1-溴甲基萘 | 3163-27-7
中文名称
1-溴甲基萘
中文别名
——
英文名称
2-(bromomethyl)naphthalene
英文别名
1-bromomethylnaphthalene;1-(Bromomethyl)naphthalene
CAS
3163-27-7
化学式
C11H9Br
mdl
——
分子量
221.096
InChiKey
RZJGKPNCYQZFGR-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:52-55°C
-
沸点:213°C/100mmHg
-
密度:1.44
-
最大波长(λmax):292nm(Cyclohexane)(lit.)
计算性质
-
辛醇/水分配系数(LogP):3.8
-
重原子数:12
-
可旋转键数:1
-
环数:2.0
-
sp3杂化的碳原子比例:0.09
-
拓扑面积:0
-
氢给体数:0
-
氢受体数:0
安全信息
-
危险等级:8
-
安全说明:S26,S36
-
海关编码:2903999090
-
危险品标志:Xi
-
危险类别码:R20/21/22
-
包装等级:II
-
危险类别:8
-
危险性防范说明:P234,P260,P264,P272,P280,P301+P330+P331,P303+P361+P353,P304+P340+P310,P305+P351+P338+P310,P333+P313,P362+P364,P390,P405,P406,P501
-
危险品运输编号:3261
-
危险性描述:H290,H314,H317
-
储存条件:存储于0-5°C环境中
SDS
Material Safety Data Sheet
Section 1. Identification of the substance
Product Name: 1-(Bromomethyl)naphthalene
Synonyms:
Section 2. Hazards identification
Harmful by inhalation, in contact with skin, and if swallowed.
H314: Causes severe skin burns and eye damage
H318: Causes serious eye damage
P260: Do not breathe dust/fume/gas/mist/vapours/spray
P303+P361+P353: IF ON SKIN (or hair): Remove/Take off immediately all contaminated clothing. Rinse skin with
water/shower
P305+P351+P338: IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses if present
and easy to do – continue rinsing
P301+P330+P331: IF SWALLOWED: Rinse mouth. Do NOT induce vomiting
P405: Store locked up
Section 3. Composition/information on ingredients.
Ingredient name: 1-(Bromomethyl)naphthalene
CAS number: 3163-27-7
Section 4. First aid measures
Skin contact: Immediately wash skin with copious amounts of water for at least 15 minutes while removing
contaminated clothing and shoes. If irritation persists, seek medical attention.
Eye contact: Immediately wash skin with copious amounts of water for at least 15 minutes. Assure adequate
flushing of the eyes by separating the eyelids with fingers. If irritation persists, seek medical
attention.
Inhalation: Remove to fresh air. In severe cases or if symptoms persist, seek medical attention.
Ingestion: Wash out mouth with copious amounts of water for at least 15 minutes. Seek medical attention.
Section 5. Fire fighting measures
In the event of a fire involving this material, alone or in combination with other materials, use dry
powder or carbon dioxide extinguishers. Protective clothing and self-contained breathing apparatus
should be worn.
Section 6. Accidental release measures
Personal precautions: Wear suitable personal protective equipment which performs satisfactorily and meets local/state/national
standards.
Respiratory precaution: Wear approved mask/respirator
Hand precaution: Wear suitable gloves/gauntlets
Skin protection: Wear suitable protective clothing
Eye protection: Wear suitable eye protection
Methods for cleaning up: Mix with sand or similar inert absorbent material, sweep up and keep in a tightly closed container
for disposal. See section 12.
Environmental precautions: Do not allow material to enter drains or water courses.
Section 7. Handling and storage
Handling: This product should be handled only by, or under the close supervision of, those properly qualified
in the handling and use of potentially hazardous chemicals, who should take into account the fire,
health and chemical hazard data given on this sheet.
Storage: Store in closed vessels, refrigerated.
Section 8. Exposure Controls / Personal protection
Engineering Controls: Use only in a chemical fume hood.
