2-环己基氨基-5-硝基吡啶 | 25948-14-5
分子结构分类
中文名称
2-环己基氨基-5-硝基吡啶
中文别名
3-溴-2,4-二氧代哌啶-1-羧酸叔丁基酯
英文名称
N-(5-nitro-2-pyridyl)cyclohexylamine
英文别名
cyclohexyl-(5-nitro-pyridin-2-yl)-amine;2-(Cyclohexylamino)-5-nitro-pyridine;2-Cyclohexylamino-5-nitropyridine;N-cyclohexyl-5-nitropyridin-2-amine
CAS
25948-14-5
化学式
C11H15N3O2
mdl
MFCD00023640
分子量
221.259
InChiKey
OGOQAAXBFKYJBE-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
计算性质
-
辛醇/水分配系数(LogP):2.8
-
重原子数:16
-
可旋转键数:2
-
环数:2.0
-
sp3杂化的碳原子比例:0.545
-
拓扑面积:70.7
-
氢给体数:1
-
氢受体数:4
安全信息
-
海关编码:2933399090
反应信息
-
作为反应物:描述:2-环己基氨基-5-硝基吡啶 、 碘甲烷 以 N-甲基乙酰胺 、 mineral oil 为溶剂, 生成 2-(N-Cyclohexyl-N-methylamino)-5-nitro-pyridine参考文献:名称:Substituted pyridine compounds and methods of use摘要:选定的新型取代吡啶化合物对于预防和治疗疾病,如TNF-α、IL-1β、IL-6和/或IL-8介导的疾病,以及其他疾病,如疼痛和糖尿病,具有有效性。该发明涵盖了新型化合物、类似物、前药和其药学上可接受的盐,用于预防和治疗涉及炎症、疼痛、糖尿病、癌症等疾病和其他疾病或病况的药物组合物和方法。该发明还涉及制备此类化合物的方法,以及在此类方法中有用的中间体。公开号:US06022884A1
-
作为产物:描述:参考文献:名称:取代硝基吡啶和硝基苯的 1H 和 13C NMR 研究摘要:已经测定了 3-硝基-、5-硝基-和 3,5-二硝基-2-甲氧基吡啶的 1H 和 13C NMR 谱。结果显示了 2-甲氧基优选的顺式构象,以及该基团的氧原子与阻碍 3-硝基取代衍生物共轭的邻硝基氧原子之间空间排斥的重要性。用 2-N-丁胺-、2-N-环己胺-和 2-(N-哌啶基)-取代的硝基吡啶也可以观察到类似的共振空间抑制,其 1H 和 13C NMR 光谱也被测定。在仲胺的情况下,可以清楚地检测到氨基质子和 3-硝基之间的氢键。2,6-二硝基-、2,4-二硝基-和2,4,6-三硝基-2-R-苯(R = OCH3、NHC4H9、NH-环-C6H11、NC5H10) 也被记录并与吡啶衍生物的那些进行比较。如果硝基的共轭不受阻碍,则电子氮杂和硝基效应是可比的。DOI:10.1002/mrc.1260240607
文献信息
-
Amination of Heteroaryl Chlorides: Palladium Catalysis or S<sub>N</sub>Ar in Green Solvents?