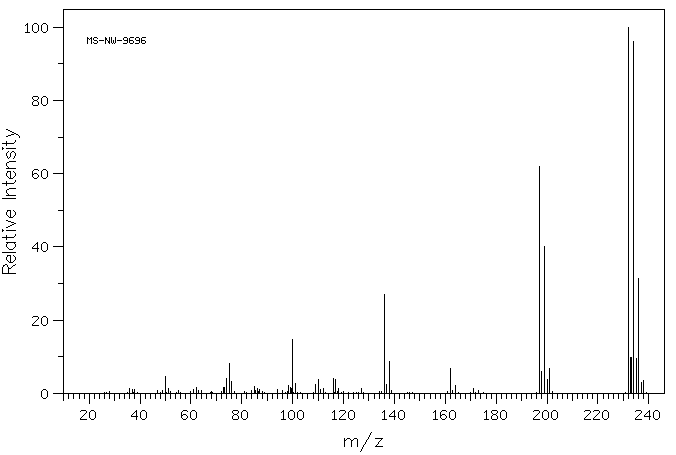
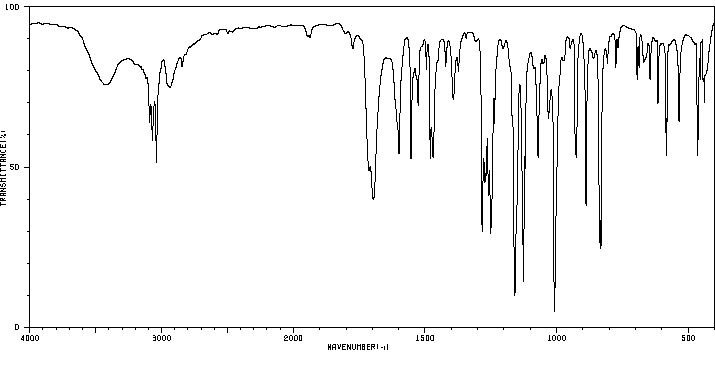
2,3,6-三氯喹喔啉 | 2958-87-4
中文名称
2,3,6-三氯喹喔啉
中文别名
2,3,6-三氯喹噁啉
英文名称
2,3,6-trichloro-quinoxaline
英文别名
6-chloro-2,3-dichloroquinoxaline;2,3,6-Trichloroquinoxaline
CAS
2958-87-4
化学式
C8H3Cl3N2
mdl
MFCD00006721
分子量
233.484
InChiKey
GZEGFNCRZUGIOB-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:143-148 °C
-
沸点:373.7°C (rough estimate)
-
密度:1.600
-
稳定性/保质期:
遵照规定使用和储存,则不会分解。
计算性质
-
辛醇/水分配系数(LogP):3.8
-
重原子数:13
-
可旋转键数:0
-
环数:2.0
-
sp3杂化的碳原子比例:0.0
-
拓扑面积:25.8
-
氢给体数:0
-
氢受体数:2
安全信息
-
危险品标志:Xi
-
危险类别码:R36/37/38
-
海关编码:2933990090
-
安全说明:S26,S37/39
-
储存条件:存放于阴凉干燥处。
SDS
| Name: | 2 3 6-Trichloroquinoxaline 98% Material Safety Data Sheet |
| Synonym: | None Known |
| CAS: | 2958-87-4 |
Synonym:None Known
Section 2 - COMPOSITION, INFORMATION ON INGREDIENTS
| CAS# | Chemical Name | content | EINECS# |
| 2958-87-4 | 2,3,6-Trichloroquinoxaline | 98% | 220-987-3 |
Risk Phrases: 36/37/38
Section 3 - HAZARDS IDENTIFICATION
EMERGENCY OVERVIEW
Irritating to eyes, respiratory system and skin.
Potential Health Effects
Eye:
Causes eye irritation. May cause chemical conjunctivitis.
Skin:
Causes skin irritation.
Ingestion:
May cause gastrointestinal irritation with nausea, vomiting and diarrhea.
Inhalation:
Causes respiratory tract irritation. Can produce delayed pulmonary edema.
