6-甲基腺素 | 443-72-1
中文名称
6-甲基腺素
中文别名
N-甲基腺嘌呤;6-(甲氨基)嘌呤;6-甲氨基嘌呤;N6-甲基腺素;6-(甲氨基)嘌呤
英文名称
N-methyladenine
英文别名
N6-methyladenine;N-methyl-9H-purin-6-amine;6-(methylamino)purine;6-Methylamino-9H-purine;6-Methyladenine;N-methyl-7H-purin-6-amine
CAS
443-72-1
化学式
C6H7N5
mdl
MFCD00075820
分子量
149.155
InChiKey
CKOMXBHMKXXTNW-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:≥300 °C
-
沸点:256.3±50.0 °C(Predicted)
-
密度:1.57±0.1 g/cm3(Predicted)
-
溶解度:可溶于DMSO(略微加热)、甲醇(略微加热)
-
物理描述:Solid
计算性质
-
辛醇/水分配系数(LogP):0.6
-
重原子数:11
-
可旋转键数:1
-
环数:2.0
-
sp3杂化的碳原子比例:0.166
-
拓扑面积:66.5
-
氢给体数:2
-
氢受体数:4
安全信息
-
WGK Germany:3
-
海关编码:2933990090
-
储存条件:2-8°C
SDS
Material Safety Data Sheet
Section 1. Identification of the substance
Product Name: 6-(Methylamino)purine
Synonyms:
Section 2. Hazards identification
Harmful by inhalation, in contact with skin, and if swallowed.
Section 3. Composition/information on ingredients.
Ingredient name: 6-(Methylamino)purine
CAS number: 443-72-1
Section 4. First aid measures
Skin contact: Immediately wash skin with copious amounts of water for at least 15 minutes while removing
contaminated clothing and shoes. If irritation persists, seek medical attention.
Eye contact: Immediately wash skin with copious amounts of water for at least 15 minutes. Assure adequate
flushing of the eyes by separating the eyelids with fingers. If irritation persists, seek medical
attention.
Inhalation: Remove to fresh air. In severe cases or if symptoms persist, seek medical attention.
Ingestion: Wash out mouth with copious amounts of water for at least 15 minutes. Seek medical attention.
Section 5. Fire fighting measures
In the event of a fire involving this material, alone or in combination with other materials, use dry
powder or carbon dioxide extinguishers. Protective clothing and self-contained breathing apparatus
should be worn.
Section 6. Accidental release measures
Personal precautions: Wear suitable personal protective equipment which performs satisfactorily and meets local/state/national
standards.
Respiratory precaution: Wear approved mask/respirator
Hand precaution: Wear suitable gloves/gauntlets
Skin protection: Wear suitable protective clothing
Eye protection: Wear suitable eye protection
Methods for cleaning up: Mix with sand or similar inert absorbent material, sweep up and keep in a tightly closed container
for disposal. See section 12.
Environmental precautions: Do not allow material to enter drains or water courses.
Section 7. Handling and storage
Handling: This product should be handled only by, or under the close supervision of, those properly qualified
in the handling and use of potentially hazardous chemicals, who should take into account the fire,
health and chemical hazard data given on this sheet.
Storage: Store in closed vessels, refrigerated.
Section 8. Exposure Controls / Personal protection
Engineering Controls: Use only in a chemical fume hood.
Personal protective equipment: Wear laboratory clothing, chemical-resistant gloves and safety goggles.
General hydiene measures: Wash thoroughly after handling. Wash contaminated clothing before reuse.
Section 9. Physical and chemical properties
Appearance: Not specified
Boiling point: No data
Melting point: No data
Flash point: No data
Density: No data
Molecular formula: C6H7N5
Molecular weight: 149.2
Section 10. Stability and reactivity
Conditions to avoid: Heat, flames and sparks.
Materials to avoid: Oxidizing agents.
Possible hazardous combustion products: Carbon monoxide, nitrogen oxides.
Section 11. Toxicological information
No data.
Section 12. Ecological information
No data.
Section 13. Disposal consideration
Arrange disposal as special waste, by licensed disposal company, in consultation with local waste
disposal authority, in accordance with national and regional regulations.
