2-(4-溴丁基)-1,3-二氧戊环 | 87227-41-6
中文名称
2-(4-溴丁基)-1,3-二氧戊环
中文别名
5-溴戊醛乙二醇缩醛;2-(4-溴丁基)-1,3-二恶烷
英文名称
2-(4-bromobutyl)-1,3-dioxolane
英文别名
2-(4-bromo-1-butyl)-1,3-dioxolane
CAS
87227-41-6
化学式
C7H13BrO2
mdl
——
分子量
209.083
InChiKey
UQTZEBQZCBQLAN-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
沸点:237.6±15.0 °C(Predicted)
-
密度:1.38
计算性质
-
辛醇/水分配系数(LogP):1.7
-
重原子数:10
-
可旋转键数:4
-
环数:1.0
-
sp3杂化的碳原子比例:1.0
-
拓扑面积:18.5
-
氢给体数:0
-
氢受体数:2
安全信息
-
安全说明:S26,S36/37/39
-
危险类别码:R36/37/38
-
海关编码:2932999099
-
包装等级:III
-
危险类别:6.1
-
危险性防范说明:P301+P310
-
危险品运输编号:2810
-
危险性描述:H301
-
储存条件:冷藏
SDS
2-(4-Bromobutyl)-1,3-dioxolane Revision number: 1
SAFETY DATA SHEET
Section 1. BASE INFORMATION
Product name: 2-(4-Bromobutyl)-1,3-dioxolane
Revision number: 1
Section 2. HAZARDS IDENTIFICATION
Classification of the GHS
PHYSICAL HAZARDS Not classified
HEALTH HAZARDS Not classified
Not classified
ENVIRONMENTAL HAZARDS
GHS label elements
None
Pictograms or hazard symbols
Signal word No signal word
None
Hazard statement
Precautionary statements None
Section 3. COMPOSITION/INFORMATION ON INGREDIENTS
Substance/mixture: Substance
Component(s): 2-(4-Bromobutyl)-1,3-dioxolane
Percent: >94.0%(GC)
CAS Number: 87227-41-6
Synonyms: 5-Bromovaleraldehyde Ethylene Acetal
Chemical Formula: C7H13BrO2
Section 4. FIRST AID MEASURES
Inhalation: Remove victim to fresh air and keep at rest in a position comfortable for breathing.
Get medical advice/attention if you feel unwell.
Skin contact: Remove/Take off immediately all contaminated clothing. Rinse skin with
water/shower. If skin irritation or rash occurs: Get medical advice/attention.
Eye contact: Rinse cautiously with water for several minutes. Remove contact lenses, if present
and easy to do. Continue rinsing. If eye irritation persists: Get medical
advice/attention.
Ingestion: Get medical advice/attention if you feel unwell. Rinse mouth.
A rescuer should wear personal protective equipment, such as rubber gloves and air-
Protection of first-aiders:
tight goggles.
Section 5. FIRE-FIGHTING MEASURES
Suitable extinguishing Dry chemical, foam, water spray, carbon dioxide.
media:
Extinguishing media not to Solid streams of water
be used:
Specific hazards: Take care as it may decompose upon combustion or in high temperatures to
generate poisonous fume.
2-(4-Bromobutyl)-1,3-dioxolane
Section 5. FIRE-FIGHTING MEASURES
Specific methods: Fire-extinguishing work is done from the windward and the suitable fire-extinguishing
method according to the surrounding situation is used. Uninvolved persons should
evacuate to a safe place. In case of fire in the surroundings: Remove movable
containers if safe to do so.
Special protective When extinguishing fire, be sure to wear personal protective equipment.
equipment for firefighters:
Section 6. ACCIDENTAL RELEASE MEASURES
Use personal protective equipment. Keep people away from and upwind of spill/leak.
Personal precautions,
protective equipment and Ensure adequate ventilation. Entry to non-involved personnel should be controlled
emergency procedures: around the leakage area by roping off, etc.
Environmental precautions: Prevent product from entering drains.
