2-溴-3-氨基-5-氯吡啶 | 90902-83-3
中文名称
2-溴-3-氨基-5-氯吡啶
中文别名
3-氨基-2-溴-5-氯吡啶;2-溴-5-氯-3-氨基吡啶
英文名称
2-bromo-5-chloropyridin-3-amine
英文别名
3-amino-2-bromo-5-chloropyridine;2-bromo-3-amino-5-chloropyridine
CAS
90902-83-3
化学式
C5H4BrClN2
mdl
MFCD07368976
分子量
207.457
InChiKey
AZOXQBMBDRLSCQ-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:141-143°C
-
沸点:312.7±37.0 °C(Predicted)
-
密度:1.834±0.06 g/cm3(Predicted)
-
最大波长(λmax):314nm(EtOH)(lit.)
计算性质
-
辛醇/水分配系数(LogP):1.8
-
重原子数:9
-
可旋转键数:0
-
环数:1.0
-
sp3杂化的碳原子比例:0.0
-
拓扑面积:38.9
-
氢给体数:1
-
氢受体数:2
安全信息
-
危险品标志:Xi
-
安全说明:S26,S39
-
危险类别码:R22,R37/38,R41
-
海关编码:2933399090
-
危险性防范说明:P261,P305+P351+P338
-
危险性描述:H315,H319,H335
-
储存条件:2-8°C
SDS
Material Safety Data Sheet
Section 1. Identification of the substance
3-Amino-2-bromo-5-chloropyridine
Product Name:
Synonyms:
Section 2. Hazards identification
Harmful by inhalation, in contact with skin, and if swallowed.
H301: Toxic if swallowed
H315: Causes skin irritation
H318: Causes serious eye damage
H335: May cause respiratory irritation
Avoid breathing dust/fume/gas/mist/vapours/spray
P261:
P280: Wear protective gloves/protective clothing/eye protection/face protection
P301+P310: IF SWALLOWED: Immediately call a POISON CENTER or doctor/physician
P305+P351+P338: IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses if present
and easy to do – continue rinsing
Section 3. Composition/information on ingredients.
3-Amino-2-bromo-5-chloropyridine
Ingredient name:
CAS number: 90902-83-3
Section 4. First aid measures
Immediately wash skin with copious amounts of water for at least 15 minutes while removing
Skin contact:
contaminated clothing and shoes. If irritation persists, seek medical attention.
Eye contact: Immediately wash skin with copious amounts of water for at least 15 minutes. Assure adequate
flushing of the eyes by separating the eyelids with fingers. If irritation persists, seek medical
attention.
Inhalation: Remove to fresh air. In severe cases or if symptoms persist, seek medical attention.
Wash out mouth with copious amounts of water for at least 15 minutes. Seek medical attention.
Ingestion:
Section 5. Fire fighting measures
In the event of a fire involving this material, alone or in combination with other materials, use dry
powder or carbon dioxide extinguishers. Protective clothing and self-contained breathing apparatus
should be worn.
Section 6. Accidental release measures
Personal precautions: Wear suitable personal protective equipment which performs satisfactorily and meets local/state/national
standards.
Respiratory precaution: Wear approved mask/respirator
Hand precaution: Wear suitable gloves/gauntlets
Skin protection: Wear suitable protective clothing
Eye protection: Wear suitable eye protection
Methods for cleaning up: Mix with sand or similar inert absorbent material, sweep up and keep in a tightly closed container
for disposal. See section 12.
Environmental precautions: Do not allow material to enter drains or water courses.
Section 7. Handling and storage
Handling: This product should be handled only by, or under the close supervision of, those properly qualified
in the handling and use of potentially hazardous chemicals, who should take into account the fire,
health and chemical hazard data given on this sheet.
Storage: Store in closed vessels.
Section 8. Exposure Controls / Personal protection
Engineering Controls: Use only in a chemical fume hood.
Personal protective equipment: Wear laboratory clothing, chemical-resistant gloves and safety goggles.
General hydiene measures: Wash thoroughly after handling. Wash contaminated clothing before reuse.
Section 9. Physical and chemical properties
Not specified
Appearance:
Boiling point: No data
Melting point: No data
Flash point: No data
Density: No data
Molecular formula: C5H4BrClN2
Molecular weight: 207.5
Section 10. Stability and reactivity
Conditions to avoid: Heat, flames and sparks.
Materials to avoid: Oxidizing agents.
