1--tetrahydro-1H-pyrimidin-2-on | 6584-29-8
中文名称
——
中文别名
——
英文名称
1--tetrahydro-1H-pyrimidin-2-on
英文别名
1-(4-chloro-phenyl)-tetrahydro-pyrimidin-2-one;1-(p-Chlorophenyl)-3,4,5,6-tetrahydro-2(1H)-pyrimidinone;1-(4-chlorophenyl)-1,3-diazinan-2-one
CAS
6584-29-8
化学式
C10H11ClN2O
mdl
——
分子量
210.663
InChiKey
SFPPEPCOGJSMRX-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
计算性质
-
辛醇/水分配系数(LogP):1.9
-
重原子数:14
-
可旋转键数:1
-
环数:2.0
-
sp3杂化的碳原子比例:0.3
-
拓扑面积:32.3
-
氢给体数:1
-
氢受体数:1
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 —— 1-(4-chlorophenyl)-3-(3-chloropropyl)urea 1073187-47-9 C10H12Cl2N2O 247.124
反应信息
-
作为反应物:描述:1-
-tetrahydro-1H-pyrimidin-2-on 、 1-(5-bromopentyloxy)-3-nitrobenzene 在 sodium hydride 作用下, 以 N,N-二甲基甲酰胺 为溶剂, 反应 3.0h, 生成参考文献:名称:设计和有效合成新型的芳硫脲衍生物作为有效的丙型肝炎病毒抑制剂摘要:设计并合成了一类新型的芳基硫脲HCV抑制剂,其在烷基连接基上具有各种功能,例如环脲,环硫脲,尿素和硫脲。在本文中,我们报告了这种新型的芳基硫脲衍生物的合成及其构效关系(SAR),这些衍生物在基于细胞的亚基因组HCV复制子测定中显示出对HCV的有效抑制活性。在测试的化合物中,发现新的咔唑衍生物64具有很强的抗HCV活性(EC 50 = 0.031),该咔唑衍生物在苯基和咔唑环之间具有八个碳原子的连接,并且在咔唑的N-9位具有甲苯基。μM),较低的细胞毒性(CC 50 > 50μM)和较高的选择性指数(SI> 1612)。DOI:10.1016/j.bmcl.2009.09.037 -
作为产物:描述:1-(4-chlorophenyl)-3-(3-chloropropyl)urea 在 sodium hydride 作用下, 以 四氢呋喃 、 N,N-二甲基甲酰胺 为溶剂, 生成 1-
-tetrahydro-1H-pyrimidin-2-on 参考文献:名称:设计和有效合成新型的芳硫脲衍生物作为有效的丙型肝炎病毒抑制剂摘要:设计并合成了一类新型的芳基硫脲HCV抑制剂,其在烷基连接基上具有各种功能,例如环脲,环硫脲,尿素和硫脲。在本文中,我们报告了这种新型的芳基硫脲衍生物的合成及其构效关系(SAR),这些衍生物在基于细胞的亚基因组HCV复制子测定中显示出对HCV的有效抑制活性。在测试的化合物中,发现新的咔唑衍生物64具有很强的抗HCV活性(EC 50 = 0.031),该咔唑衍生物在苯基和咔唑环之间具有八个碳原子的连接,并且在咔唑的N-9位具有甲苯基。μM),较低的细胞毒性(CC 50 > 50μM)和较高的选择性指数(SI> 1612)。DOI:10.1016/j.bmcl.2009.09.037
表征谱图
-
氢谱1HNMR
-
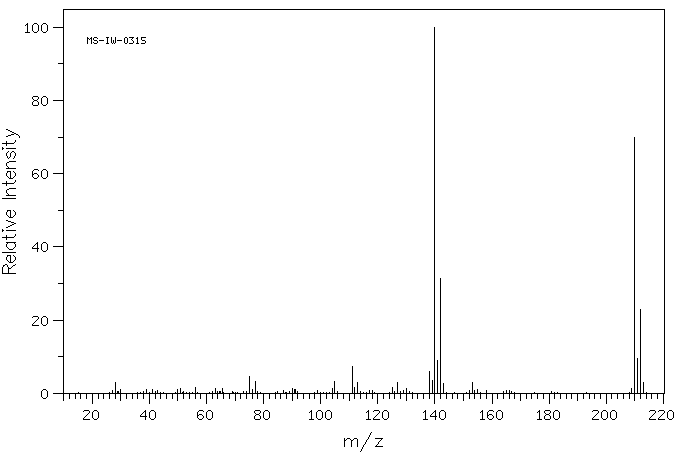
质谱MS
-
碳谱13CNMR
-
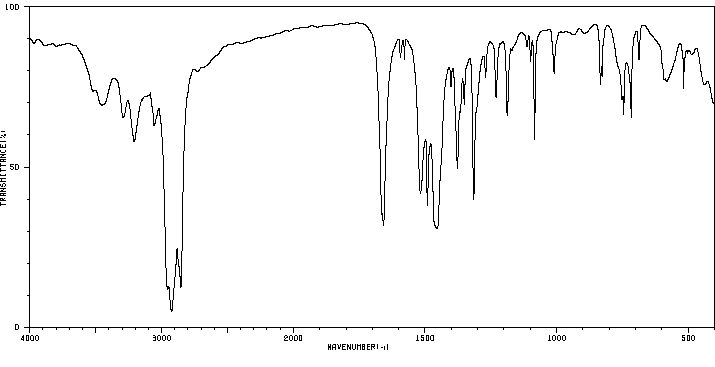
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(S)-3-(2-(二氟甲基)吡啶-4-基)-7-氟-3-(3-(嘧啶-5-基)苯基)-3H-异吲哚-1-胺
(6-羟基嘧啶-4-基)乙酸
(4,5-二甲氧基-1,2,3,6-四氢哒嗪)
鲁匹替丁
马西替坦杂质7
马西替坦杂质4
马西替坦杂质
马西替坦原料药杂质D
马西替坦原料药杂质B
马西替坦
顺式-4-{[5-溴-2-(2,5-二甲基-1H-吡咯-1-基)-6-甲基嘧啶-4-基]氨基}环己醇
非沙比妥
非巴氨酯
非尼啶醇
青鲜素钾盐
雷特格韦钾盐
雷特格韦相关化合物E(USP)
雷特格韦杂质8
雷特格韦EP杂质H
雷特格韦-RT9
雷特格韦
阿西莫司杂质3
阿西莫司
阿脲四水合物
阿脲一水合物
阿维霉素
阿米美啶
阿米洛利
阿米妥钠
阿洛巴比妥
阿普瑞西他滨
阿普比妥
阿巴卡韦相关化合物B(USP)
阿卡明
阿伐那非杂质V
阿伐那非杂质1
阿伐那非杂质
阿伐那非中间体
阿伐那非
铂(2+)二氯化6-甲基-1,3-二{2-[(2-甲基丙基)硫烷基]乙基}嘧啶-2,4(1H,3H)-二酮(1:1)
钴1,2,3,6-四氢-2,6-二氧代嘧啶-4-羧酸酯(1:2)
钠5-烯丙基-4,6-二氧代-1,4,5,6-四氢-2-嘧啶醇酸酯
钠5-乙基-4,6-二氧代-1,4,5,6-四氢-2-嘧啶醇酸酯
钠5-(2-溴丙-2-烯基)-5-丁烷-2-基-4,6-二氧代-1H-嘧啶-2-醇
醌肟腙
酒石酸噻吩嘧啶
那可比妥
辛基2,6-二氧代-1,2,3,6-四氢-4-嘧啶羧酸酯
赛乐西帕杂质3
赛乐西帕KSM3