Chronic:
Effects may be delayed.
Section 4 - FIRST AID MEASURES
Eyes: Immediately flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid.
Skin:
Get medical aid. Flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes. Wash clothing before reuse.
Ingestion:
Never give anything by mouth to an unconscious person. Get medical aid. Do NOT induce vomiting. If conscious and alert, rinse mouth and drink 2-4 cupfuls of milk or water.
Inhalation:
Remove from exposure and move to fresh air immediately. If breathing is difficult, give oxygen. Get medical aid. Do NOT use mouth-to-mouth resuscitation. If breathing has ceased apply artificial respiration using oxygen and a suitable mechanical device such as a bag and a mask.
Notes to Physician:
Treat symptomatically and supportively.
Section 5 - FIRE FIGHTING MEASURES
General Information:
As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear. During a fire, irritating and highly toxic gases may be generated by thermal decomposition or combustion. Runoff from fire control or dilution water may cause pollution.
Extinguishing Media:
In case of fire, use water, dry chemical, chemical foam, or alcohol-resistant foam. Use agent most appropriate to extinguish fire.
Section 6 - ACCIDENTAL RELEASE MEASURES
General Information: Use proper personal protective equipment as indicated in Section 8.
Spills/Leaks:
Vacuum or sweep up material and place into a suitable disposal container. Clean up spills immediately, observing precautions in the Protective Equipment section. Avoid generating dusty conditions.
Provide ventilation.
Section 7 - HANDLING and STORAGE
Handling:
Wash thoroughly after handling. Remove contaminated clothing and wash before reuse. Minimize dust generation and accumulation. Avoid contact with eyes, skin, and clothing. Keep container tightly closed.
Avoid ingestion and inhalation. Use with adequate ventilation.
Storage:
Keep container closed when not in use. Store in a tightly closed container. Store in a cool, dry, well-ventilated area away from incompatible substances.
Section 8 - EXPOSURE CONTROLS, PERSONAL PROTECTION
Engineering Controls:
Facilities storing or utilizing this material should be equipped with an eyewash facility and a safety shower. Use adequate ventilation to keep airborne concentrations low.
Exposure Limits CAS# 2958-87-4: Personal Protective Equipment Eyes: Wear appropriate protective eyeglasses or chemical safety goggles as described by OSHA's eye and face protection regulations in 29 CFR 1910.133 or European Standard EN166.
Skin:
Wear appropriate protective gloves and clothing to prevent skin exposure.
Clothing:
Wear appropriate protective clothing to prevent skin exposure.
Respirators:
Follow the OSHA respirator regulations found in 29 CFR 1910.134 or European Standard EN 149. Use a NIOSH/MSHA or European Standard EN 149 approved respirator if exposure limits are exceeded or if irritation or other symptoms are experienced.
Section 9 - PHYSICAL AND CHEMICAL PROPERTIES
Physical State: Powder
Color: light brown
Odor: None reported.
pH: Not available.
Vapor Pressure: Not available.
Viscosity: Not available.
Boiling Point: Not available.
Freezing/Melting Point: 144.00 - 146.00 deg C
Autoignition Temperature: Not applicable.
Flash Point: Not applicable.
Explosion Limits, lower: Not available.
Explosion Limits, upper: Not available.
Decomposition Temperature:
Solubility in water:
Specific Gravity/Density:
Molecular Formula: C8H3Cl3N2
Molecular Weight: 233.48
Section 10 - STABILITY AND REACTIVITY
Chemical Stability:
Stable at room temperature in closed containers under normal storage and handling conditions.
Conditions to Avoid:
Incompatible materials, dust generation, excess heat, strong oxidants.
Incompatibilities with Other Materials:
Oxidizing agents.
Hazardous Decomposition Products:
Hydrogen chloride, nitrogen oxides, carbon monoxide, irritating and toxic fumes and gases, carbon dioxide.
Hazardous Polymerization: Has not been reported.
Section 11 - TOXICOLOGICAL INFORMATION
RTECS#:
CAS# 2958-87-4: VD3625000 LD50/LC50:
Not available.