Methods and materials for Absorb spilled material in a suitable absorbent (e.g. rag, dry sand, earth, saw-dust).
containment and cleaning In case of large amount of spillage, contain a spill by bunding. Adhered or collected
up: material should be promptly disposed of, in accordance with appropriate laws and
regulations.
Section 7. HANDLING AND STORAGE
Handling
Technical measures: Handling is performed in a well ventilated place. Wear suitable protective equipment.
Prevent generation of vapor or mist. Wash hands and face thoroughly after handling.
Use a ventilation, local exhaust if vapor or aerosol will be generated.
Advice on safe handling: Avoid contact with skin, eyes and clothing.
Storage
Keep container tightly closed. Store in a refrigerator.
Storage conditions:
Store away from incompatible materials such as oxidizing agents.
Law is followed.
Packaging material:
Section 8. EXPOSURE CONTROLS / PERSONAL PROTECTION
Engineering controls: Install a closed system or local exhaust as possible so that workers should not be
exposed directly. Also install safety shower and eye bath.
Personal protective equipment
Respiratory protection: Vapor respirator. Follow local and national regulations.
Hand protection: Protective gloves.
Eye protection: Safety glasses. A face-shield, if the situation requires.
Skin and body protection: Protective clothing. Protective boots, if the situation requires.
Section 9. PHYSICAL AND CHEMICAL PROPERTIES
Physical state (20°C): Liquid
Form: clear
Color: Very pale yellow - Pale yellow
Odor: No data available
pH: No data available
Melting point/freezing point:No data available
Boiling Point/Range: No data available
Flash Point: No data available
Explosive limits
Lower: No data available
Upper: No data available
Density: 1.38
Solubility: No data available
Section 10. STABILITY AND REACTIVITY
Stability: Stable under proper conditions.
No special reactivity has been reported.
Reactivity:
Conditions to avoid: Heat-sensitive
2-(4-Bromobutyl)-1,3-dioxolane
Section 10. STABILITY AND REACTIVITY
Incompartible materials: oxidizing agents
Hazardous Decomposition Carbon monoxide, Carbon dioxide, Hydrogen bromide
Products:
Section 11. TOXICOLOGICAL INFORMATION
Acute Toxicity: No data available
Skin corrosion/irritation: No data available
Serious eye No data available
damage/irritation:
Germ cell mutagenicity: No data available
Carcinogenicity:
IARC = No data available
NTP = No data available
Reproductive toxicity: No data available
Section 12. ECOLOGICAL INFORMATION
Ecotoxicity:
Fish: No data available
No data available
Crustacea:
Algae: No data available
Persistence / degradability: No data available
Bioaccumulative No data available
potential(BCF):
Mobillity in soil
No data available
log Pow:
Soil adsorption (Koc): No data available
No data available
Henry's Law
constant(PaM3/mol):
Section 13. DISPOSAL CONSIDERATIONS
Recycle to process, if possible. Consult your local regional authorities. You may be able to burn in a chemical
incinerator equipped with an afterburner and scrubber system. Observe all federal, state and local regulations when
disposing of the substance.
Section 14. TRANSPORT INFORMATION
Does not correspond to the classification standard of the United Nations
Hazards Class:
UN-No: Not Listed
Section 15. REGULATORY INFORMATION
Safe management ordinance of dangerous chemical product (State Council announces on January 26,
2002): Safe use and production, the storage of a dangerous chemical, transport, loading and unloading were
prescribed.
