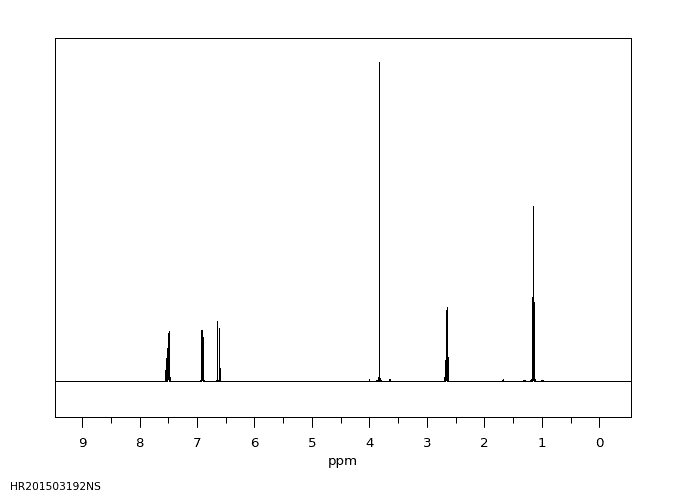(E)-1-(4-methoxyphenyl)pent-1-en-3-one | 82297-64-1
中文名称
——
中文别名
——
英文名称
(E)-1-(4-methoxyphenyl)pent-1-en-3-one
英文别名
ethyl p-methoxystyryl ketone;1-(4-Methoxyphenyl)pent-1-en-3-one
CAS
82297-64-1
化学式
C12H14O2
mdl
——
分子量
190.242
InChiKey
SLDQOBRACOQXGE-QPJJXVBHSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:86.5-87.0 °C
-
沸点:333.2±17.0 °C(Predicted)
-
密度:1.030±0.06 g/cm3(Predicted)
-
物理描述:Solid
-
溶解度:insoluble in water, soluble in organic solvents, oils
计算性质
-
辛醇/水分配系数(LogP):2.5
-
重原子数:14
-
可旋转键数:4
-
环数:1.0
-
sp3杂化的碳原子比例:0.25
-
拓扑面积:26.3
-
氢给体数:0
-
氢受体数:2
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 4-甲氧基肉桂醛 4-Methoxycinnamaldehyde 24680-50-0 C10H10O2 162.188 —— (E)-1-(4-methoxyphenyl)penta-1,4-dien-3-ol —— C12H14O2 190.242 1-(2,2-二溴乙烯基)-4-甲氧基苯 1-(2,2-dibromovinyl)-4-methoxybenzene 60512-57-4 C9H8Br2O 291.97
反应信息
-
作为反应物:描述:(E)-1-(4-methoxyphenyl)pent-1-en-3-one 在 三乙烯二胺 、 bis(1,5-cyclooctadiene)nickel (0) 、 (S)-4-(tert-butyl)-2-(pyrimidin-2-yl)-4,5-dihydrooxazole 、 频那醇硼烷 作用下, 以 甲苯 为溶剂, 反应 0.67h, 以80%的产率得到(S,E)-1-(4-methoxyphenyl)pent-1-en-3-ol参考文献:名称:钴催化烯丙醇与乙烯基三氟甲磺酸酯的对映体特异性动态动力学交叉电乙烯基化摘要:不对称的交叉亲电偶联已成为生产手性分子的有前途的工具。然而,这种化学与镍以外的金属的潜力仍然未知。在此,我们报道了钴催化的烯丙醇与乙烯基三氟甲磺酸酯的对映体特异性乙烯基化反应。这项工作建立了一种合成对映体富集的 1,4-二烯的新方法。该反应通过动态动力学耦合方法进行,这不仅允许烯丙醇直接官能化,而且对于实现高化学选择性也是必不可少的。钴的使用使反应能够以高对映体特异性进行,这是镍催化剂无法实现的。DOI:10.1021/jacs.1c08695
-
作为产物:描述:4-甲氧基肉桂醛 在 [Ru(η(3):η(3)-C10H16)Cl(O2CCH3)] 、 sodium chloride 作用下, 以 四氢呋喃 、 aq. phosphate buffer 为溶剂, 反应 2.0h, 生成 (E)-1-(4-methoxyphenyl)pent-1-en-3-one参考文献:名称:活细胞内钌催化的氧化还原异构化摘要:定制的钌(IV)络合物可以在生理相关条件下甚至在活的哺乳动物细胞内催化烯丙醇异构化为饱和羰基衍生物。该反应涉及氢化钌中间体,具有生物正交性和生物相容性,可用于“细胞内”生成荧光和生物活性探针。总的来说,我们的研究揭示了一种新型的基于金属的细胞干预工具,并进一步证明了有机金属机制与细胞复杂环境的兼容性。DOI:10.1021/jacs.9b00837
文献信息
-
