1,5-癸二炔 | 53963-03-4
中文名称
1,5-癸二炔
中文别名
——
英文名称
1,5-decadiyne
英文别名
deca-1,5-diyne
CAS
53963-03-4
化学式
C10H14
mdl
MFCD00041651
分子量
134.221
InChiKey
IWIDILFDSIIOSB-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
沸点:80°C 12mm
-
密度:0.82
-
稳定性/保质期:
如果按照规格使用和储存,则不会分解,未有已知危险反应。
计算性质
-
辛醇/水分配系数(LogP):3.4
-
重原子数:10
-
可旋转键数:3
-
环数:0.0
-
sp3杂化的碳原子比例:0.6
-
拓扑面积:0
-
氢给体数:0
-
氢受体数:0
安全信息
-
危险等级:3
-
安全说明:S26,S36/37/39
-
危险类别码:R36/37/38
-
海关编码:2901299090
-
储存条件:请将贮藏器密封,并存放在阴凉、干燥处。同时,确保工作环境有良好的通风或排气设施。
SDS
1,5-癸二炔 修改号码:2
模块 1. 化学品
产品名称: 1,5-Decadiyne
修改号码: 2
模块 2. 危险性概述
GHS分类
物理性危害
易燃液体 第3级
健康危害 未分类
环境危害 未分类
GHS标签元素
图标或危害标志
信号词 警告
危险描述 易燃液体和蒸气
防范说明
[预防] 远离热源/火花/明火/热表面。禁烟。
保持容器密闭。
使用防爆的电气/通风/照明设备。采取预防措施以防静电和火花引起的着火。
穿戴防护手套/护目镜/防护面具。
[急救措施] 皮肤接触:立即去除/脱掉所有被污染的衣物。用水清洗皮肤/淋浴。
[储存] 存放于通风良好处。保持凉爽。
[废弃处置] 根据当地政府规定把物品/容器交与工业废弃处理机构。
模块 3. 成分/组成信息
单一物质/混和物 单一物质
化学名(中文名): 1,5-癸二炔
百分比: >97.0%(GC)
CAS编码: 53963-03-4
分子式: C10H14
模块 4. 急救措施
吸入: 将受害者移到新鲜空气处,保持呼吸通畅,休息。若感不适请求医/就诊。
1,5-癸二炔 修改号码:2
模块 4. 急救措施
皮肤接触: 立即去除/脱掉所有被污染的衣物。用水清洗皮肤/淋浴。
若皮肤刺激或发生皮疹:求医/就诊。
眼睛接触: 用水小心清洗几分钟。如果方便,易操作,摘除隐形眼镜。继续清洗。
如果眼睛刺激:求医/就诊。
食入: 若感不适,求医/就诊。漱口。
紧急救助者的防护: 救援者需要穿戴个人防护用品,比如橡胶手套和气密性护目镜。
模块 5. 消防措施
合适的灭火剂: 干粉,泡沫,二氧化碳
不适用的灭火剂: 水(有可能扩大灾情。)
特定方法: 从上风处灭火,根据周围环境选择合适的灭火方法。
非相关人员应该撤离至安全地方。
周围一旦着火:喷水,保持容器冷却。如果安全,消除一切火源。
消防员的特殊防护用具: 灭火时,一定要穿戴个人防护用品。
模块 6. 泄漏应急处理
个人防护措施,防护用具, 使用个人防护用品。远离溢出物/泄露处并处在上风处。确保足够通风。
紧急措施: 泄露区应该用安全带等圈起来,控制非相关人员进入。
环保措施: 防止进入下水道。
控制和清洗的方法和材料: 回收到密闭容器前用干砂或惰性吸收剂吸收泄漏物。一旦大量泄漏,筑堤控制。附着
物或收集物应该根据相关法律法规废弃处置。
副危险性的防护措施 移除所有火源。一旦发生火灾应该准备灭火器。使用防火花工具和防爆设备。
模块 7. 操作处置与储存
处理
技术措施: 在通风良好处进行处理。穿戴合适的防护用具。防止烟雾产生。远离热源/火花/明火
/热表面。禁烟。采取措施防止静电积累。使用防爆设备。处理后彻底清洗双手和脸。
注意事项: 如果可能,使用封闭系统。如果蒸气或浮质产生,使用通风、局部排气。
操作处置注意事项: 避免接触皮肤、眼睛和衣物。
贮存
储存条件: 保持容器密闭。存放于凉爽、阴暗、通风良好处。
远离不相容的材料比如氧化剂存放。
包装材料: 依据法律。
模块 8. 接触控制和个体防护
工程控制: 尽可能安装封闭体系或局部排风系统。同时安装淋浴器和洗眼器。
个人防护用品
呼吸系统防护: 防毒面具。依据当地和政府法规。
手部防护: 防护手套。
眼睛防护: 安全防护镜。如果情况需要,佩戴面具。
皮肤和身体防护: 防护服。如果情况需要,穿戴防护靴。
模块 9. 理化特性
液体
外形(20°C):
外观: 透明
颜色: 无色-黄色
气味: 无资料
pH: 无数据资料