Carcinogenicity:
2,3,6-Trichloroquinoxaline - Not listed by ACGIH, IARC, or NTP.
Other:
See actual entry in RTECS for complete information.
Section 12 - ECOLOGICAL INFORMATION
Section 13 - DISPOSAL CONSIDERATIONS
Dispose of in a manner consistent with federal, state, and local regulations.
Section 14 - TRANSPORT INFORMATION
IATA
Not regulated as a hazardous material.
IMO
Not regulated as a hazardous material.
RID/ADR
Not regulated as a hazardous material.
Section 15 - REGULATORY INFORMATION
European/International Regulations
European Labeling in Accordance with EC Directives
Hazard Symbols: XI
Risk Phrases:
R 36/37/38 Irritating to eyes, respiratory system
and skin.
Safety Phrases:
S 26 In case of contact with eyes, rinse immediately
with plenty of water and seek medical advice.
S 37/39 Wear suitable gloves and eye/face
protection.
WGK (Water Danger/Protection)
CAS# 2958-87-4: No information available.
Canada
CAS# 2958-87-4 is listed on Canada's NDSL List.
CAS# 2958-87-4 is not listed on Canada's Ingredient Disclosure List.
US FEDERAL
TSCA
CAS# 2958-87-4 is listed on the TSCA inventory.
SECTION 16 - ADDITIONAL INFORMATION
N/A
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 6-氯喹喔啉 6-chloroquinoxaline 5448-43-1 C8H5ClN2 164.594 2,3-二羟基-6-氯喹喔啉 6-chloroquinoxaline-2,3(1H,4H)-dione 6639-79-8 C8H5ClN2O2 196.593 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 —— 3,6-dichloroquinoxalin-2(1H)-one 1374748-37-4 C8H4Cl2N2O 215.039 —— 2-amino-3,6-dichloroquinoxaline 89978-28-9 C8H5Cl2N3 214.054 —— 3,7-dichloroquinoxalin-2-amine 89978-29-0 C8H5Cl2N3 214.054 —— 3,6-dichloro-2-methoxy-quinoxaline 281209-15-2 C9H6Cl2N2O 229.065 —— 2,6-dichloro-3-methoxyquinoxaline 91895-09-9 C9H6Cl2N2O 229.065 —— 2,6-dichloro-3-hydrazinoquinoxaline 91895-34-0 C8H6Cl2N4 229.068 —— 2,6-dichloro-3-(N,N-dimethylamino)quinoxaline 81057-47-8 C10H9Cl2N3 242.108 —— 6-Chlor-2,3-diamino-chinoxalin 52312-40-0 C8H7ClN4 194.623 —— 2-(3,7-dichloroquinoxalin-2-ylamino)ethanol 1217300-50-9 C10H9Cl2N3O 258.107 —— 2-(3,6-dichloroquinoxalin-2-ylamino)propan-1-ol 1217300-57-6 C11H11Cl2N3O 272.134 —— 2-(3,7-dichloroquinoxalin-2-ylamino)propan-1-ol 1217300-56-5 C11H11Cl2N3O 272.134 (3,7-二氯-喹喔啉-2-基)-(2,2-二甲氧基-乙基)-胺 (3,7-Dichloro-quinoxalin-2-yl)-(2,2-dimethoxy-ethyl)-amine 1026883-72-6 C12H13Cl2N3O2 302.16 - 1
- 2
反应信息
-
作为反应物:描述:2,3,6-三氯喹喔啉 在 盐酸 、 sodium 、 一水合肼 、 溶剂黄146 、 三氯氧磷 作用下, 以 乙醇 为溶剂, 反应 68.5h, 生成 7-chloro-4-(diethylamino)<1,2,3>triazolo<4,3-a>quinoxaline参考文献:名称:4-氨基[1,2,4]三唑并[4,3-a]喹喔啉。一类新型的有效腺苷受体拮抗剂和潜在的快速发作抗抑郁药。摘要:制备了一系列的4-氨基[1,2,4]三唑并[4,3-a]喹喔啉。此类化合物中的许多化合物在急性给药后可降低大鼠Porsolt行为绝望模型的固定性,因此可能具有作为新型且速效抗抑郁药的治疗潜力。该测试中的最佳活性与1位上的氢,CF3或小烷基,与4位上的小烷基取代的NH2,NH-乙酰基或胺以及氢或8个卤素取代基有关在芳香环上。此外,许多这些4-氨基[1,2,4]三唑并[4,3-a]喹喔啉与腺苷A1和A2受体狂热结合,在某些情况下非常选择性地结合。这些化合物的A1亲和力是通过抑制大鼠大脑皮层膜中tri化的CHA(N6-环己基腺苷)结合而测定的,A2亲和性是通过抑制tri化的NECA(5'-(N-乙基氨基甲酰基)腺苷)与大鼠纹状体匀浆中的结合来测定的。存在冷的N6-环戊基腺苷。结构-活性关系(SAR)研究表明,最佳的A1亲和力与1位的乙基,CF3或C2F5、4位的NH-iPr或NH-环烷基以及8-DOI:10.1021/jm00170a031