Possible hazardous combustion products: Carbon monoxide, nitrogen oxides, hydrogen chloride, hydrogen bromide.
Section 11. Toxicological information
No data.
Section 12. Ecological information
No data.
Section 13. Disposal consideration
Arrange disposal as special waste, by licensed disposal company, in consultation with local waste
disposal authority, in accordance with national and regional regulations.
Section 14. Transportation information
UN Number: UN2811 Class: 6.1 Packing group: III
Proper shipping name: TOXIC SOLIDS, ORGANIC, N.O.S. (3-Amino-2-bromo-5-chloropyridine)
Section 15. Regulatory information
No chemicals in this material are subject to the reporting requirements of SARA Title III, Section
302, or have known CAS numbers that exceed the threshold reporting levels established by SARA
Title III, Section 313.
SECTION 16 - ADDITIONAL INFORMATION
N/A
Section 1. Identification of the substance
3-Amino-2-bromo-5-chloropyridine
Product Name:
Synonyms:
Section 2. Hazards identification
Harmful by inhalation, in contact with skin, and if swallowed.
H301: Toxic if swallowed
H315: Causes skin irritation
H318: Causes serious eye damage
H335: May cause respiratory irritation
Avoid breathing dust/fume/gas/mist/vapours/spray
P261:
P280: Wear protective gloves/protective clothing/eye protection/face protection
P301+P310: IF SWALLOWED: Immediately call a POISON CENTER or doctor/physician
P305+P351+P338: IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses if present
and easy to do – continue rinsing
Section 3. Composition/information on ingredients.
3-Amino-2-bromo-5-chloropyridine
Ingredient name:
CAS number: 90902-83-3
Section 4. First aid measures
Immediately wash skin with copious amounts of water for at least 15 minutes while removing
Skin contact:
contaminated clothing and shoes. If irritation persists, seek medical attention.
Eye contact: Immediately wash skin with copious amounts of water for at least 15 minutes. Assure adequate
flushing of the eyes by separating the eyelids with fingers. If irritation persists, seek medical
attention.
Inhalation: Remove to fresh air. In severe cases or if symptoms persist, seek medical attention.
Wash out mouth with copious amounts of water for at least 15 minutes. Seek medical attention.
Ingestion:
Section 5. Fire fighting measures
In the event of a fire involving this material, alone or in combination with other materials, use dry
powder or carbon dioxide extinguishers. Protective clothing and self-contained breathing apparatus
should be worn.
Section 6. Accidental release measures
Personal precautions: Wear suitable personal protective equipment which performs satisfactorily and meets local/state/national
standards.
Respiratory precaution: Wear approved mask/respirator
Hand precaution: Wear suitable gloves/gauntlets
Skin protection: Wear suitable protective clothing
Eye protection: Wear suitable eye protection
Methods for cleaning up: Mix with sand or similar inert absorbent material, sweep up and keep in a tightly closed container
for disposal. See section 12.
Environmental precautions: Do not allow material to enter drains or water courses.
Section 7. Handling and storage
Handling: This product should be handled only by, or under the close supervision of, those properly qualified
in the handling and use of potentially hazardous chemicals, who should take into account the fire,
health and chemical hazard data given on this sheet.
Storage: Store in closed vessels.
Section 8. Exposure Controls / Personal protection
Engineering Controls: Use only in a chemical fume hood.
Personal protective equipment: Wear laboratory clothing, chemical-resistant gloves and safety goggles.
General hydiene measures: Wash thoroughly after handling. Wash contaminated clothing before reuse.
Section 9. Physical and chemical properties
Not specified
Appearance:
Boiling point: No data
Melting point: No data
Flash point: No data
Density: No data
Molecular formula: C5H4BrClN2
Molecular weight: 207.5
Section 10. Stability and reactivity
Conditions to avoid: Heat, flames and sparks.
Materials to avoid: Oxidizing agents.
Possible hazardous combustion products: Carbon monoxide, nitrogen oxides, hydrogen chloride, hydrogen bromide.
Section 11. Toxicological information
No data.
Section 12. Ecological information
No data.
Section 13. Disposal consideration
Arrange disposal as special waste, by licensed disposal company, in consultation with local waste
disposal authority, in accordance with national and regional regulations.