2-(4-Bromobutyl)-1,3-dioxolane
SECTION 16 - ADDITIONAL INFORMATION
N/A
SAFETY DATA SHEET
Section 1. BASE INFORMATION
Product name: 2-(4-Bromobutyl)-1,3-dioxolane
Revision number: 1
Section 2. HAZARDS IDENTIFICATION
Classification of the GHS
PHYSICAL HAZARDS Not classified
HEALTH HAZARDS Not classified
Not classified
ENVIRONMENTAL HAZARDS
GHS label elements
None
Pictograms or hazard symbols
Signal word No signal word
None
Hazard statement
Precautionary statements None
Section 3. COMPOSITION/INFORMATION ON INGREDIENTS
Substance/mixture: Substance
Component(s): 2-(4-Bromobutyl)-1,3-dioxolane
Percent: >94.0%(GC)
CAS Number: 87227-41-6
Synonyms: 5-Bromovaleraldehyde Ethylene Acetal
Chemical Formula: C7H13BrO2
Section 4. FIRST AID MEASURES
Inhalation: Remove victim to fresh air and keep at rest in a position comfortable for breathing.
Get medical advice/attention if you feel unwell.
Skin contact: Remove/Take off immediately all contaminated clothing. Rinse skin with
water/shower. If skin irritation or rash occurs: Get medical advice/attention.
Eye contact: Rinse cautiously with water for several minutes. Remove contact lenses, if present
and easy to do. Continue rinsing. If eye irritation persists: Get medical
advice/attention.
Ingestion: Get medical advice/attention if you feel unwell. Rinse mouth.
A rescuer should wear personal protective equipment, such as rubber gloves and air-
Protection of first-aiders:
tight goggles.
Section 5. FIRE-FIGHTING MEASURES
Suitable extinguishing Dry chemical, foam, water spray, carbon dioxide.
media:
Extinguishing media not to Solid streams of water
be used:
Specific hazards: Take care as it may decompose upon combustion or in high temperatures to
generate poisonous fume.
2-(4-Bromobutyl)-1,3-dioxolane
Section 5. FIRE-FIGHTING MEASURES
Specific methods: Fire-extinguishing work is done from the windward and the suitable fire-extinguishing
method according to the surrounding situation is used. Uninvolved persons should
evacuate to a safe place. In case of fire in the surroundings: Remove movable
containers if safe to do so.
Special protective When extinguishing fire, be sure to wear personal protective equipment.
equipment for firefighters:
Section 6. ACCIDENTAL RELEASE MEASURES
Use personal protective equipment. Keep people away from and upwind of spill/leak.
Personal precautions,
protective equipment and Ensure adequate ventilation. Entry to non-involved personnel should be controlled
emergency procedures: around the leakage area by roping off, etc.
Environmental precautions: Prevent product from entering drains.
Methods and materials for Absorb spilled material in a suitable absorbent (e.g. rag, dry sand, earth, saw-dust).
containment and cleaning In case of large amount of spillage, contain a spill by bunding. Adhered or collected
up: material should be promptly disposed of, in accordance with appropriate laws and
regulations.
Section 7. HANDLING AND STORAGE
Handling
Technical measures: Handling is performed in a well ventilated place. Wear suitable protective equipment.
Prevent generation of vapor or mist. Wash hands and face thoroughly after handling.
Use a ventilation, local exhaust if vapor or aerosol will be generated.
Advice on safe handling: Avoid contact with skin, eyes and clothing.
Storage
Keep container tightly closed. Store in a refrigerator.
Storage conditions:
Store away from incompatible materials such as oxidizing agents.
Law is followed.
Packaging material:
Section 8. EXPOSURE CONTROLS / PERSONAL PROTECTION
Engineering controls: Install a closed system or local exhaust as possible so that workers should not be
exposed directly. Also install safety shower and eye bath.
Personal protective equipment
Respiratory protection: Vapor respirator. Follow local and national regulations.
Hand protection: Protective gloves.
Eye protection: Safety glasses. A face-shield, if the situation requires.
Skin and body protection: Protective clothing. Protective boots, if the situation requires.
Section 9. PHYSICAL AND CHEMICAL PROPERTIES
Physical state (20°C): Liquid
Form: clear
Color: Very pale yellow - Pale yellow
Odor: No data available
pH: No data available
Melting point/freezing point:No data available
Boiling Point/Range: No data available
Flash Point: No data available
Explosive limits
Lower: No data available
Upper: No data available
Density: 1.38
Solubility: No data available
Section 10. STABILITY AND REACTIVITY
Stability: Stable under proper conditions.