Personal protective equipment: Wear laboratory clothing, chemical-resistant gloves and safety goggles.
General hydiene measures: Wash thoroughly after handling. Wash contaminated clothing before reuse.
Section 9. Physical and chemical properties
Not specified
Appearance:
Boiling point: No data
Melting point: No data
Flash point: No data
Density: No data
Molecular formula: C11H9Br
Molecular weight: 221.1
Section 10. Stability and reactivity
Conditions to avoid: Heat, flames and sparks.
Materials to avoid: Oxidizing agents.
Possible hazardous combustion products: Carbon monoxide, hydrogen bromide.
Section 11. Toxicological information
No data.
Section 12. Ecological information
No data.
Section 13. Disposal consideration
Arrange disposal as special waste, by licensed disposal company, in consultation with local waste
disposal authority, in accordance with national and regional regulations.
Section 14. Transportation information
UN Number: UN3261 Class: 8 Packing group: II
Proper shipping name: CORROSIVE SOLID, ACIDIC, ORGANIC, N.O.S. (1-(Bromomethyl)naphthalene)
Section 15. Regulatory information
No chemicals in this material are subject to the reporting requirements of SARA Title III, Section
302, or have known CAS numbers that exceed the threshold reporting levels established by SARA
Title III, Section 313.
SECTION 16 - ADDITIONAL INFORMATION
N/A
Section 1. Identification of the substance
Product Name: 1-(Bromomethyl)naphthalene
Synonyms:
Section 2. Hazards identification
Harmful by inhalation, in contact with skin, and if swallowed.
H314: Causes severe skin burns and eye damage
H318: Causes serious eye damage
P260: Do not breathe dust/fume/gas/mist/vapours/spray
P303+P361+P353: IF ON SKIN (or hair): Remove/Take off immediately all contaminated clothing. Rinse skin with
water/shower
P305+P351+P338: IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses if present
and easy to do – continue rinsing
P301+P330+P331: IF SWALLOWED: Rinse mouth. Do NOT induce vomiting
P405: Store locked up
Section 3. Composition/information on ingredients.
Ingredient name: 1-(Bromomethyl)naphthalene
CAS number: 3163-27-7
Section 4. First aid measures
Skin contact: Immediately wash skin with copious amounts of water for at least 15 minutes while removing
contaminated clothing and shoes. If irritation persists, seek medical attention.
Eye contact: Immediately wash skin with copious amounts of water for at least 15 minutes. Assure adequate
flushing of the eyes by separating the eyelids with fingers. If irritation persists, seek medical
attention.
Inhalation: Remove to fresh air. In severe cases or if symptoms persist, seek medical attention.
Ingestion: Wash out mouth with copious amounts of water for at least 15 minutes. Seek medical attention.
Section 5. Fire fighting measures
In the event of a fire involving this material, alone or in combination with other materials, use dry
powder or carbon dioxide extinguishers. Protective clothing and self-contained breathing apparatus
should be worn.
Section 6. Accidental release measures
Personal precautions: Wear suitable personal protective equipment which performs satisfactorily and meets local/state/national
standards.
Respiratory precaution: Wear approved mask/respirator
Hand precaution: Wear suitable gloves/gauntlets
Skin protection: Wear suitable protective clothing
Eye protection: Wear suitable eye protection
Methods for cleaning up: Mix with sand or similar inert absorbent material, sweep up and keep in a tightly closed container
for disposal. See section 12.
Environmental precautions: Do not allow material to enter drains or water courses.
Section 7. Handling and storage
Handling: This product should be handled only by, or under the close supervision of, those properly qualified
in the handling and use of potentially hazardous chemicals, who should take into account the fire,
health and chemical hazard data given on this sheet.
Storage: Store in closed vessels, refrigerated.
Section 8. Exposure Controls / Personal protection
Engineering Controls: Use only in a chemical fume hood.
Personal protective equipment: Wear laboratory clothing, chemical-resistant gloves and safety goggles.
General hydiene measures: Wash thoroughly after handling. Wash contaminated clothing before reuse.