Section 14. Transportation information
Non-harzardous for air and ground transportation.
Section 15. Regulatory information
No chemicals in this material are subject to the reporting requirements of SARA Title III, Section
302, or have known CAS numbers that exceed the threshold reporting levels established by SARA
Title III, Section 313.
SECTION 16 - ADDITIONAL INFORMATION
N/A
Section 1. Identification of the substance
Product Name: 6-(Methylamino)purine
Synonyms:
Section 2. Hazards identification
Harmful by inhalation, in contact with skin, and if swallowed.
Section 3. Composition/information on ingredients.
Ingredient name: 6-(Methylamino)purine
CAS number: 443-72-1
Section 4. First aid measures
Skin contact: Immediately wash skin with copious amounts of water for at least 15 minutes while removing
contaminated clothing and shoes. If irritation persists, seek medical attention.
Eye contact: Immediately wash skin with copious amounts of water for at least 15 minutes. Assure adequate
flushing of the eyes by separating the eyelids with fingers. If irritation persists, seek medical
attention.
Inhalation: Remove to fresh air. In severe cases or if symptoms persist, seek medical attention.
Ingestion: Wash out mouth with copious amounts of water for at least 15 minutes. Seek medical attention.
Section 5. Fire fighting measures
In the event of a fire involving this material, alone or in combination with other materials, use dry
powder or carbon dioxide extinguishers. Protective clothing and self-contained breathing apparatus
should be worn.
Section 6. Accidental release measures
Personal precautions: Wear suitable personal protective equipment which performs satisfactorily and meets local/state/national
standards.
Respiratory precaution: Wear approved mask/respirator
Hand precaution: Wear suitable gloves/gauntlets
Skin protection: Wear suitable protective clothing
Eye protection: Wear suitable eye protection
Methods for cleaning up: Mix with sand or similar inert absorbent material, sweep up and keep in a tightly closed container
for disposal. See section 12.
Environmental precautions: Do not allow material to enter drains or water courses.
Section 7. Handling and storage
Handling: This product should be handled only by, or under the close supervision of, those properly qualified
in the handling and use of potentially hazardous chemicals, who should take into account the fire,
health and chemical hazard data given on this sheet.
Storage: Store in closed vessels, refrigerated.
Section 8. Exposure Controls / Personal protection
Engineering Controls: Use only in a chemical fume hood.
Personal protective equipment: Wear laboratory clothing, chemical-resistant gloves and safety goggles.
General hydiene measures: Wash thoroughly after handling. Wash contaminated clothing before reuse.
Section 9. Physical and chemical properties
Appearance: Not specified
Boiling point: No data
Melting point: No data
Flash point: No data
Density: No data
Molecular formula: C6H7N5
Molecular weight: 149.2
Section 10. Stability and reactivity
Conditions to avoid: Heat, flames and sparks.
Materials to avoid: Oxidizing agents.
Possible hazardous combustion products: Carbon monoxide, nitrogen oxides.
Section 11. Toxicological information
No data.
Section 12. Ecological information
No data.
Section 13. Disposal consideration
Arrange disposal as special waste, by licensed disposal company, in consultation with local waste
disposal authority, in accordance with national and regional regulations.
Section 14. Transportation information
Non-harzardous for air and ground transportation.
Section 15. Regulatory information
No chemicals in this material are subject to the reporting requirements of SARA Title III, Section
302, or have known CAS numbers that exceed the threshold reporting levels established by SARA
Title III, Section 313.