作者:Katie Walsh、Helen F. Sneddon、Christopher J. MoodyDOI:10.1002/cssc.201300239日期:2013.8reaction of heteroaryl chlorides in the pyrimidine, pyrazine and quinazoline series with amines in water in the presence of KF results in a facile SNAr reaction and N‐arylation. The reaction is less satisfactory with pyridines unless an additional electron‐withdrawing group is present. The results showed that the transition‐metal‐free SNAr reaction not only compares favourably to palladium‐catalysed
-
Mittel zum Färben von Haaren申请人:RÜTGERSWERKE AKTIENGESELLSCHAFT公开号:EP0137524A1公开(公告)日:1985-04-17Die Erfindung beschreibt neue Kuppler- oder Nuanceurkomponenten, die in Kombination mit einschlägigen Entwicklern zum Färben von Haaren verwendet werden. Diese Kupplerkomponenten sind Nitropyridinderivate der allgemeinen Formel wobei die Substituenten X, Y, R, und R2 die in den Ansprüchen gegebenen Bedeutungen haben.本发明描述了与相关显影剂结合使用的新型耦合剂或微染剂成分,用于染发。这些耦合剂成分是通式如下的硝基吡啶衍生物 其中取代基 X、Y、R 和 R2 的含义见权利要求书。
-
US06184237B2申请人:——公开号:——公开(公告)日:——
-
SUBSTITUTED PYRIDINE COMPOUNDS AS ANTI-INFLAMMATORY AGENTS申请人:AMGEN INC.公开号:EP1028945B9公开(公告)日:2007-11-28
-
THERAPEUTIC QUINAZOLINE DERIVATIVES申请人:AstraZeneca AB公开号:EP1218355A1公开(公告)日:2002-07-03
表征谱图
-
氢谱1HNMR
-
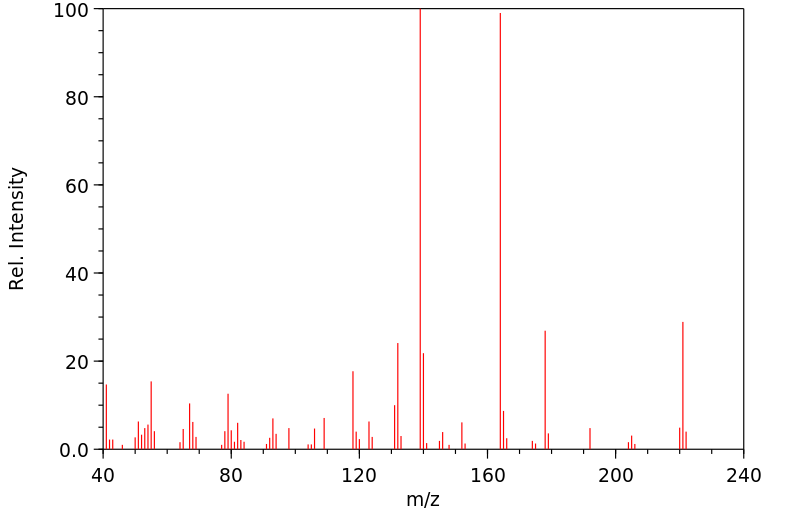
质谱MS
-
碳谱13CNMR
-
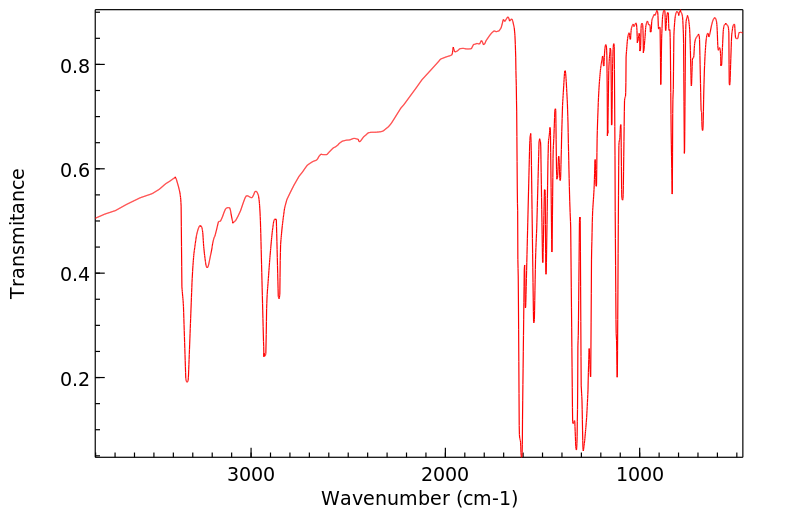
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
顺式-2-硝基环己基乙酸酯
顺式-2-硝基-6-甲基环己酮
雷尼替丁杂质18
铝硝基甲烷三氯化物
钾离子载体III
重氮(硝基)甲烷
醛基-七聚乙二醇-叠氮
过氧亚甲基
辛腈,4-氟-4-硝基-7-羰基-
辛烷,1,2-二氯-1-硝基-
赤霉素A4+7(GA4:GA7=65:35)
苄哒唑
羟胺-四聚乙二醇-叠氮
羟胺-三乙二醇-叠氮
米索硝唑
磷酸十二醇酯
碘硝基甲烷
碘化e1,1-二甲基-4-羰基-3,5-二(3-苯基-2-亚丙烯基)哌啶正离子
硝酰胺
硝基脲银(I)复合物
硝基甲醇
硝基甲烷-d3
硝基甲烷-13C,d3
硝基甲烷-13C
硝基甲烷-(15)N
硝基甲烷
硝基甲基甲醇胺
硝基环辛烷
硝基环戊烷
硝基环戊基阴离子
硝基环庚烷
硝基环己烷锂盐
硝基环己烷钾盐
硝基环己烷
硝基环丁烷
硝基氨基甲酸
硝基新戊烷
硝基二乙醇胺
硝基乙醛缩二甲醇
硝基乙醛缩二乙醇
硝基乙腈
硝基乙烷-D5
硝基乙烷-1,1-d2
硝基乙烷
硝基乙烯
硝基丙烷
硝基丙二醛(E,E)-二肟
硝基丙二腈
硝基-(3-硝基-[4]吡啶基)-胺
硝乙醛肟