Section 9. Physical and chemical properties
Not specified
Appearance:
Boiling point: No data
Melting point: No data
Flash point: No data
Density: No data
Molecular formula: C11H9Br
Molecular weight: 221.1
Section 10. Stability and reactivity
Conditions to avoid: Heat, flames and sparks.
Materials to avoid: Oxidizing agents.
Possible hazardous combustion products: Carbon monoxide, hydrogen bromide.
Section 11. Toxicological information
No data.
Section 12. Ecological information
No data.
Section 13. Disposal consideration
Arrange disposal as special waste, by licensed disposal company, in consultation with local waste
disposal authority, in accordance with national and regional regulations.
Section 14. Transportation information
UN Number: UN3261 Class: 8 Packing group: II
Proper shipping name: CORROSIVE SOLID, ACIDIC, ORGANIC, N.O.S. (1-(Bromomethyl)naphthalene)
Section 15. Regulatory information
No chemicals in this material are subject to the reporting requirements of SARA Title III, Section
302, or have known CAS numbers that exceed the threshold reporting levels established by SARA
Title III, Section 313.
SECTION 16 - ADDITIONAL INFORMATION
N/A
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 甲基萘 1-Methylnaphthalene 90-12-0 C11H10 142.2 1-萘甲醇 1-naphthalene methanol 4780-79-4 C11H10O 158.2 1-萘甲醛 1-naphthaldehyde 66-77-3 C11H8O 156.184 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 甲基萘 1-Methylnaphthalene 90-12-0 C11H10 142.2 1-溴-4-(溴甲基)萘 1-bromo-4-bromomethyl-naphthalene 79996-99-9 C11H8Br2 299.993 1-萘甲醇 1-naphthalene methanol 4780-79-4 C11H10O 158.2 1-乙基萘 1-ethylnapthelene 1127-76-0 C12H12 156.227 1-(碘甲基)-萘 1-(iodomethyl)naphthalene 24471-54-3 C11H9I 268.097 1-萘甲醛 1-naphthaldehyde 66-77-3 C11H8O 156.184 1-(氟甲基)萘 1-(fluoromethyl)naphthalene 55831-10-2 C11H9F 160.191 氰基萘 1-Cyanonaphthalene 86-53-3 C11H7N 153.183 1-萘甲基胺 (naphth-1-yl)methylamine 118-31-0 C11H11N 157.215 抑芽醚 1-(methoxymethyl)naphthalene 5903-23-1 C12H12O 172.227 1-萘乙胺盐酸盐 2-naphthalen-1-yl-ethylamine 4735-50-6 C12H13N 171.242 - 1
- 2
反应信息
-
作为反应物:描述:参考文献:名称:Synthesis and Biological Activity of New 3-Hydroxy-3-methylglutaryl Coenzyme A (HMG-CoA) Synthase Inhibitors: 2-Oxetanones with a Side Chain Mimicking the Folded Structure of 1233A.摘要:为模拟1233A(1)的折叠侧链构象,该化合物是一种3-羟基-3-甲基戊二酰辅酶A(HMG-CoA)合酶抑制剂,合成了侧链含有芳环的1233A类似物。其中2-恶坦酮部分保持不变。在1233A及其合成类似物中,反式-3-羟甲基-4-[2-(7-甲氧羰基-1-萘基)乙基]-2-恶坦酮(23)显示了最高的HMG-CoA合酶体外抑制活性。讨论了侧链上的构效关系。DOI:10.1248/cpb.42.512
-
作为产物:描述:参考文献:名称:一种用卤素取代羟基的温和方法:2.统一的程序和立体化学研究摘要:N,N-二甲基-和N,N-二异丙基-1-卤代-2-甲基-1-丙烯胺是容易获得的用于醇和羟基酸的轻度脱氧卤代的试剂。在这项研究中,我们表明可以通过改变试剂上烷基的大小来调节试剂的反应性:用异丙基取代甲基可以显着提高反应性。然后,我们使用容易获得的α描述了所有脱氧卤化反应的统一程序-氯亚胺作为试剂,带有(溴化,碘化)或不带有(氯化)碱性溴化物或碘化物。最后,我们表明仲醇的脱氧卤化反应具有高度的立体定向性,通常发生构型反转。DOI:10.1016/j.tet.2020.131441
-