SECTION 16 - ADDITIONAL INFORMATION
N/A
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 腺嘌呤 adenine 73-24-5 C5H5N5 135.128 6-肼基嘌呤 6-Hydrazinopurin 5404-86-4 C5H6N6 150.143 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 腺嘌呤 adenine 73-24-5 C5H5N5 135.128 —— N6-methyl-8-oxoadenine 89073-90-5 C6H7N5O 165.154 —— 8-bromo-6-methylaminopurine 89073-89-2 C6H6BrN5 228.051 N,9-二甲基嘌呤-6-胺 N6,N9-dimethyladenine 2009-52-1 C7H9N5 163.182 N(6)-(甲基亚硝基)腺苷 methyl-nitroso-(7(9)H-purin-6-yl)-amine 21928-82-5 C6H6N6O 178.153 —— [(6-methylamino)-9H-purin-9-yl]acetaldehyde —— C8H9N5O 191.192 —— 9,9'-ethylenebis[6-(methylamino)purine] 96750-61-7 C14H16N10 324.348 —— 9,9'-tetramethylenebis[6-(methylamino)purine] —— C16H20N10 352.402 —— 9-trimethylsilyl-6-methylaminopurine 32865-83-1 C9H15N5Si 221.337 —— 9,9'-(1,5-pentanediyl)bis[6-(methylamino)purine] —— C17H22N10 366.429 —— 9,9'-(1,7-heptanediyl)bis[6-(methylamino)purine] —— C19H26N10 394.482 —— 9,9'-(1,6-hexanediyl)bis[6-(methylamino)purine] —— C18H24N10 380.456 —— 9,9'-(1,9-nonanediyl)bis[6-(methylamino)purine] —— C21H30N10 422.536 —— 9-(5-bromopentyl)-6-(methylamino)purine —— C11H16BrN5 298.186 —— N6-methyl-9-[(4-hydroxy)-3-hydroxymethylbutyl]adenine —— C11H17N5O2 251.288 —— N6-methyl-9-[(1,3-dihydroxy-2-propoxy)methyl]adenine —— C10H15N5O3 253.261 —— methyl-(9-benzyl-9H-purin-6-yl)amine 81060-73-3 C13H13N5 239.28 2-[6-(甲基氨基)嘌呤-9-基]乙酸 2-(6-(methylamino)-9H-purin-9-yl)acetic acid 918334-37-9 C8H9N5O2 207.192 —— 1-methylisoguanine 23468-94-2 C6H7N5O 165.154 2-[[2-[6-(甲基氨基)嘌呤-7-基]乙酰基]氨基]乙酸 N-{[6-(Methylamino)-7H-purin-7-yl]acetyl}glycine 918334-39-1 C10H12N6O3 264.244 - 1
- 2
反应信息
-
作为反应物:描述:参考文献:名称:腺嘌呤阳离子的荧光摘要:DOI:10.1021/j100390a010
-
作为产物:描述:Benzotriazol-1-ylmethyl-(9H-purin-6-yl)-amine 在 sodium tetrahydroborate 作用下, 以 1,4-二氧六环 为溶剂, 反应 20.0h, 以75%的产率得到6-甲基腺素参考文献:名称:Katritzky, Alan R.; Rachwal, Stanislaw; Rachwal, Bogumila, Journal of the Chemical Society. Perkin transactions I, 1987, p. 805 - 810摘要:DOI:
文献信息
-
PHOSPHORAMIDITE BUILDING BLOCKS FOR SUGAR-CONJUGATED OLIGONUCLEOTIDES申请人:AM CHEMICALS LLC公开号:US20160083414A1公开(公告)日:2016-03-24Novel nucleoside phosphoramidite building blocks for preparation of synthetic oligonucleotides containing at least one phosphotriester linkage conjugated to a monosaccharide and synthetic processes for making the same are disclosed. Furthermore, oligomeric compounds are prepared using said building blocks, preferably followed by removal of protecting groups to provide monosaccharide-conjugated oligonucleotides.
