧氯丙烷-3,3’-亚氨基二丙胺的聚合物
环氧氯丙烷-2-13C
环氧氯丙烷
环氧氟丙烷
环氧树脂(环氧氯丙烷和二乙二醇)
环氧树脂
环氧柏木烷