Copper catalyzed oxygen assisted C(CNOH)–C(alkyl) bond cleavage: a facile conversion of aryl/aralkyl/vinyl ketones to aromatic acids作者:Pochampalli Sathyanarayana、Owk Ravi、Prathap Reddy Muktapuram、Surendar Reddy BathulaDOI:10.1039/c5ob01569c日期:——A novel copper-catalyzed aerobic oxidative C(NOH)–C(alkyl) bond cleavage reaction of aryl/aralkyl/vinyl ketones for the synthesis of aromatic/acrylic acids is described. A series of ketones having aryl/aralkyl/vinyl at the one end and methyl to any higher alkyl at the other end can be selectively cleaved and converted into the corresponding acids via oxime intermediates.
-
Regioselective Heck Vinylation of Electron-Rich Olefins with Vinyl Halides: Is the Neutral Pathway in Operation?作者:Matthew McConville、Ourida Saidi、John Blacker、Jianliang XiaoDOI:10.1021/jo802781m日期:2009.4.3Highly regioselective vinylation of electron-rich olefins by bromo- as well as chlorostyrenes is effected by palladium catalysis with either mono- or bidentate phosphines in a molecular solvent, with no need for halide scavengers, ionic liquids, or ionic additives. The use of the hemilabile 1,3-bis(diphenylphosphino)propane monoxide (dpppO) as a ligand led to faster reactions of more challenging 2-substituted
-
Palladium-Catalysed Heck Reactions of Alk-1-en-3-ones with Aryl Bromides: A Very Simple Access to (<i>E</i>)-1-Arylalk-1-en-3-ones作者:Mhamed Lemhadri、Yacoub Fall、Henri Doucet、Maurice SantelliDOI:10.1055/s-0028-1087976日期:——When appropriate reaction conditions are used, very high yields of (E)-1-arylalk-1-en-3-one derivatives can be obtained by palladium-catalysed reactions of alk-1-en-3-ones with aryl bromides. The tetraphosphine cis,cis,cis-1,2,3,4-tetrakis(diphenylphosphinomethyl)cyclopentane in combination with [Pd(C3H5)Cl]2 was found to be a very efficient catalyst for this reaction. In general, higher reaction rates were