(E)-1-(4-methoxyphenyl)-3-(4-(methylthio)phenyl)prop-2-en-1-one
分子结构分类
中文名称
——
中文别名
——
英文名称
(E)-1-(4-methoxyphenyl)-3-(4-(methylthio)phenyl)prop-2-en-1-one
英文别名
(E)-1-(4-methoxyphenyl)-3-(4-methylsulfanylphenyl)prop-2-en-1-one
CAS
——
化学式
C17H16O2S
mdl
——
分子量
284.379
InChiKey
ZGIOCWWESCNBFZ-LFYBBSHMSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
计算性质
-
辛醇/水分配系数(LogP):4.2
-
重原子数:20
-
可旋转键数:5
-
环数:2.0
-
sp3杂化的碳原子比例:0.12
-
拓扑面积:51.6
-
氢给体数:0
-
氢受体数:3
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 对甲氧基苯乙酮 1-(4-methoxyphenyl)ethanone 100-06-1 C9H10O2 150.177
反应信息
-
作为产物:描述:4-(甲基巯基)苯甲醛 、 对甲氧基苯乙酮 在 sodium hydroxide 作用下, 以 乙醇 为溶剂, 生成 (E)-1-(4-methoxyphenyl)-3-(4-(methylthio)phenyl)prop-2-en-1-one参考文献:名称:Chalcones and Bis-Chalcones Analogs as DPPH and ABTS Radical Scavengers摘要:背景:许多合成支架物以及天然产物被确定为有效的抗氧化剂。本研究涉及对不同取代、具有药用特性的化合物类别“查尔酮和双查尔酮”进行抗氧化潜力评估。 方法:对一系列合成查尔酮1-13和双查尔酮14-18进行体外自由基清除活性测试。 结果:所有分子1-18在IC50范围内显示出明显的2,2-二苯基-1-苯基-亚硝基肼(DPPH)和2,2'-联苯二(3-乙基苯并噻唑啉-6-磺酸)(ABTS)自由基清除潜力,分别为0.58 ± 0.14 - 1.72 ± 0.03和0.49 ± 0.3 - 1.48 ± 0.06 μM。抗坏血酸(DPPH和ABTS的IC50分别为0.5 ± 0.1和0.46 ± 0.17 μM)被用作标准自由基清除剂。 结论:结构活性关系(SAR)显示了各种基团,包括-SMe和-OMe在清除活性中的积极参与。DOI:10.2174/1570180817999201001155032
文献信息
-
Alkene Synthesis by Photo‐Wolff‐Kischner Reaction of Sulfur Ylides and <i>N</i> ‐Tosylhydrazones作者:Pan‐Pan Gao、Dong‐Mei Yan、Ming‐Hang Bi、Min Jiang、Wen‐Jing Xiao、Jia‐Rong ChenDOI:10.1002/chem.202102671日期:2021.10.13A visible-light-driven and room temperature photo-Wolff-Kischner reaction of sulfur ylides and N-tosylhydrazones has been developed for the first time to provide modular access to alkene synthesis. The high functional group tolerance and broad substrate scope were demonstrated by more than 60 examples. Both E- and Z-olefinic stereochemistry in the products could be controlled with excellent stereoselectivity
-
CHALCONE DERIVATIVE COMPOUNDS申请人:NIPPON OIL AND FATS COMPANY, LIMITED公开号:EP0328669A1公开(公告)日:1989-08-23Chalcone derivative compounds represented by general formula (I) or (II), wherein R₁ and R₂ each represents a halogen atom, a hydroxy group, an amino group, a dimethylamino group, a nitro group, a cyano group, a phenyl group, an acetyl group, an alkyl group containing 1 to 18 carbon atoms or an alkyloxy group containing 1 to 22 carbon atoms, n₁ and n₂ each represents an integer of 0 to 21, and m₁ and m₂ each represents an integer of 0 to 5.