No special reactivity has been reported.
Reactivity:
Conditions to avoid: Heat-sensitive
2-(4-Bromobutyl)-1,3-dioxolane
Section 10. STABILITY AND REACTIVITY
Incompartible materials: oxidizing agents
Hazardous Decomposition Carbon monoxide, Carbon dioxide, Hydrogen bromide
Products:
Section 11. TOXICOLOGICAL INFORMATION
Acute Toxicity: No data available
Skin corrosion/irritation: No data available
Serious eye No data available
damage/irritation:
Germ cell mutagenicity: No data available
Carcinogenicity:
IARC = No data available
NTP = No data available
Reproductive toxicity: No data available
Section 12. ECOLOGICAL INFORMATION
Ecotoxicity:
Fish: No data available
No data available
Crustacea:
Algae: No data available
Persistence / degradability: No data available
Bioaccumulative No data available
potential(BCF):
Mobillity in soil
No data available
log Pow:
Soil adsorption (Koc): No data available
No data available
Henry's Law
constant(PaM3/mol):
Section 13. DISPOSAL CONSIDERATIONS
Recycle to process, if possible. Consult your local regional authorities. You may be able to burn in a chemical
incinerator equipped with an afterburner and scrubber system. Observe all federal, state and local regulations when
disposing of the substance.
Section 14. TRANSPORT INFORMATION
Does not correspond to the classification standard of the United Nations
Hazards Class:
UN-No: Not Listed
Section 15. REGULATORY INFORMATION
Safe management ordinance of dangerous chemical product (State Council announces on January 26,
2002): Safe use and production, the storage of a dangerous chemical, transport, loading and unloading were
prescribed.
2-(4-Bromobutyl)-1,3-dioxolane
SECTION 16 - ADDITIONAL INFORMATION
N/A
上下游信息
反应信息
-
作为反应物:描述:2-(4-溴丁基)-1,3-二氧戊环 在 盐酸 、 氢氧化钾 、 sodium hexamethyldisilazane 、 红铝 、 copper(I) bromide 作用下, 以 四氢呋喃 、 甲苯 、 乙腈 、 苯 为溶剂, 反应 68.67h, 生成 (Z)-氧代环十七碳-8-烯-2-酮参考文献:名称:Kumulierte Ylide XX.1Synthesen (E)-α,β-ungesättigter macrocyclischer Lactone durch intramolekulare Wittig-Olefinierung via Triphenylphosphoranylidenketen2摘要:累积脲 XX.1 通过三苯基膦亚基乙烯分子内 Wittig 烯化反应合成 (E)-δ,δ-不饱和大环内酯2 描述了通过 (Ï-氧代烷氧基)羰基亚甲基三苯基膦分子内 Wittig 反应封闭大环内酯环的两种方法。通过将适当的(游离或受保护的)Ï-羟基烷烃加入到累积的三苯基膦亚基乙烯炔中,很容易获得后者。然后举例说明了这些方法在天然产物合成中的应用。DOI:10.1055/s-1989-27271
-