熔点: 无资料
1,5-癸二炔 修改号码:2
模块 9. 理化特性
沸点/沸程 79 °C/2.7kPa
闪点: 无资料
爆炸特性
爆炸下限: 无资料
爆炸上限: 无资料
密度: 0.82
溶解度: 无资料
模块 10. 稳定性和反应性
稳定性: 一般情况下稳定。
反应性: 未报道特殊反应性。
须避免接触的物质 氧化剂
危险的分解产物: 一氧化碳, 二氧化碳
模块 11. 毒理学信息
急性毒性: 无资料
对皮肤腐蚀或刺激: 无资料
对眼睛严重损害或刺激: 无资料
生殖细胞变异原性: 无资料
致癌性:
IARC = 无资料
NTP = 无资料
生殖毒性: 无资料
模块 12. 生态学信息
生态毒性:
鱼类: 无资料
甲壳类: 无资料
藻类: 无资料
残留性 / 降解性: 无资料
潜在生物累积 (BCF): 无资料
土壤中移动性
log水分配系数: 无资料
土壤吸收系数 (Koc): 无资料
亨利定律 无资料
constaNT(PaM3/mol):
模块 13. 废弃处置
如果可能,回收处理。请咨询当地管理部门。建议在装有后燃和洗涤装置的化学焚烧炉中焚烧。废弃处置时请遵守
国家、地区和当地的所有法规。
模块 14. 运输信息
联合国分类: 第3类 易燃液体 。
UN编号: 3295
正式运输名称: 碳氢化合物, 液体, 不另作详细说明
包装等级: III
模块 15. 法规信息
《危险化学品安全管理条例》(2002年1月26日国务院发布): 针对危险化学品的安全使用、生产、储存、运输、装
卸等方面均作了相应的规定。
1,5-癸二炔 修改号码:2
模块16 - 其他信息
N/A
模块 1. 化学品
产品名称: 1,5-Decadiyne
修改号码: 2
模块 2. 危险性概述
GHS分类
物理性危害
易燃液体 第3级
健康危害 未分类
环境危害 未分类
GHS标签元素
图标或危害标志
信号词 警告
危险描述 易燃液体和蒸气
防范说明
[预防] 远离热源/火花/明火/热表面。禁烟。
保持容器密闭。
使用防爆的电气/通风/照明设备。采取预防措施以防静电和火花引起的着火。
穿戴防护手套/护目镜/防护面具。
[急救措施] 皮肤接触:立即去除/脱掉所有被污染的衣物。用水清洗皮肤/淋浴。
[储存] 存放于通风良好处。保持凉爽。
[废弃处置] 根据当地政府规定把物品/容器交与工业废弃处理机构。
模块 3. 成分/组成信息
单一物质/混和物 单一物质
化学名(中文名): 1,5-癸二炔
百分比: >97.0%(GC)
CAS编码: 53963-03-4
分子式: C10H14
模块 4. 急救措施
吸入: 将受害者移到新鲜空气处,保持呼吸通畅,休息。若感不适请求医/就诊。
1,5-癸二炔 修改号码:2
模块 4. 急救措施
皮肤接触: 立即去除/脱掉所有被污染的衣物。用水清洗皮肤/淋浴。
若皮肤刺激或发生皮疹:求医/就诊。
眼睛接触: 用水小心清洗几分钟。如果方便,易操作,摘除隐形眼镜。继续清洗。
如果眼睛刺激:求医/就诊。
食入: 若感不适,求医/就诊。漱口。
紧急救助者的防护: 救援者需要穿戴个人防护用品,比如橡胶手套和气密性护目镜。
模块 5. 消防措施
合适的灭火剂: 干粉,泡沫,二氧化碳
不适用的灭火剂: 水(有可能扩大灾情。)
特定方法: 从上风处灭火,根据周围环境选择合适的灭火方法。
非相关人员应该撤离至安全地方。
周围一旦着火:喷水,保持容器冷却。如果安全,消除一切火源。
消防员的特殊防护用具: 灭火时,一定要穿戴个人防护用品。
模块 6. 泄漏应急处理
个人防护措施,防护用具, 使用个人防护用品。远离溢出物/泄露处并处在上风处。确保足够通风。
紧急措施: 泄露区应该用安全带等圈起来,控制非相关人员进入。
环保措施: 防止进入下水道。
控制和清洗的方法和材料: 回收到密闭容器前用干砂或惰性吸收剂吸收泄漏物。一旦大量泄漏,筑堤控制。附着
物或收集物应该根据相关法律法规废弃处置。
副危险性的防护措施 移除所有火源。一旦发生火灾应该准备灭火器。使用防火花工具和防爆设备。
模块 7. 操作处置与储存
处理
技术措施: 在通风良好处进行处理。穿戴合适的防护用具。防止烟雾产生。远离热源/火花/明火
/热表面。禁烟。采取措施防止静电积累。使用防爆设备。处理后彻底清洗双手和脸。
注意事项: 如果可能,使用封闭系统。如果蒸气或浮质产生,使用通风、局部排气。
操作处置注意事项: 避免接触皮肤、眼睛和衣物。
贮存
储存条件: 保持容器密闭。存放于凉爽、阴暗、通风良好处。
远离不相容的材料比如氧化剂存放。
包装材料: 依据法律。
模块 8. 接触控制和个体防护