-
POLY(PHOSPHOESTERS) FOR DELIVERY OF NUCLEIC ACIDS申请人:TRANSLATE BIO, INC.公开号:US20200277446A1公开(公告)日:2020-09-03Disclosed are polymers comprising the moiety A, which is a moiety of formula I: and pharmaceutically acceptable salts thereof, wherein R, R 1 , R 2 , L, n1 and n2 are as defined herein. These polymers are useful for delivering nucleic acids to subject. These polymers and pharmaceutically acceptable compositions comprising such polymers and nucleic acids can be useful for treating various diseases, disorders and conditions.揭示了包含A基团的聚合物,该基团是化学式I的基团:及其药用可接受的盐,其中R、R1、R2、L、n1和n2如本文所定义。这些聚合物对于将核酸传递给受试者是有用的。这些聚合物和包含这些聚合物和核酸的药用可接受组合物可用于治疗各种疾病、疾病和症状。
-
[EN] COMPOSITIONS AND METHODS FOR SYNTHESIS OF PHOSPHORYLATED MOLECULES<br/>[FR] COMPOSITIONS ET PROCÉDÉS DE SYNTHÈSE DE MOLÉCULES PHOSPHORYLÉES
-
SOLID SUPPORTS AND PHOSPHORAMIDITE BUILDING BLOCKS FOR OLIGONUCLEOTIDE CONJUGATES申请人:AM Chemicals LLC公开号:US20180016232A1公开(公告)日:2018-01-18Novel non-nucleoside solid supports and phosphoramidite building blocks for preparation of synthetic oligonucleotides containing at least one non-nucleosidic moiety conjugated to a ligand of practical interest and synthetic processes for making the same are disclosed. Furthermore, oligomeric compounds are prepared using said solid supports and phosphoramidite building blocks, preferably followed by removal of protecting groups to provide oligonucleotides conjugated to ligands of interest.
-
CARBOHYDRATE CONJUGATES AS DELIVERY AGENTS FOR OLIGONUCLEOTIDES申请人:Alnylam Pharmaceuticals, Inc.公开号:US20160051691A1公开(公告)日:2016-02-25The present invention provides iRNA agents comprising at least one subunit of the formula (I): wherein: A and B are each independently for each occurrence O, N(R N ) or S; X and Y are each independently for each occurrence H, OH, a hydroxyl protecting group, a phosphate group, a phosphodiester