4-(苄氧基)-3-甲氧基-2-硝基苯甲酸 | 3584-32-5
中文名称
4-(苄氧基)-3-甲氧基-2-硝基苯甲酸
中文别名
4-苄氧基-3-甲氧基-2-硝基苯甲酸
英文名称
4-(benzyloxy)-3-methoxy-2-nitrobenzoic acid
英文别名
2-Nitro-3-methoxy-4-benzyloxybenzoesaeure;2-Nitro-vanillinsaeure-benzylaether;4-Benzyloxy-3-methoxy-2-nitro-benzoesaeure;3-methoxy-2-nitro-4-phenylmethoxybenzoic acid
CAS
3584-32-5
化学式
C15H13NO6
mdl
——
分子量
303.271
InChiKey
AWQSWWDWMMXFIH-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
计算性质
-
辛醇/水分配系数(LogP):2.7
-
重原子数:22
-
可旋转键数:5
-
环数:2.0
-
sp3杂化的碳原子比例:0.13
-
拓扑面积:102
-
氢给体数:1
-
氢受体数:6
安全信息
-
海关编码:2918990090
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 4-(苄氧基)-3-甲氧基-2-硝基苯甲醛 4-(benzyloxy)-3-methoxy-2-nitrobenzaldehyde 2450-27-3 C15H13NO5 287.272 4-羟基-3-甲氧基-2-硝基苯甲醛 2-nitrovanillin 2450-26-2 C8H7NO5 197.147 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 2-氨基-4-(苄氧基)-3-甲氧基苯甲酸 4-(benzyloxy)-3-methoxyanthranilic acid 23938-73-0 C15H15NO4 273.288 —— 4-(benzyloxy)-3-methoxy-N-(2,3,4,5-tetramethoxyphenyl)anthranilic acid 135082-42-7 C25H27NO8 469.491
反应信息
-
作为反应物:描述:4-(苄氧基)-3-甲氧基-2-硝基苯甲酸 在 哌啶 、 吡啶 、 copper(I) oxide 、 氯化亚砜 、 sodium dithionite 、 sodium acetate 、 铜 、 sodium hydride 、 potassium carbonate 、 三乙胺 、 三氯氧磷 作用下, 以 二乙二醇二甲醚 、 水 、 N,N-二甲基甲酰胺 、 苯 为溶剂, 反应 35.0h, 生成 6-(benzyloxy)-10-<2-(diethylamino)ethyl>-1,2,3,4,5-pentamethoxyacridin-9-one参考文献:名称:Synthesis of the acridone alkaloids, glyfoline and congeners. Structure-activity relationship studies of cytotoxic acridones摘要:Glyfoline (4, 1,6-dihydroxY-10-methyl-2,3,4,5-tetramethoxyacridin-9-one) and its congeners were synthesized for evaluation of their cytotoxicity. A detailed structure-activity relationships (SAR) of these acridone derivatives were also studied. To study the SAR of glyfoline analogues, substituent(s) at C-1 and C-6 and at the heterocyclic nitrogen of glyfoline nucleus were modified. Nitro- and amino-substituted glyfoline analogues were also synthesized to study the effects of substituent(s) (electron-withdrawing vs electron-donating) on their cytotoxicity. These compounds were synthesized via the Ullmann condensation of anthranilic acids with iodobenzenes or 2-chlorobenzoic acids with aniline-derivatives. The SAR studies showed that 1-hydroxy 9-acridones were more active than their 1-OMe derivatives against cell growth of human leukemic HL-60 cells in culture. Replacement of NMe of glyfoline with NH or N(CH2)2NEt2 resulted in either total loss or dramatic reduction of cytotoxity. Glyfoline congeners with nitro function at the A-ring were inactive, while compounds with amino substituent were shown to be cytotoxic in vitro.DOI:10.1021/jm00092a022
-