作为产物:参考文献:名称:Metabolism of phencyclidine. The role of the carbinolamine intermediate in the formation of lactam and amino acid metabolites of nitrogen heterocycles摘要:The transformation of phencyclidine in a mouse liver microsome preparation to several oxidative metabolites was studied. With use of GLC and HPLC methods with internal standards, phencyclidine and six metabolites were quantitated and the amino acid 12, resulting from the alpha-oxidation of the piperidine ring, was produced in 10-50 times greater amounts than the other metabolites. While most piperidines and pyrrolidines produce an amino acid and a corresponding lactam, it was found that phencyclidine was not converted to the lactam 11.DOI:10.1021/jm00379a011
文献信息
-
COMPOUNDS HAVING MUSCARINIC RECEPTOR ANTAGONIST AND BETA2 ADRENERGIC RECEPTOR AGONIST ACTIVITY申请人:CHIESI FARMACEUTICI S.p.A.公开号:US20160235734A1公开(公告)日:2016-08-18Compounds of formula I, defined herein, act both as muscarinic receptor antagonists and beta2 adrenergic receptor agonists and are useful for treating broncho-obstructive and inflammatory diseases.具有本公式I定义的化合物既作为毒蕈碱受体拮抗剂又作为β2肾上腺素能受体激动剂,并且用于治疗支气管阻塞和炎症性疾病。
-
Reductions, Reductive Alkylations, and Intramolecular Cyclizations of Acyl Silanes with Samarium Diiodide or Tributyltin Hydride作者:Tsung-Hsun Chuang、Jim-Min Fang、Weir-Torn Jiaang、Yeun-Min TsaiDOI:10.1021/jo951980m日期:1996.1.1bearing other substituents such as a bromine atom and alkenyl, succinimide, and carbonyl groups, were prepared, and their reactions with samarium diiodide or tributylstannane were studied. The reactions of acyl silanes occurred in various manners such as reductions, reductive alkylations, intramolecular radical cyclizations, pinacol couplings, aldol reactions, and Tishchenko reactions, depending on the nature制备了包括脂肪族,芳香族和双酰基硅烷在内的一系列酰基硅烷,以及带有其他取代基(如溴原子和烯基,琥珀酰亚胺和羰基)的酰基硅烷,并使其与二碘化sa反应或三丁基锡烷进行了研究。取决于基材的性质和反应条件,酰基硅烷的反应以各种方式发生,例如还原,还原烷基化,分子内自由基环化,频哪醇偶联,醛醇缩合反应和季申科反应。通常将酰基硅烷还原而得到相应的α-甲硅烷基醇,而不转移甲硅烷基。实现了5-己烯丙基硅烷和1-silyl-1,5-pentanedione的分子内自由基环化反应,得到α-甲硅烷基环戊醇和1,2-环戊二醇衍生物,分别。在四氢呋喃中用二碘化sa处理时,在叔丁醇存在下,1-(三甲基甲硅烷基)-1,6-己二酮进行了频哪醇偶联反应,而在甲醇存在下进行了Tishchenko反应。1-甲硅烷基-1,5-戊二酮的Tishchenko反应得到δ-甲硅烷基-δ-内酯。用二碘化treating处理后,分别进行1-(三甲基甲硅烷基)-1
-
[EN] COMPOUNDS AND USES THEREOF<br/>[FR] COMPOSÉS ET LEURS UTILISATIONS申请人:FOGHORN THERAPEUTICS INC公开号:WO2021207291A1公开(公告)日:2021-10-14The present disclosure features compounds useful for the treatment of BAF complex-related disorders.本公开内容涉及用于治疗BAF复合物相关疾病的化合物。
-
Synthesis of Cp*CH2PPh2 and its use as a ligand for the nickel-catalysed cross-coupling reaction of alkyl halides with aryl Grignard reagents作者:Minoru Uemura、Hideki Yorimitsu、Koichiro OshimaDOI:10.1039/b612173j日期:——A new ligand, Cp*CH2PPh2 (Cp* = 1,2,3,4,5-pentamethyl-2,4-cyclopentadienyl), was prepared, and was used as a ligand for nickel-catalysed cross-coupling reaction of alkyl halides with aryl Grignard reagents, which nickel-phosphine complexes had never made possible.