-
Chalcones and bis-chalcones: As potential α-amylase inhibitors; synthesis, in vitro screening, and molecular modelling studies作者:Adebayo Tajudeen Bale、Khalid Mohammed Khan、Uzma Salar、Sridevi Chigurupati、Tolulope Fasina、Farman Ali、Kanwal、Abdul Wadood、Muhammad Taha、Sitansu Sekhar Nanda、Mehreen Ghufran、Shahnaz PerveenDOI:10.1016/j.bioorg.2018.05.003日期:2018.9Despite of a diverse range of biological activities associated with chalcones and bis-chalcones, they are still neglected by the medicinal chemist for their possible alpha-amylase inhibitory activity. So, the current study is based on the evaluation of this class for the identification of new leads as alpha-amylase inhibitors. For that purpose, a library of substituted chalcones 1-13 and bis-chalcones 14-18 were synthesized and characterized by spectroscopic techniques EI-MS and H-1 NMR. CHN analysis was carried out and found in agreement with the calculated values. All compounds were evaluated for in vitro alpha-amylase inhibitory activity and demonstrated good activities in the range of IC50 = 1.25 +/- 1.05-2.40 +/- 0.09 mu M as compared to the standard acarbose (IC50 = 1.04 +/- 0.3 mu M). Limited structure-activity relationship (SAR) was established by considering the effect of different groups attached to aryl rings on varying inhibitory activity. SMe group in chalcones and OMe group in bis-chalcones were found more influential on the activity than other groups. However, in order to predict the involvement of different groups in the binding interactions with the active site of alpha-amylase enzyme, in silico studies were also conducted.
-
Synthesis, antimycobacterial activity evaluation, and QSAR studies of chalcone derivatives作者:P.M. Sivakumar、S. Prabu Seenivasan、Vanaja Kumar、Mukesh DobleDOI:10.1016/j.bmcl.2006.12.112日期:2007.3In order to develop relatively small molecules as antimycobacterial agents, twenty-five chalcones were synthesized, their activity was evaluated, and quantitative structure-activity relationship (QSAR) was developed. The synthesis was based on the Claisen-Schimdt scheme and the resultant compounds were tested for antitubercular activity by luciferase reporter phage (LRP) assay. Compound C-24 was found to be the most active (similar to 99%) in this series based on the percentage reduction in Relative Light Units at both 50 and 100 mu g/ml levels, followed by compound C-21. Four compounds at the 50 mu g/ml and eight compounds at the 100 mu g/ml showed activity above 90% level. QSAR model was developed between activity and spatial, topological, and ADME descriptors for the 50 mu g/ml data. The statistical measures such as r, r(2), q(2), and F values obtained for the training set were in acceptable range and hence this relationship was used for the test set. The predictive ability of the model is satisfactory (q(2) = 0.56) and it can be used for designing similar group of compounds. (c) 2007 Elsevier Ltd. All rights reserved.
表征谱图
-
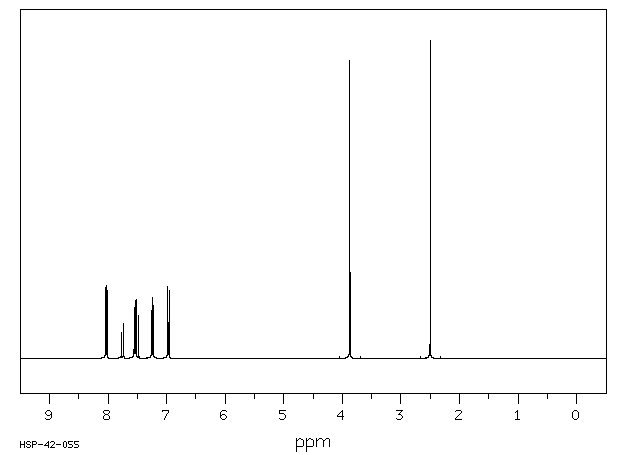
氢谱1HNMR
-
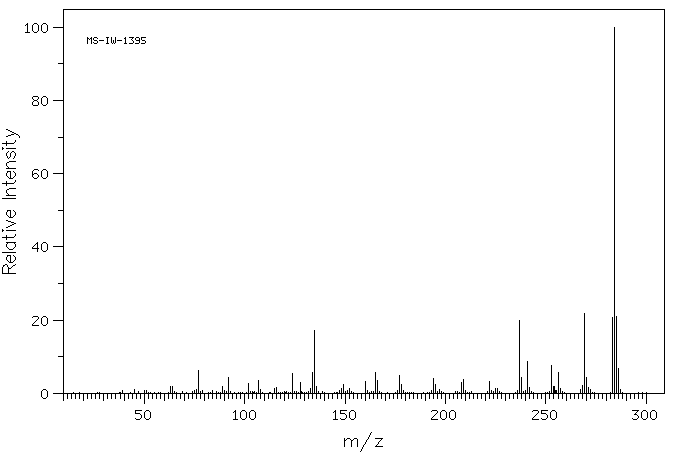
质谱MS
-
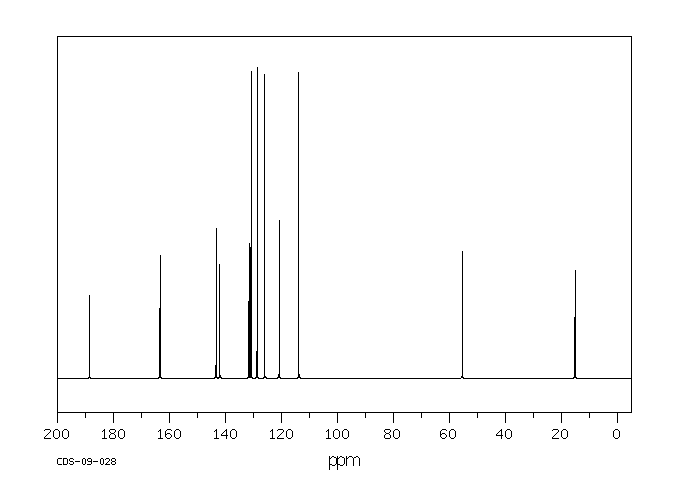
碳谱13CNMR
-
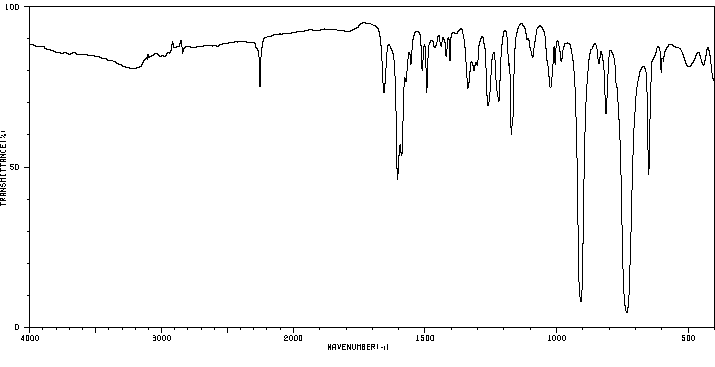
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(2Z)-1,3-二苯基-2-丙烯-1-酮,2-丙烯-1-酮,1,3-二苯基-,(2Z)-
龙血素D
龙血素A
龙血素 B
黄色当归醇F
黄色当归醇B
黄腐醇; 黄腐酚
黄腐醇 D; 黄腐酚 D
黄腐酚B
黄腐酚
黄腐酚
黄卡瓦胡椒素 C
高紫柳查尔酮
阿普非农
阿司巴汀
阿伏苯宗
金鸡菊查耳酮
邻肉桂酰苯甲酸
达泊西汀杂质25
豆蔻明
补骨脂色烯查耳酮
补骨脂查耳酮
补骨脂呋喃查耳酮
补骨脂乙素
蜡菊亭; 4,2',4'-三羟基-6'-甲氧基查耳酮
苯酚,4-[3-(2-羟基苯基)-1-苯基丙基]-2-(3-苯基丙基)-
苯磺酰胺,N-[4-[3-(3-羟基苯基)-1-羰基-2-丙烯基]苯基]-
苯磺酰胺,N-[3-[3-(4-羟基-3-甲氧苯基)-1-羰基-2-丙烯基]苯基]-
苯磺酰胺,4-甲氧基-N,N-二甲基-2-(3-羰基-3-苯基-1-丙烯基)-,(E)-
苯磺酰氯化,4,5-二甲氧基-2-(3-羰基-3-苯基-1-丙烯基)-,(E)-
苯磺酰氯,4-甲氧基-3-(3-羰基-3-苯基-1-丙烯基)-,(E)-
苯甲醇,4-甲氧基-a-[2-(4-甲氧苯基)乙烯基]-
苯甲酸-[4-(3-氧代-3-苯基-丙烯基)-苯胺]
苯甲酸,3-[3-(4-溴苯基)-1-羰基-2-丙烯基]-4-羟基-
苯甲酰(2-羟基苯酰)甲烷
苯甲腈,4-(1-羟基-3-羰基-3-苯基丙基)-
苯基[2-(1-萘基)乙烯基]甲酮
苯基-(三苯基-丙-2-炔基)-醚
苯基-(2-苯基-2,3-二氢-苯并噻唑-2-基)-甲酮
苯亚甲基苯乙酮
苯乙酰腈,a-(1-氨基-2-苯基亚乙基)-
苯丙酸,a-苯甲酰-b-羰基-,苯基(苯基亚甲基)酰肼
苯,1-(2,2-二甲基-3-苯基丙基)-2-甲基-
苏木查耳酮
苄桂哌酯
苄基(4-氯-2-(3-氧代-1,3-二苯基丙基)苯基)氨基甲酸酯
芦荟提取物
腈苯唑
胀果甘草宁C
聚磷酸根皮酚