工程控制: 尽可能安装封闭体系或局部排风系统。同时安装淋浴器和洗眼器。
个人防护用品
呼吸系统防护: 防毒面具。依据当地和政府法规。
手部防护: 防护手套。
眼睛防护: 安全防护镜。如果情况需要,佩戴面具。
皮肤和身体防护: 防护服。如果情况需要,穿戴防护靴。
模块 9. 理化特性
液体
外形(20°C):
外观: 透明
颜色: 无色-黄色
气味: 无资料
pH: 无数据资料
熔点: 无资料
1,5-癸二炔 修改号码:2
模块 9. 理化特性
沸点/沸程 79 °C/2.7kPa
闪点: 无资料
爆炸特性
爆炸下限: 无资料
爆炸上限: 无资料
密度: 0.82
溶解度: 无资料
模块 10. 稳定性和反应性
稳定性: 一般情况下稳定。
反应性: 未报道特殊反应性。
须避免接触的物质 氧化剂
危险的分解产物: 一氧化碳, 二氧化碳
模块 11. 毒理学信息
急性毒性: 无资料
对皮肤腐蚀或刺激: 无资料
对眼睛严重损害或刺激: 无资料
生殖细胞变异原性: 无资料
致癌性:
IARC = 无资料
NTP = 无资料
生殖毒性: 无资料
模块 12. 生态学信息
生态毒性:
鱼类: 无资料
甲壳类: 无资料
藻类: 无资料
残留性 / 降解性: 无资料
潜在生物累积 (BCF): 无资料
土壤中移动性
log水分配系数: 无资料
土壤吸收系数 (Koc): 无资料
亨利定律 无资料
constaNT(PaM3/mol):
模块 13. 废弃处置
如果可能,回收处理。请咨询当地管理部门。建议在装有后燃和洗涤装置的化学焚烧炉中焚烧。废弃处置时请遵守
国家、地区和当地的所有法规。
模块 14. 运输信息
联合国分类: 第3类 易燃液体 。
UN编号: 3295
正式运输名称: 碳氢化合物, 液体, 不另作详细说明
包装等级: III
模块 15. 法规信息
《危险化学品安全管理条例》(2002年1月26日国务院发布): 针对危险化学品的安全使用、生产、储存、运输、装
卸等方面均作了相应的规定。
1,5-癸二炔 修改号码:2
模块16 - 其他信息
N/A
上下游信息
反应信息
-
作为反应物:参考文献:名称:Mild chemo-selective hydration of terminal alkynes catalysed by AgSbF6摘要:报道了一种在温和条件下,由AgSbF6催化的广泛范围的未活化末端炔烃的选择性氢化反应。DOI:10.1039/c1cc12928g
-
作为产物:参考文献:名称:Intermediate for Gossyplure, the sex pheromone of the pink bollworm摘要:粉红铃蛾(Pectinophora gossypiella)的性信息素是大约1:1的(Z,Z)和(Z,E)-7,11-十六烯基乙酸酯的混合物。这种性信息素,即上述异构体的混合物,被称为Gossyplure。Gossyplure的一个组分(Z,E)-7,11-十六烯基乙酸酯是谷仓粉螟(Sitotroga cerealella)的性信息素,被称为Angoulure。Gossyplure的另一个异构体成分,即Z,Z异构体,不能引起雄性谷仓粉螟的反应,因此Gossyplure中相对较大的Z,Z异构体量干扰了Z,E异构体的作用,使得Gossyplure不能替代Angoulure。Gossyplure的异构成分是通过对1,5-己二炔的单负离子进行丁基化制备的1,5-癸二炔,然后用六亚甲基卤水合物或其保护衍生物(如四氢吡喃醚)烷基化,得到7,11-十六炔基基团,经过乙酰化和部分还原后得到Gossyplure的一个异构体成分(Z,Z)-7,11-十六烯基乙酸酯。Gossyplure的另一个异构体成分Z,E异构体,也称为Angoulure,是谷仓粉螟的性信息素,以类似的方式从十碳炔(通过1,5-癸二炔的钠液氨还原制备)合成。公开号:US03996270A1
-
作为试剂:描述:1,4-Bis(ethenyl)benzene;iodobenzene;styrene 、 乙炔 在 water TEA 、 1,5-癸二炔 、 N,N-二甲基甲酰胺 、 potassium carbonate 、 四丁基溴化铵 、 氩 、 四(三苯基膦)钯 、 乙酸铵 、 乙二醇二甲醚 、 水 、 盐酸 、 甲醇 、 二氯甲烷 作用下, 以 N,N-二甲基甲酰胺 为溶剂, 反应 18.17h, 生成 1,5-癸二炔参考文献:名称:Attached tags for use in combinatorial chemistry synthesis摘要:本发明涉及一种编码和识别化学组合物库中个体成员的方法,该库在多个固体支持体上合成,经历混合和分离合成。该过程提供了一种将固体支持体标记为编码标识符的方法,该编码标识符附着于固体支持体上,并且可以通过红外或拉曼光谱直接附着于支持体上进行解码。公开号:US06355490B1