Section 14. Transportation information
UN Number: UN2811 Class: 6.1 Packing group: III
Proper shipping name: TOXIC SOLIDS, ORGANIC, N.O.S. (3-Amino-2-bromo-5-chloropyridine)
Section 15. Regulatory information
No chemicals in this material are subject to the reporting requirements of SARA Title III, Section
302, or have known CAS numbers that exceed the threshold reporting levels established by SARA
Title III, Section 313.
SECTION 16 - ADDITIONAL INFORMATION
N/A
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 5-氯-2-溴-3-硝基吡啶 2-bromo-5-chloro-3-nitropyridine 75806-86-9 C5H2BrClN2O2 237.44 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 —— N-(2-bromo-5-chloropyridin-3-yl)-2-chloroacetamide 1382722-12-4 C7H5BrCl2N2O 283.939
反应信息
-
作为反应物:描述:2-溴-3-氨基-5-氯吡啶 在 bis-triphenylphosphine-palladium(II) chloride 、 copper(l) iodide 、 sodium hydride 、 三乙胺 作用下, 以 四氢呋喃 、 N,N-二甲基甲酰胺 、 mineral oil 为溶剂, 反应 10.0h, 生成 6-氯-1H-吡咯并[3,2-b]吡啶参考文献:名称:[EN] FUSED BICYCLIC COMPOUNDS FOR THE TREATMENT OF DISEASE
[FR] COMPOSÉS BICYCLIQUES FUSIONNÉS POUR LE TRAITEMENT DE MALADIES摘要:本文描述了融合双环化合物、组合物以及它们用于治疗疾病的方法。公开号:WO2016103037A1 -
作为产物:描述:参考文献:名称:NaI/PPh3 介导的硝基芳烃的光化学还原和胺化摘要:发现基于 NaI 和 PPh 3组合的温和无过渡金属和无光敏剂的光氧化还原系统能够高度选择性地还原硝基芳烃。该协议允许广泛的可还原官能团,如卤素(Cl、Br 甚至 I)、醛、酮、羧基和氰基。此外,当使用邻硝基双芳烃时,NaI 和化学计量 PPh 3的光氧化还原催化也提供了 Cadogan 型还原胺化的替代入口。DOI:10.1021/acs.orglett.1c01654
文献信息
-
[EN] 3-SUBSTITUTED 2-AMINO-INDOLE DERIVATIVES<br/>[FR] DÉRIVÉS DE 2-AMINO-INDOLE 3-SUBSTITUÉS申请人:TAKEDA PHARMACEUTICAL公开号:WO2015198045A1公开(公告)日:2015-12-30The present invention provides compounds of formula (I) (Formula (I)) and pharmaceutically acceptable salts thereof, wherein Q, X% X4,X5 X6, X7,R1, R2, R3 and R8 are as defined in the specification, processes for the preparation of such compounds, pharmaceutical compositions containing them and the use of such compounds in therapy.本发明提供了公式(I)(公式(I))的化合物及其药用可接受的盐,其中Q,X,X4,X5,X6,X7,R1,R2,R3和R8如说明书中所定义,这些化合物的制备方法,包含它们的药物组合物以及这些化合物在治疗中的用途。
-