observed with electron-poor aryl bromides, but the electron-rich aryl bromides 1-bromo-4-(dimethylamino)benzene and 4-bromoanisole also led to the arylated enones in high yields. Even with sterically very congested aryl bromides such as 9-bromoanthracene, 1-bromo-2,4,6-trimethylbenzene or 1-bromo-2,4,6-triisopropylbenzene, the expected (E)-1-arylalk-1-en-3-ones were obtained in moderate to good yields. These enones appear to be unstable under the reaction conditions, and the addition of a small amount of hydroquinone to the reaction mixture was found to be crucial, especially for the vinylation of electron-deficient aryl bromides. A variety of alk-1-en-3-ones has been employed, and better results in terms of substrate/catalyst ratios were obtained when oct-1-en-3-one or hex-1-en-3-one was used than when but-1-en-3-one or pent-1-en-3-one was used. It should be noted that several reactions can be performed with as little as 0.1-0.001 mol% catalyst.在适当的反应条件下,通过钯催化的alk-1-en-3-ones与芳基溴化物的反应,可以获得高产率的(E)-1-芳基烯-1-烯-3-酮衍生物。铑催化剂中发现四膦配体顺,顺,顺-1,2,3,4-四(二苯膦甲基)环戊烷与[Pd(C3H5)Cl]2结合是一种非常高效的催化剂。通常情况下,电子贫乏的芳基溴化物显示出更高的反应速率,但电子富集的芳基溴化物1-溴-4-(二甲基氨基)苯和4-溴苯甲醚也能导致高产率的芳基化烯酮。即使在空间位阻很大的芳基溴化物如9-溴蒽、1-溴-2,4,6-三甲基苯或1-溴-2,4,6-三异丙基苯的情况下,期望的(E)-1-芳基烯-1-烯-3-酮也能在中等至良好的产率下获得。这些烯酮在反应条件下似乎是不稳定的,在反应混合物中加入少量的氢醌被发现是至关重要的,特别是对于电子缺乏的芳基溴化物的乙烯基化。已使用了多种alk-1-en-3-ones,当使用辛-1-烯-3-酮或己-1-烯-3-酮时,相对于底物/催化剂比例,得到了更好的结果,而不是丁-1-烯-3-酮或戊-1-烯-3-酮。值得注意的是,有几种反应可以在仅0.1-0.001 mol%催化剂的情况下进行。
-
Cationic Rhodium(I)/Bisphosphane Complex-Catalyzed Isomerization of Secondary Propargylic Alcohols to α,β-Enones作者:Ken Tanaka、Takeaki Shoji、Masao HiranoDOI:10.1002/ejoc.200700071日期:2007.6alcohols to α,β-enones. A kinetic resolution of secondary propargylic alcohols proceeded with moderate selectivity with [Rh((R)-BINAP)]OTf as a catalyst. Mechanistic studies revealed that the isomerization proceeds through intramolecular 1,3- and 1,2-hydrogen migration pathways. The isomerization of propargylic diol derivatives was also investigated, which revealed that 1,4-diketones, furans, and α,β-enones