文献信息
-
Scope of the Intramolecular Titanocene-Catalyzed Pauson−Khand Type Reaction<sup>1</sup>作者:Frederick A. Hicks、Natasha M. Kablaoui、Stephen L. BuchwaldDOI:10.1021/ja990682u日期:1999.6.1A Pauson−Khand type conversion of enynes to bicyclic cyclopentenones employing the commercially available precatalyst titanocene dicarbonyl is described. This methodology shows excellent functional group tolerance for a group 4 metallocene-catalyzed process. The scope and limitations of this cyclization with respect to 1,6-, 1,7- and 1,8-enynes with a variety of terminal alkyne substituents, chiral
-
SO<sub>2</sub>F<sub>2</sub>-Mediated Oxidative Dehydrogenation and Dehydration of Alcohols to Alkynes作者:Gao-Feng Zha、Wan-Yin Fang、You-Gui Li、Jing Leng、Xing Chen、Hua-Li QinDOI:10.1021/jacs.8b10069日期:2018.12.19Direct synthesis of alkynes from inexpensive, abundant alcohols was achieved in high yields (greater than 40 examples, up to 95% yield) through a SO2F2-promoted dehydration and dehydrogenation process. This straightforward transformation of sp3-sp3 (C-C) bonds to sp-sp (C≡C) bonds requires only inexpensive and readily available reagents (no transition metals) under mild conditions. The crude alkynes
-
Rhodium(I)-Catalyzed Ring-Closing Reaction of Allene-Alkene-Alkynes: One-Step Construction of Tricyclo[6.4.0.0<sup>2,6</sup> ] and Bicyclo[6.3.0] Skeletons from Linear Carbon Chains作者:Yasuaki Kawaguchi、Asami Nagata、Kei Kurokawa、Haruna Yokosawa、Chisato MukaiDOI:10.1002/chem.201801239日期:2018.5.2afforded bicyclo[6.3.0]undecatriene derivatives instead of tricyclic compounds, the latter of which are well known as a basic skeleton of naturally occurring octanoids. On the basis of two experiments with deuterated substrates, a plausible reaction mechanism for the construction of these products was proposed.