group, an activated phosphate group, an activated phosphite group, a phosphoramidite, a solid support, —P(Z′)(Z″)O-nucleoside, —P(Z′)(Z″)O-oligonucleotide, a lipid, a PEG, a steroid, a lipophile, a polymer, —P(Z′)(Z″)O-Linker-OP(Z′″)(Z″″)O-oligonucleotide, a nucleotide, an oligonucleotide, —P(Z′)(Z″)-formula(I), —P(Z′)(Z″)— or -Linker-R; R is L G , -Linker-L G , or has the structure shown below: L G is independently for each occurrence a carbohydrate, e.g., monosaccharide, disaccharide, trisaccharide, tetrasaccharide, oligosaccharide, polysaccharide; R N is independently for each occurrence H, methyl, ethyl, propyl, isopropyl, butyl, or benzyl; and Z′, Z″, Z′″ and Z″″ are each independently for each occurrence O or S.本发明提供了包含至少一个式(I)的亚单位的iRNA试剂: 其中: A和B分别独立于每次出现O、N(RN)或S; X和Y分别独立于每次出现H、OH、一个羟基保护基团、一个磷酸基团、一个磷酸二酯基团、一个活化磷酸基团、一个活化亚磷酸基团、一个磷酰胺基团、一个固相支持、-P(Z')(Z″)O-核苷、-P(Z')(Z″)O-寡核苷酸、一个脂质、一个PEG、一个类固醇、一个亲脂物质、一个聚合物、-P(Z')(Z″)O-连接子-OP(Z′″)(Z″″)O-寡核苷酸、一个核苷酸、一个寡核苷酸、-P(Z')(Z″)-式(I)、-P(Z')(Z″)-或-连接子-R; R是LG、-连接子-LG,或具有下面所示结构: LG独立于每次出现的是一种碳水化合物,例如,单糖、双糖、三糖、四糖、寡糖、多糖; RN独立于每次出现的是H、甲基、乙基、丙基、异丙基、丁基或苄基; Z'、Z″、Z′″和Z″″分别独立于每次出现的是O或S。
表征谱图
-
氢谱1HNMR
-
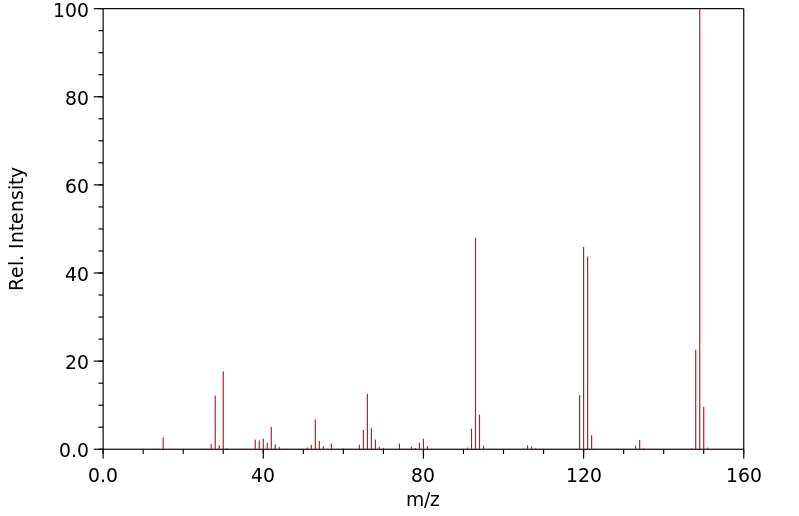
质谱MS
-
碳谱13CNMR
-
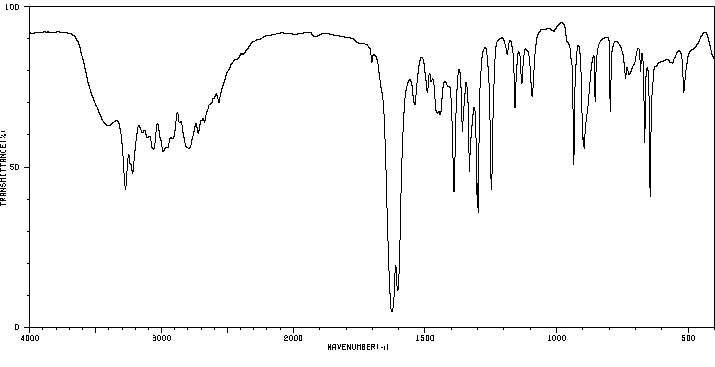
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
黄嘌呤钠盐
黄嘌呤
鸟嘌呤肟
鸟嘌呤盐酸盐
鸟嘌呤
顺式-二氨基二(O(6),9-二甲基鸟嘌呤-7)铂(II)二氯化物
顺式-2-(6-氨基-9H-嘌呤-9-基)-环己醇
阿罗茶碱
阿比茶碱
阿普西特-N-氧化物
阿昔洛韦钠
阿昔洛韦杂质K
阿昔洛韦杂质H
阿昔洛韦单磷酸盐
阿昔洛韦三磷酸酯
阿昔洛韦
阿德福韦酯杂质E
阿德福韦酯杂质12
阿德福韦酯杂质12
阿德福韦酯N6羟甲基杂质
阿德福韦酯 杂质C (阿德福韦单乙酯、单特戊酸甲酯)
阿德福韦酯
阿德福韦单特戊酸甲酯
阿德福韦-d4二磷酸三乙胺盐
阿德福韦
阿帕茶碱
阿司匹林,非那西汀和咖啡因
野杆菌素84
西潘茶碱
螺菲林
茶麻黄碱
茶苯海明
茶碱乙酸
茶碱一水合物
茶碱-D6
茶碱-8-丁酸
茶碱-2-氨基乙醇
茶碱
茶丙洛尔
苯酰胺,N-[9-[(2R)-2-羟基丙基]-9H-嘌呤-6-基]-
苯酰胺,N-(三甲基甲硅烷基)-N-[7-(三甲基甲硅烷基)-7H-嘌呤-6-基]-
苯酚,2-(3,4-二氢-2H-1-苯并吡喃-2-基)-
苯磺酸,4-(2,3,6,7-四氢-1,3,7-三甲基-2,6-二羰基-1H-嘌呤-8-基)-
苯甲酸咖啡鹼
苯甲腈,4-[(6,7-二氢-6-羰基-3H-嘌呤-3-基)甲基]-
苯呤司特
苄吡喃腺嘌呤
芬乙茶碱
芬乙茶碱
艾米替诺福韦