作为产物:描述:参考文献:名称:New Compounds-Some Derivatives of Benzylvanillin and Benzylvanillic Acid摘要:DOI:10.1021/ja01179a600
文献信息
-
Multisubstituted 1-hydroxy-9-acridones with anticancer activity申请人:Sloan-Kettering Institute for Cancer Research公开号:US05296602A1公开(公告)日:1994-03-22The present invention provides a compound having the structure: ##STR1## The present invention also provides a method for synthesizing a compound having the above-identified structure as well as the intermediate compounds produced according to that method. The present invention further provides a pharmaceutical composition comprising the above compounds. Lastly, the present invention provides a method of inhibiting growth of tumor cells.本发明提供了一种具有以下结构的化合物:##STR1## 本发明还提供了一种合成具有上述结构的化合物的方法,以及根据该方法产生的中间化合物。本发明还提供了包括上述化合物的药物组合物。最后,本发明提供了一种抑制肿瘤细胞生长的方法。
-
Synthesis of glyfoline, a constituent of glycosmis citrifolia (Willd.) Lindl. and a potential anticancer agent作者:Tsann-Long Su、Krzysztof Dziewiszek、Tian-Shung WuDOI:10.1016/s0040-4039(00)74267-5日期:1991.3Condensation of 4-benzyloxy-3-methoxyanthranilic acid (3) with 5-iodo-1,2,3,4-tetramethoxybenzene (4) gave 6-benzyloxy-1,2,3,4,5-pentamethoxy-9-acridone (5), which was then converted into glyfoline (1) via N-methylation, selective de-O-methylation, and debenzylation. The synthetic glyfoline (1) is identical with an authentic sample isolated from plant.4-苄氧基-3- methoxyanthranilic酸(缩合3)与5-碘-1,2,3,4-四甲氧基苯(4),得到6-苄氧基1,2,3,4,5-五甲氧基-9-吖啶酮(5),其随后通过N-甲基化,选择性脱-O-甲基化和脱苄基作用转化为乙二醛(1)。合成的草甘膦(1)与从植物中分离出的真实样品完全相同。
-
The aporphine series. Part I. Developments of the Bischler–Napieralski–Pschorr synthetic method. The synthesis of (±)-isobulbocapnine and (±)-actinodaphnine methyl ether作者:D. H. Hey、L. C. LoboDOI:10.1039/jr9540002246日期:——
-
Synthesis and Biological Evaluation of Novel Analogues of the Pan Class I Phosphatidylinositol 3-Kinase (PI3K) Inhibitor 2-(Difluoromethyl)-1-[4,6-di(4-morpholinyl)-1,3,5-triazin-2-yl]-1<i>H</i>-benzimidazole (ZSTK474)作者:Gordon W. Rewcastle、Swarna A. Gamage、Jack U. Flanagan、Raphael Frederick、William A. Denny、Bruce C. Baguley、Philip Kestell、Ripudaman Singh、Jackie D. Kendall、Elaine S. Marshall、Claire L. Lill、Woo-Jeong Lee、Sharada Kolekar、Christina M. Buchanan、Stephen M. F. Jamieson、Peter R. ShepherdDOI:10.1021/jm200688y日期:2011.10.27A structure activity relationship (SAR) study of the pan class I PI 3-kinase inhibitor 2-(difluoromethyl)-1-[4,6-di(4-morpholinyl)-1,3,5-triazin-2-yl]-1H-benzimidazole (ZSTK474) identified substitution at the 4 and 6 positions of the benzimidazole ring as having significant effects on the potency of substituted derivatives. The 6-amino-4-methoxy