-
作为产物:描述:参考文献:名称:4-氨基[1,2,4]三唑并[4,3-a]喹喔啉。一类新型的有效腺苷受体拮抗剂和潜在的快速发作抗抑郁药。摘要:制备了一系列的4-氨基[1,2,4]三唑并[4,3-a]喹喔啉。此类化合物中的许多化合物在急性给药后可降低大鼠Porsolt行为绝望模型的固定性,因此可能具有作为新型且速效抗抑郁药的治疗潜力。该测试中的最佳活性与1位上的氢,CF3或小烷基,与4位上的小烷基取代的NH2,NH-乙酰基或胺以及氢或8个卤素取代基有关在芳香环上。此外,许多这些4-氨基[1,2,4]三唑并[4,3-a]喹喔啉与腺苷A1和A2受体狂热结合,在某些情况下非常选择性地结合。这些化合物的A1亲和力是通过抑制大鼠大脑皮层膜中tri化的CHA(N6-环己基腺苷)结合而测定的,A2亲和性是通过抑制tri化的NECA(5'-(N-乙基氨基甲酰基)腺苷)与大鼠纹状体匀浆中的结合来测定的。存在冷的N6-环戊基腺苷。结构-活性关系(SAR)研究表明,最佳的A1亲和力与1位的乙基,CF3或C2F5、4位的NH-iPr或NH-环烷基以及8-DOI:10.1021/jm00170a031
文献信息
-
Fragment Based Design of New H<sub>4</sub> Receptor−Ligands with Anti-inflammatory Properties in Vivo作者:Rogier A. Smits、Herman D. Lim、Agnes Hanzer、Obbe P. Zuiderveld、Elena Guaita、Maristella Adami、Gabriella Coruzzi、Rob Leurs、Iwan J. P. de EschDOI:10.1021/jm7014217日期:2008.4.1and evaluated a series of compounds at the human histamine H 4 receptor (H 4R) from which 2-(4-methyl-piperazin-1-yl)-quinoxaline ( 3) was identified as a new lead structure for H 4R ligands. Exploration of the structure-activity relationship (SAR) of this scaffold led to the identification of 6,7-dichloro 3-(4-methylpiperazin-1-yl)quinoxalin-2(1 H)-one (VUF 10214, 57) and 2-benzyl-3-(4-methyl-pip
-
HETEROCYCLIC INHIBITORS OF HISTAMINE RECEPTORS FOR THE TREATMENT OF DISEASE申请人:Borchardt Allen J.公开号:US20100120741A1公开(公告)日:2010-05-13The present invention relates to compounds and methods which may be useful as inhibitors of H 1 R and/or H 4 R for the treatment or prevention of inflammatory, autoimmune, allergic, and ocular diseases.本发明涉及化合物和方法,这些化合物和方法可能用作H1R和/或H4R的抑制剂,用于治疗或预防炎症性、自身免疫性、过敏性和眼部疾病。
-
Method of using [1,2,4]triazolo[4,3-a]quinoxaline-4-amine derivatives as申请人:Pfizer Inc.公开号:US04547501A1公开(公告)日:1985-10-15A series of novel [1,2,4]triazolo[4,3-a]quinoxaline-4-amine derivatives wherein the amine group is optionally substituted with lower alkyl, phenylalkyl having up to three carbon atoms in the alkyl moiety or alkanoyl having from two to five carbon atoms, or the amine group alternatively completes a piperazino ring, the quinoxaline ring is optionally substituted with fluorine, chlorine, bromine or methoxy, and the triazolo ring is optionally substituted with lower alkyl, lower perfluoroalkyl or phenyl are disclosed. These novel compounds are useful for treatment of symptoms associated with depression. Also disclosed are pharmaceutical compositions containing the novel compounds of this invention and a method of using the compounds in the treatment of depression and fatigue.