-
Merging Halogen-Atom Transfer (XAT) and Cobalt Catalysis to Override E2-Selectivity in the Elimination of Alkyl Halides: A Mild Route toward <i>contra</i>-Thermodynamic Olefins作者:Huaibo Zhao、Alastair J. McMillan、Timothée Constantin、Rory C. Mykura、Fabio Juliá、Daniele LeonoriDOI:10.1021/jacs.1c06768日期:2021.9.15mechanistically distinct tactic to carry E2-type eliminations on alkyl halides. This strategy exploits the interplay of α-aminoalkyl radical-mediated halogen-atom transfer (XAT) with desaturative cobalt catalysis. The methodology is high-yielding, tolerates many functionalities, and was used to access industrially relevant materials. In contrast to thermal E2 eliminations where unsymmetrical substrates give regioisomeric
表征谱图
-
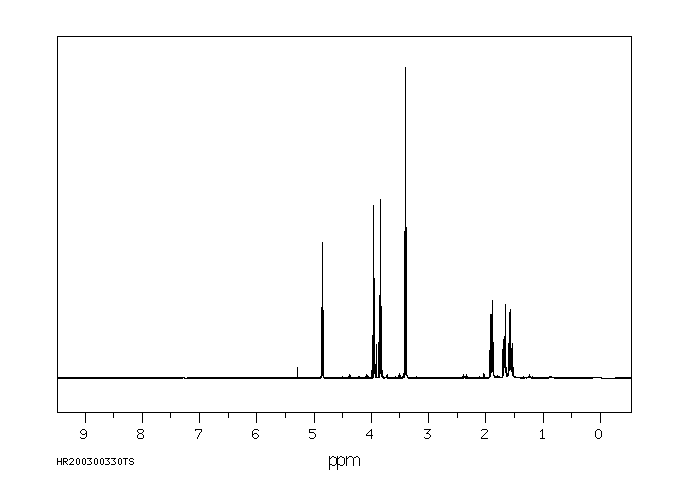
氢谱1HNMR
-
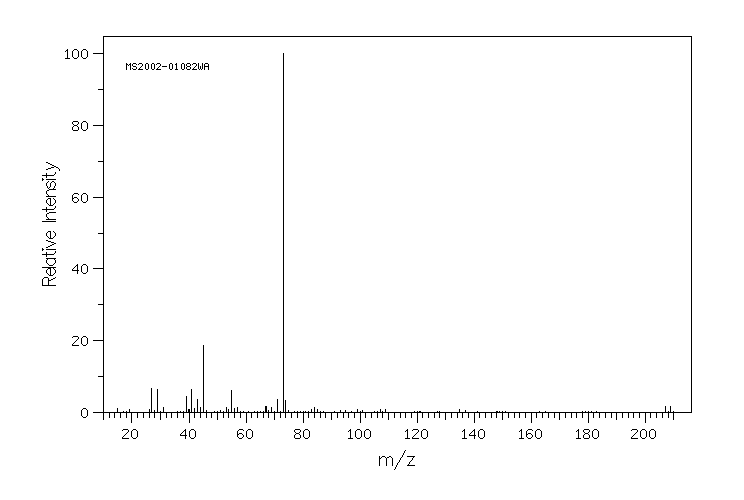
质谱MS
-
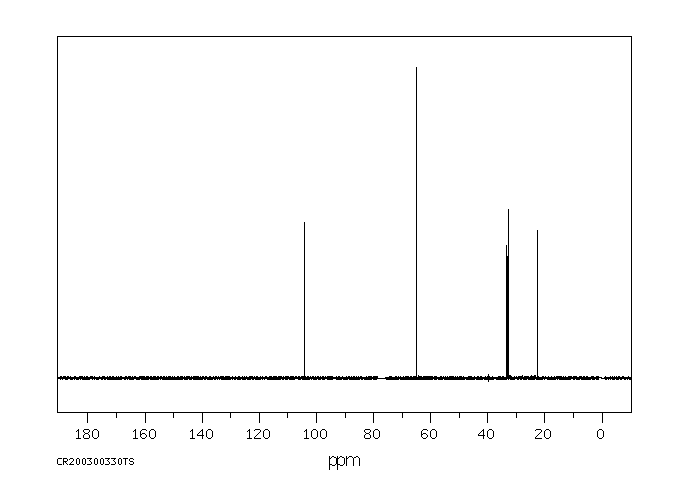
碳谱13CNMR
-
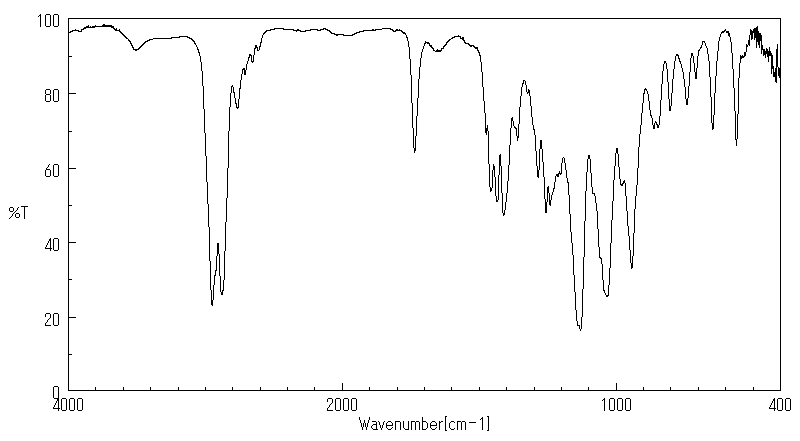