HETEROARYLAZOLE COMPOUND AND PEST CONTROL AGENT申请人:Nippon Soda Co., Ltd.公开号:US20210380556A1公开(公告)日:2021-12-09An object of the present invention is to provide a heteroaryl azole compound that is excellent in pest control activity, particularly, insecticidal activity and/or miticidal activity, is excellent in safety, and may be industrially advantageously synthesized. The compound of the present invention is a compound represented by the formula (I), an N-oxide compound, stereoisomer, tautomer or hydrate thereof or a salt of any of these compounds. In the formula (I), A represents CH or a nitrogen atom; B 1 represents CX 1 or a nitrogen atom; X 1 , X 2 and X 3 each independently represent a hydrogen atom, a substituted or unsubstituted C1-6 alkyl group, a substituted or unsubstituted C2-6 alkenyl group, or the like; R 1 represents a substituted or unsubstituted C1-6 alkylthio group, a substituted or unsubstituted C1-6 alkylsulfinyl group, or the like; R 2 represents a substituted or unsubstituted C1-6 alkyl group; and R represents a substituted or unsubstituted C1-6 alkyl group.本发明的一个目的是提供一种在害虫控制活性方面表现出色,特别是在杀虫活性和/或杀螨活性方面表现出色,安全性优秀,并且可以在工业上有利地合成的杂环芳醇类化合物。本发明的化合物是由式(I)表示的化合物,其N-氧化物化合物,立体异构体,互变异构体或其水合物,或这些化合物的任何盐。在式(I)中,A代表CH或氮原子;B1代表CX1或氮原子;X1,X2和X3分别独立地代表氢原子,取代或未取代的C1-6烷基基团,取代或未取代的C2-6烯基基团,或类似物;R1代表取代或未取代的C1-6烷基硫基团,取代或未取代的C1-6烷基亚硫氧基团,或类似物;R2代表取代或未取代的C1-6烷基基团;R代表取代或未取代的C1-6烷基基团。
-
KV3 ENHANCERS FOR THE TREATMENT OF COGNITIVE DISORDERS申请人:F. Hoffmann-La Roche AG公开号:EP3901152A1公开(公告)日:2021-10-27The invention provides new heterocyclic compounds having the general formula (I) wherein A, B, L, Y1 to Y4 and R1 to R6 are as described herein.这项发明提供了具有一般式(I)的新杂环化合物 其中A、B、L、Y1至Y4和R1至R6如本文所述。
-