-
Copper-Catalyzed Carbamoylation of Terminal Alkynes with Formamides via Cross-Dehydrogenative Coupling作者:Jin-Ji Wu、Yinwu Li、Hai-Yun Zhou、A-Hao Wen、Chu-Chu Lun、Su-Yang Yao、Zhuofeng Ke、Bao-Hui YeDOI:10.1021/acscatal.5b02881日期:2016.2.5efficient approach for direct carbamoylation of terminal alkynes with formamides affording propiolamides has been developed by copper-catalyzed oxidative cross coupling of C(sp)-H and C(sp2)-H bonds in the presence of a pincer ligand with two imidazolyl groups. The catalytic reaction is compatible with diverse functional groups but sensitive to the electronic effect of terminal alkyne and the steric effect
-
Copper/Guanidine-Catalyzed Asymmetric Alkynylation of Isatins作者:Quangang Chen、Yu Tang、Tianyu Huang、Xiaohua Liu、Lili Lin、Xiaoming FengDOI:10.1002/anie.201600711日期:2016.4.18The highly enantioselective alkynylation of isatins, catalyzed by a bifunctional guanidine/CuI catalyst under mild reaction conditions, is described. The reaction is broad in scope with respect to alkyl/aryl‐substituted terminal alkynes and substituted isatins, thus affording bioactive propargylic alcohols in excellent yields and enantioselectivities.
表征谱图
-
氢谱1HNMR
-
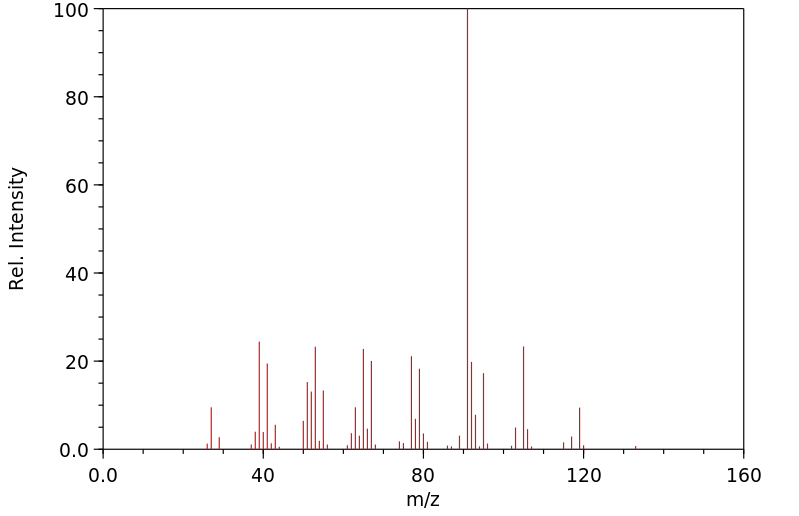
质谱MS
-
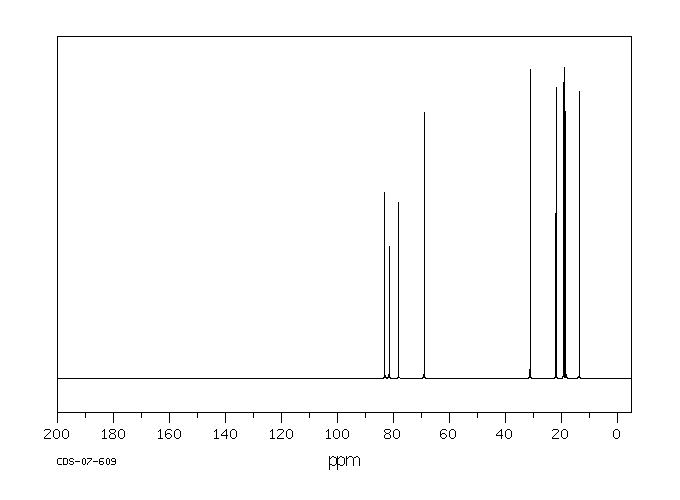
碳谱13CNMR
-
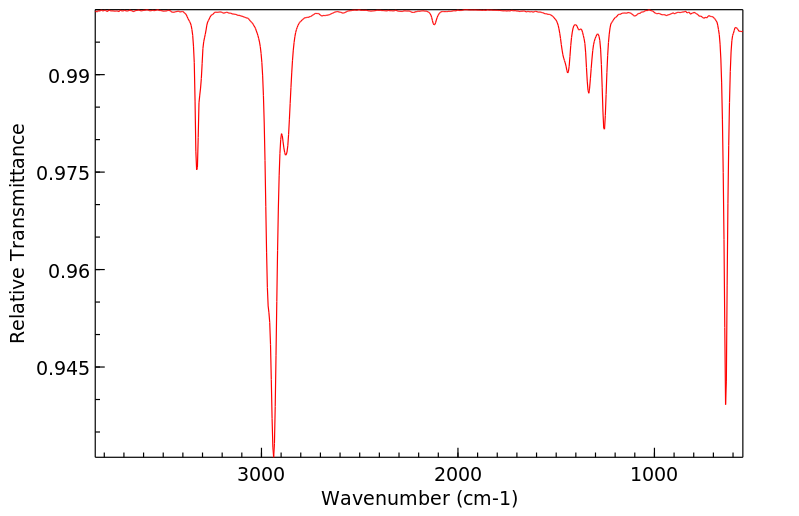
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
锗烷,三甲基[3-(三甲基甲锡烷基)-2-炔丙基]-
锗烷,三甲基-2-炔丙基-
铜,1-戊炔基-
甲基炔丙基硫化物
甲基乙炔和丙二烯混合物
甲基丙-2-炔基氰基二硫代亚氨酸酯
甲基-D3-乙炔
环戊基乙炔
环己基乙炔
环丙乙炔
炔丙胺
炔丙基膦
炔丙基碘化物
炔丙基叔丁基二甲基硅烷
炔丙基三甲基硅烷
炔丙基三乙基硅烷
氘乙炔
戊-1-炔-3-胺
戊-1,3-二炔
戊-1,2-二烯-4-炔
异氰基-乙炔
己基(己-5-炔基)甲基硅烷
己-1-炔银
四碳化铀
反式-4-(2-丙炔基)-环己烷甲醇
双(三甲基锡)乙炔
双(三氟甲基)锌
十四碳-1,4-二炔
十四碳-1,3-二炔
十八碳-1,17-二炔
十八炔
十三碳-1,7-二炔
十三碳-1,12-二炔
十一碳-1,5-二炔
亚硫酸二(2-丙炔基)酯
二甲基炔丙基溴化硫
二炔丙基硫醚
二乙炔基-二甲基-锗烷
二丙-1-炔基汞
二[2-甲氧基乙基汞(II)]乙炔
二(三正丁基甲锡烷基)乙炔
二(3-羟基-1-丙炔基)汞(II)
乙炔锂乙二胺配合物
乙炔银
乙炔基环己烷钠
乙炔基环丙烷氯化镁
乙炔基(三甲基)锗烷
乙炔基(三甲基)硅烷铜(1+)
乙炔基(三甲基)硅烷溴化镁
乙炔基(三甲基)硅烷氯化镁