analogue displayed a greater than 1000-fold potency enhancement over the corresponding 6-aza-4-methoxy analogue against all three class Ia PI 3-kinase enzymes (p110 alpha, p110 beta, and p110 delta) and also displayed significant potency against two mutant forms of the p110 alpha isoform (H1047R and E545K). This compound was also evaluated in vivo against a U87MG human glioblastoma tumor xenograft model in Ragl(-/-) mice, and at a dose of 50 mg/kg given by ip injection at a qd x 10 dosing schedule it dramatically reduced cancer growth by 81% compared to untreated controls.
-
US5296602A申请人:——公开号:US5296602A公开(公告)日:1994-03-22
表征谱图
-
氢谱1HNMR
-
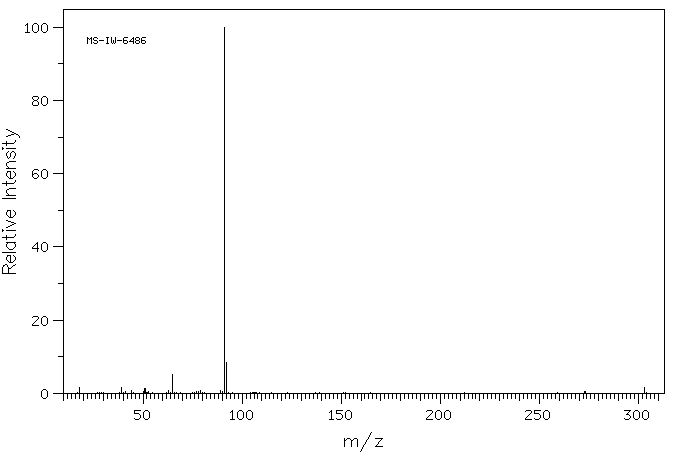
质谱MS
-
碳谱13CNMR
-
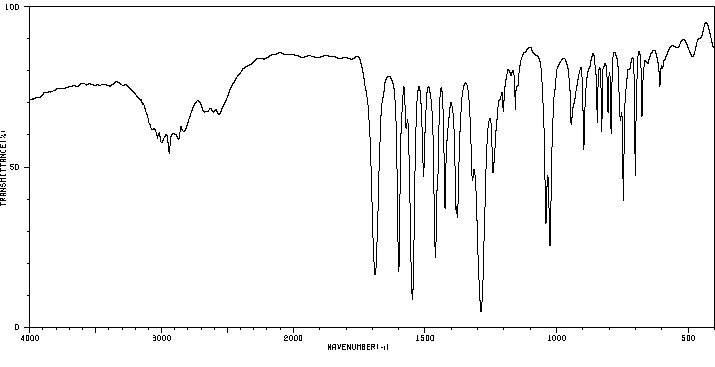
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(βS)-β-氨基-4-(4-羟基苯氧基)-3,5-二碘苯甲丙醇
(S,S)-邻甲苯基-DIPAMP
(S)-(-)-7'-〔4(S)-(苄基)恶唑-2-基]-7-二(3,5-二-叔丁基苯基)膦基-2,2',3,3'-四氢-1,1-螺二氢茚
(S)-盐酸沙丁胺醇
(S)-3-(叔丁基)-4-(2,6-二甲氧基苯基)-2,3-二氢苯并[d][1,3]氧磷杂环戊二烯
(S)-2,2'-双[双(3,5-三氟甲基苯基)膦基]-4,4',6,6'-四甲氧基联苯
(S)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(R)富马酸托特罗定
(R)-(-)-盐酸尼古地平
(R)-(-)-4,12-双(二苯基膦基)[2.2]对环芳烷(1,5环辛二烯)铑(I)四氟硼酸盐
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[((6-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(4-叔丁基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(3-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-4,7-双(3,5-二-叔丁基苯基)膦基-7“-[(吡啶-2-基甲基)氨基]-2,2”,3,3'-四氢1,1'-螺二茚满
(R)-3-(叔丁基)-4-(2,6-二苯氧基苯基)-2,3-二氢苯并[d][1,3]氧杂磷杂环戊烯
(R)-2-[((二苯基膦基)甲基]吡咯烷
(R)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(N-(4-甲氧基苯基)-N-甲基-3-(1-哌啶基)丙-2-烯酰胺)
(5-溴-2-羟基苯基)-4-氯苯甲酮
(5-溴-2-氯苯基)(4-羟基苯基)甲酮
(5-氧代-3-苯基-2,5-二氢-1,2,3,4-oxatriazol-3-鎓)
(4S,5R)-4-甲基-5-苯基-1,2,3-氧代噻唑烷-2,2-二氧化物-3-羧酸叔丁酯
(4S,4''S)-2,2''-亚环戊基双[4,5-二氢-4-(苯甲基)恶唑]
(4-溴苯基)-[2-氟-4-[6-[甲基(丙-2-烯基)氨基]己氧基]苯基]甲酮
(4-丁氧基苯甲基)三苯基溴化磷
(3aR,8aR)-(-)-4,4,8,8-四(3,5-二甲基苯基)四氢-2,2-二甲基-6-苯基-1,3-二氧戊环[4,5-e]二恶唑磷
(3aR,6aS)-5-氧代六氢环戊基[c]吡咯-2(1H)-羧酸酯
(2Z)-3-[[(4-氯苯基)氨基]-2-氰基丙烯酸乙酯
(2S,3S,5S)-5-(叔丁氧基甲酰氨基)-2-(N-5-噻唑基-甲氧羰基)氨基-1,6-二苯基-3-羟基己烷
(2S,2''S,3S,3''S)-3,3''-二叔丁基-4,4''-双(2,6-二甲氧基苯基)-2,2'',3,3''-四氢-2,2''-联苯并[d][1,3]氧杂磷杂戊环
(2S)-(-)-2-{[[[[3,5-双(氟代甲基)苯基]氨基]硫代甲基]氨基}-N-(二苯基甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[((1S,2S)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[[((1R,2R)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2-硝基苯基)磷酸三酰胺
(2,6-二氯苯基)乙酰氯
(2,3-二甲氧基-5-甲基苯基)硼酸
(1S,2S,3S,5S)-5-叠氮基-3-(苯基甲氧基)-2-[(苯基甲氧基)甲基]环戊醇
(1S,2S,3R,5R)-2-(苄氧基)甲基-6-氧杂双环[3.1.0]己-3-醇
(1-(4-氟苯基)环丙基)甲胺盐酸盐
(1-(3-溴苯基)环丁基)甲胺盐酸盐
(1-(2-氯苯基)环丁基)甲胺盐酸盐
(1-(2-氟苯基)环丙基)甲胺盐酸盐
(1-(2,6-二氟苯基)环丙基)甲胺盐酸盐
(-)-去甲基西布曲明
龙蒿油
龙胆酸钠
龙胆酸叔丁酯
龙胆酸
龙胆紫-d6
龙胆紫