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
顺式-2-甲基-4-叔-丁基-1,3-二氧戊环
过氧竹红菌素
辛醛丙二醇缩醛
碘丙甘油
甜瓜醛丙二醇缩醛
甘油缩甲醛
甘油缩甲醛
环辛基甲醛乙烯缩醛
环戊二烯内过氧化物
环己丙胺,1-(1,3-二噁戊环-2-基)-
环丙羧酸,2-乙酰基-,甲基酯,(1R-顺)-(9CI)
氯乙醛缩乙二醇
柠檬醛乙二醇缩醛
异戊醛丙二醇缩醛
异丁醛-丙二醇缩醛
奥普碘铵
多米奥醇
多效缩醛
壬醛丙二醇缩醛
四吖戊啶,5-(1-吡咯烷基)-
亲和素
二氰苯乙烯酮乙烯缩醛
乙酮,1-(2-环辛烯-1-基)-,(-)-(9CI)
乙基1,3-二氧戊环-4-羧酸酯
丙炔醛乙二醇缩醛
三甲基-[(2-甲基-1,3-二氧戊环-4-基)甲基]铵碘化物
三氟乙烯臭氧化物
三丁基(1,3-二恶烷-2-基甲基)溴化鏻
[2-(2-碘乙基)-1,3-二氧戊环-4-基]甲醇
6,8-二氧杂二螺[2.1.4.2]十一烷
6,7-二氧杂双环[3.2.1]辛-2-烯-8-羧酸
5H,8H-呋喃并[3,4:1,5]环戊二烯并[1,2-d]-1,3-二噁唑(9CI)
5-过氧化氢基-5-甲基-1,2-二恶烷-3-酮
5-嘧啶羧酸,4-(2-呋喃基)-1,2,3,4-四氢-6-甲基-2-羰基-,1-甲基乙基酯
5-(哌嗪-1-基)苯并呋喃-2-甲酰胺
5-(1,3-二氧杂烷-2-基)呋喃-2-磺酰氯
5-(1,3-二氧戊环-2-基)戊腈
5,5-二羟基戊醛
4a-乙基-2,4a,5,6,7,7a-六氢-4-(3-羟基苯基)-1-甲基-1H-1-吡喃并英并啶
4-硝基-4-丙基辛醛乙烯缩醛
4-甲基-4-硝基辛醛乙烯缩醛
4-甲基-2-戊基-1,3-二氧戊环
4-甲基-2-十一烷基-1,3-二氧戊环
4-甲基-2-[(1E)-1-戊烯-1-基]-1,3-二氧戊环
4-甲基-2-(三氯甲基)-1,3-二氧戊环
4-甲基-2-(2-(甲硫基)乙基)-1,3-二氧戊环
4-甲基-2-(1-丙烯基)-1,3-二氧戊环
4-甲基-1,3-二氧戊环
4-烯丙基-4-甲基-2-乙烯基-1,3-二氧戊环
4-溴-3,5,5-三甲基二氧戊环-3-醇