[EN] MACROCYCLES WITH HETROCYCLIC P2' GROUPS AS FACTOR XIA INHIBITORS<br/>[FR] MACROCYCLES À GROUPES HÉTÉROCYCLIQUES P2' SERVANT D'INHIBITEURS DU FACTEUR XIA申请人:BRISTOL MYERS SQUIBB CO公开号:WO2015116886A1公开(公告)日:2015-08-06The present invention provides compounds of Formula (I): or stereoisomers, tautomers, or pharmaceutically acceptable salts thereof, wherein all the variables are as defined herein. These compounds are selective factor XIa inhibitors or dual inhibitors of FXIa and plasma kallikrein. This invention also relates to pharmaceutical compositions comprising these compounds and methods of treating thromboembolic and/or inflammatory disorders using the same.
-
HETEROARYLS AND USES THEREOF申请人:MILLENNIUM PHARMACEUTICALS, INC.公开号:US20150225422A1公开(公告)日:2015-08-13The present invention provides a compound of formula I: and pharmaceutically acceptable salts thereof, wherein X, R 1 , R 2 , R 3 , R 4 , R 5 , L 1 , L 2 , m, and n, are as described in the specification. Such compounds are inhibitors of VPS34 and thus useful for treating proliferative, inflammatory, or cardiovascular disorders.
表征谱图
-
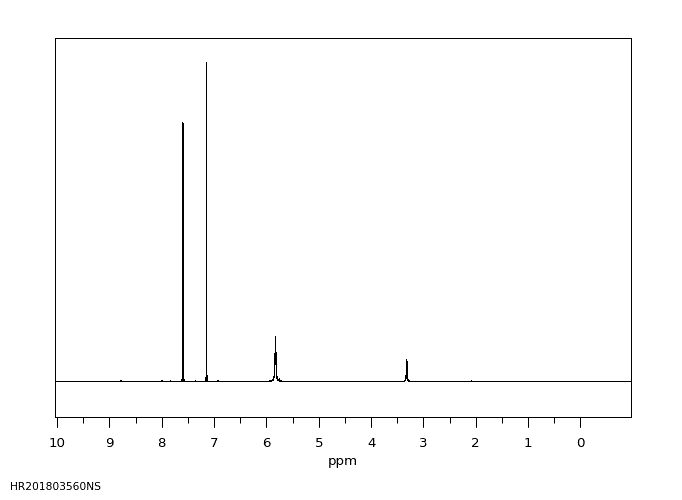
氢谱1HNMR
-
质谱MS
-
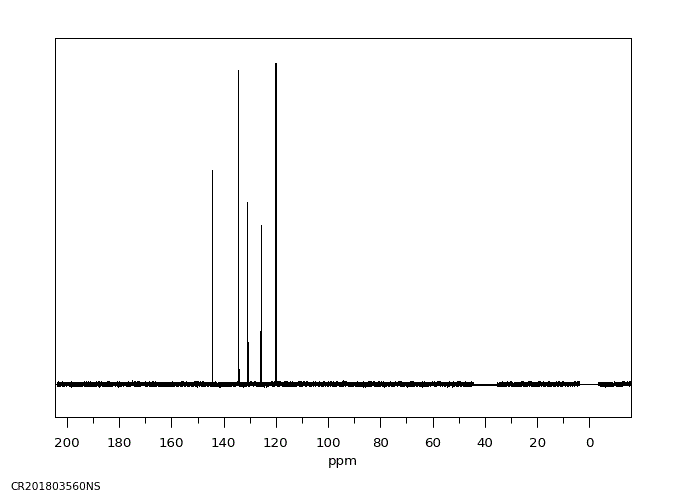
碳谱13CNMR
-
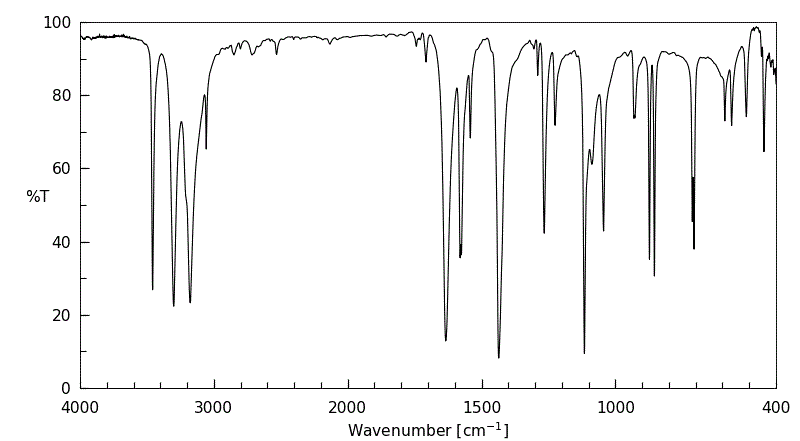
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(S)-氨氯地平-d4
(R,S)-可替宁N-氧化物-甲基-d3
(R)-(+)-2,2'',6,6''-四甲氧基-4,4''-双(二苯基膦基)-3,3''-联吡啶(1,5-环辛二烯)铑(I)四氟硼酸盐
(R)-N'-亚硝基尼古丁
(R)-DRF053二盐酸盐
(5E)-5-[(2,5-二甲基-1-吡啶-3-基-吡咯-3-基)亚甲基]-2-亚磺酰基-1,3-噻唑烷-4-酮
(5-溴-3-吡啶基)[4-(1-吡咯烷基)-1-哌啶基]甲酮
(5-氨基-6-氰基-7-甲基[1,2]噻唑并[4,5-b]吡啶-3-甲酰胺)
(2S,2'S)-(-)-[N,N'-双(2-吡啶基甲基]-2,2'-联吡咯烷双(乙腈)铁(II)六氟锑酸盐
(2S)-2-[[[9-丙-2-基-6-[(4-吡啶-2-基苯基)甲基氨基]嘌呤-2-基]氨基]丁-1-醇
(2R,2''R)-(+)-[N,N''-双(2-吡啶基甲基)]-2,2''-联吡咯烷四盐酸盐
(1'R,2'S)-尼古丁1,1'-Di-N-氧化物
黄色素-37
麦斯明-D4
麦司明
麝香吡啶
鲁非罗尼
鲁卡他胺
高氯酸N-甲基甲基吡啶正离子
高氯酸,吡啶
高奎宁酸
马来酸溴苯那敏
马来酸氯苯那敏-D6
马来酸左氨氯地平
顺式-双(异硫氰基)(2,2'-联吡啶基-4,4'-二羧基)(4,4'-二-壬基-2'-联吡啶基)钌(II)
顺式-二氯二(4-氯吡啶)铂
顺式-二(2,2'-联吡啶)二氯铬氯化物
顺式-1-(4-甲氧基苄基)-3-羟基-5-(3-吡啶)-2-吡咯烷酮
顺-双(2,2-二吡啶)二氯化钌(II) 水合物
顺-双(2,2'-二吡啶基)二氯化钌(II)二水合物
顺-二氯二(吡啶)铂(II)
顺-二(2,2'-联吡啶)二氯化钌(II)二水合物
韦德伊斯试剂
非那吡啶
非洛地平杂质C
非洛地平
非戈替尼
非布索坦杂质66
非尼拉朵
非尼拉敏
雷索替丁
阿雷地平
阿瑞洛莫
阿扎那韦中间体
阿培利司N-6
阿伐曲波帕杂质40
间硝苯地平
间-硝苯地平
镉,二碘四(4-甲基吡啶)-
锌,二溴二[4-吡啶羧硫代酸(2-吡啶基亚甲基)酰肼]-