-
Non-peptide GLP-1 agonists申请人:Teng Min公开号:US06927214B1公开(公告)日:2005-08-09Novel non-peptide GLP-1 agonists, pharmaceutical compositions comprising them, use of the non-peptide GLP-1 agonists for the preparation of pharmaceutical compositions and methods for the treatment and/or prevention of disorders and diseases wherein an activation of the human GLP-1 receptor is beneficial, especially metabolic disorders such as IGT, Type 1 diabetes, Type 2 diabetes and obesity.新型非肽类GLP-1激动剂,包括它们的药物组合物,使用非肽类GLP-1激动剂制备药物组合物以及治疗和/或预防激活人类GLP-1受体有益的疾病和疾病的方法,特别是代谢性疾病,如IGT、1型糖尿病、2型糖尿病和肥胖症。
-
Palladium-Catalyzed Intramolecular Fujiwara-Hydroarylation: Synthesis of Benzo[<i>a</i>]phenazines Derivatives作者:Sonu Kumar、Rakesh K. Saunthwal、Mohammad Mujahid、Trapti Aggarwal、Akhilesh K. VermaDOI:10.1021/acs.joc.6b02096日期:2016.10.21metalation followed by concomitant intramolecular trans-insertion of C–C triple bond to aryl-Pd complex. The results were supported by various control experiments including with electron–deficient arenes and deuterium labeling studies. The deuterium labeling studies supports electrophilic palladation of aromatic C–H over activation of C–C triple bond of alkyne. The structure of synthesized compounds
表征谱图
-
氢谱1HNMR
-
质谱MS
-
碳谱13CNMR
-
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(12羟基吲[2,1-b〕喹唑啉-6(12H)-酮)
黑暗猝灭剂BHQ-3,BHQ-3NHS
鸭嘴花酚碱
鸭嘴花碱酮;(S)-2,3-二氢-3,7-二羟基吡咯并[2,1-b]喹唑啉-9(1H)-酮
鸭嘴花碱酮
鸭嘴花碱盐酸盐
鲁米诺单钠盐
鲁米诺
骆驼蓬碱
颜料蓝64
颜料蓝60
顺式-卤夫酮
顺式-(喹喔啉-2-基)丙烯腈1,4-二氧化物
非奈利酮
青黛酮
雷替曲塞杂质1
阿法替尼杂质J
阿法替尼杂质I
阿法替尼杂质28
阿法替尼杂质18
阿法替尼杂质13
阿法替尼杂质
阿法替尼中间体
阿法替尼
阿法替尼
阿朴藏红
阿巴康唑
阿夫唑嗪杂质A
阿夫唑嗪杂质
阿夫唑嗪EP杂质C
阿夫唑嗪
阿喹司特
阿呋唑嗪杂质
阿呋唑嗪杂质
铜迈星
铁诱导细胞死亡激活剂
钠四丙基硼酸酯
酸性蓝98
酸性红101
酮色林醇
酞联氮基[2,3-b]酞嗪-5,14-二酮,7,12-二氢-
酞嗪-5-羧酸
酞嗪-2-氧化物
酚藏花红
酚嗪
酒石酸溴莫尼定
邻苯二甲酰肼
还原黄6GD
还原蓝6
达尼喹酮