-
Dual ligand-promoted palladium-catalyzed nondirected C–H alkenylation of aryl ethers作者:Biao Yin、Manlin Fu、Lei Wang、Jiang Liu、Qing ZhuDOI:10.1039/d0cc00940g日期:——Direct C-H functionalization of aryl ethers remains challenging owing to their low reactivity and selectivity. Herein, a novel strategy for nondirected C-H alkenylation of aryl ethers promoted by a dual ligand catalyst was demonstrated. This catalytic system readily achieved the highly efficient alkenylation of alkyl aryl ethers (anisole, phenetole, n-propyl phenyl ether, n-butyl phenyl ether and benzyl
表征谱图
-
氢谱1HNMR
-
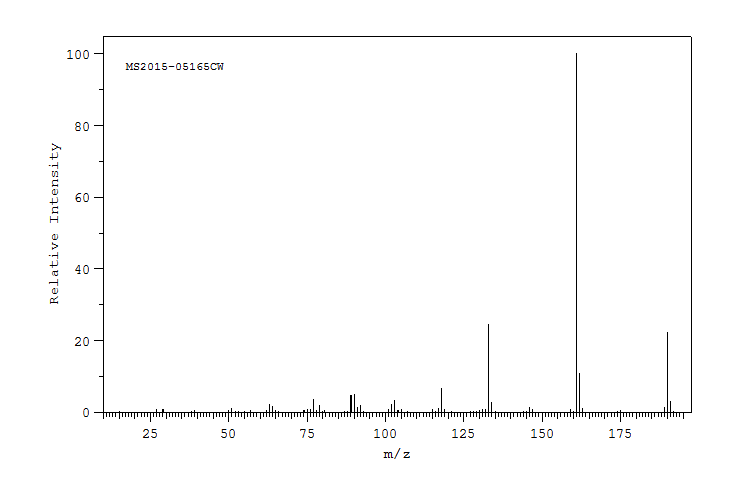
质谱MS
-
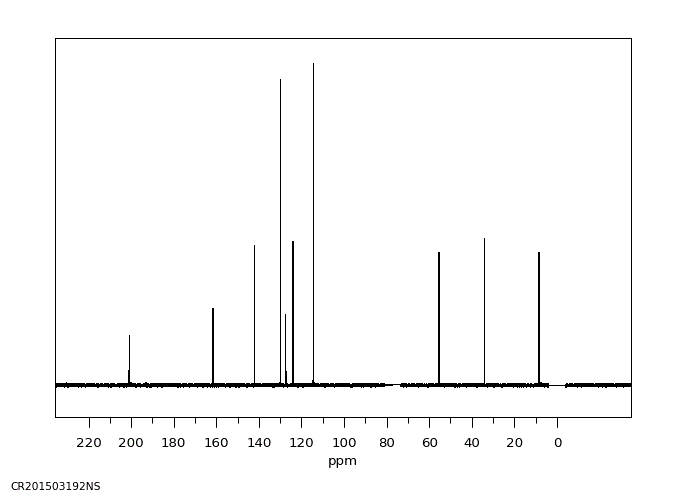
碳谱13CNMR
-
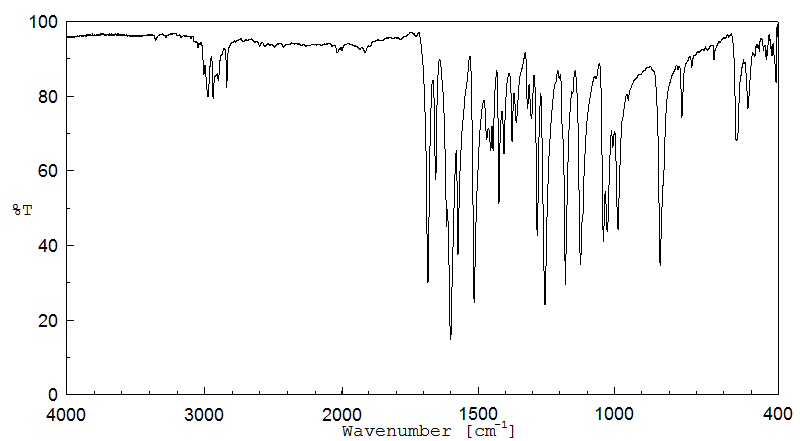
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(R)-3-(叔丁基)-4-(2,6-二异丙氧基苯基)-2,3-二氢苯并[d][1,3]氧杂磷杂环戊烯
(2S,3R)-3-(叔丁基)-2-(二叔丁基膦基)-4-甲氧基-2,3-二氢苯并[d][1,3]氧杂磷杂戊环
(2S,2''S,3S,3''S)-3,3''-二叔丁基-4,4''-二甲氧基-2,2'',3,3''-四氢-2,2''-联苯并[d][1,3]氧杂磷杂戊环
(2R,2''R,3R,3''R)-3,3''-二叔丁基-4,4''-二甲氧基-2,2'',3,3''-四氢-2,2''-联苯并[d][1,3]氧杂磷杂戊环
(2-氟-3-异丙氧基苯基)三氟硼酸钾
(+)-6,6'-{[(1R,3R)-1,3-二甲基-1,3基]双(氧)}双[4,8-双(叔丁基)-2,10-二甲氧基-丙二醇
麦角甾烷-6-酮,2,3,22,23-四羟基-,(2a,3a,5a,22S,23S)-
鲁前列醇
顺式6-(对甲氧基苯基)-5-己烯酸
顺式-铂戊脒碘化物
顺式-四氢-2-苯氧基-N,N,N-三甲基-2H-吡喃-3-铵碘化物
顺式-4-甲氧基苯基1-丙烯基醚
顺式-2,4,5-三甲氧基-1-丙烯基苯
顺式-1,3-二甲基-4-苯基-2-氮杂环丁酮
非那西丁杂质7
非那西丁杂质3
非那西丁杂质22
非那西丁杂质18
非那卡因
非布司他杂质37
非布司他杂质30
非布丙醇
雷诺嗪
阿达洛尔
阿达洛尔
阿莫噁酮
阿莫兰特
阿维西利
阿索卡诺
阿米维林
阿立酮
阿曲汀中间体3
阿普洛尔
阿普斯特杂质67
阿普斯特中间体
阿普斯特中间体
阿托西汀EP杂质A
阿托莫西汀杂质24
阿托莫西汀杂质10
阿托莫西汀EP杂质C
阿尼扎芬
阿利克仑中间体3
间苯胺氢氟乙酰氯
间苯二酚二缩水甘油醚
间苯二酚二异丙醇醚
间苯二酚二(2-羟乙基)醚
间苄氧基苯乙醇
间甲苯氧基乙酸肼
间甲苯氧基乙腈
间甲苯异氰酸酯