作为试剂:参考文献:名称:AZOLIUM AND PURINIUM SALT ANTICANCER AND ANTIMICROBIAL AGENTS摘要:单电荷和多电荷咪唑阳离子(ICs)已被确定为一类具有强效抗肿瘤、抗菌和抗微生物特性的化学成分。所公开的咪唑阳离子在对癌细胞的作用上表现出与当前临床标准药物顺铂相当或更强的效力。然而,这些咪唑阳离子在不像顺铂这样的重金属抗肿瘤药物所知的毒性副作用下,就能实现这种功效。公开号:US20140142307A1
文献信息
-
Copper promoted synthesis of substituted quinolines from benzylic azides and alkynes作者:Ching-Zong Luo、Parthasarathy Gandeepan、Yun-Ching Wu、Wei-Chen Chen、Chien-Hong ChengDOI:10.1039/c5ra23065a日期:——novel copper promoted synthesis of substituted quinolines from various benzylic azides and internal alkynes has been demonstrated. The reaction features a broad substrate scope, high product yields and excellent regioselectivity. In contrast to the known two-step process of acid promoted [4 + 2] cycloaddition reaction and oxidation, the present methodology allows the synthesis of quinolines in a single
-
Synthesis and spectroscopic properties of 1,4-diarylbutenynes作者:Alois H. A. Tinnemans、Wim H. LaarhovenDOI:10.1039/p29760001104日期:——Several new diarylbutenynes have been synthesized and analyses of the n.m.r. and u.v. spectra are described. For planar trans-isomers a conformational preference was found for 1-(α-naphthyl)- but not for 1-(β-naphthyl)-4-arylbutenynes. This difference is also found between compounds with C-1 attached to the α- or β-position of a larger aryl group.
-
Mercapto-acylamino acid antihypertensives申请人:Schering Corporation公开号:US05061710A1公开(公告)日:1991-10-29Novel mercapto-acylamino acids useful in the treatment of hypertension and combinations of mercapto-acylamino acids and atrial natriuretic factors or angiotensin converting enzyme inhibitors useful for treating hypertension are disclosed.
-
A Unified Catalytic Asymmetric (4+1) and (5+1) Annulation Strategy to Access Chiral Spirooxindole‐Fused Oxacycles作者:Min Gao、Yanshu Luo、Qianlan Xu、Yukun Zhao、Xiangnan Gong、Yuanzhi Xia、Lin HuDOI:10.1002/anie.202105282日期:2021.9A unified catalytic asymmetric (N+1) (N=4, 5) annulation reaction of oxindoles with bifunctional peroxides has been achieved in the presence of a chiral phase-transfer catalyst (PTC). This general strategy utilizes peroxides as unique bielectrophilic four- or five-atom synthons to participate in the C−C and the subsequent umpolung C−O bond-forming reactions with one-carbon unit nucleophiles, thus providing
-
NNN-pincer-copper complex immobilized on magnetic nanoparticles as a powerful hybrid catalyst for aerobic oxidative coupling and cycloaddition reactions in water作者:Nasrin Zohreh、Mahboobeh JahaniDOI:10.1016/j.molcata.2016.11.007日期:2017.1such as SEM, TEM, FT-IR, TGA, ICP, XRD, and elemental analysis. The finely engineered supported catalyst is employed in the aerobic oxidative coupling of terminal alkynes and click reaction using only 0.38 and 0.04 mol% catalyst, respectively. All reactions perform under solvent-free condition or green solvent H 2 O. Also, the catalyst is readily recovered and reused for up to 8 and 6 subsequent runs
表征谱图
-
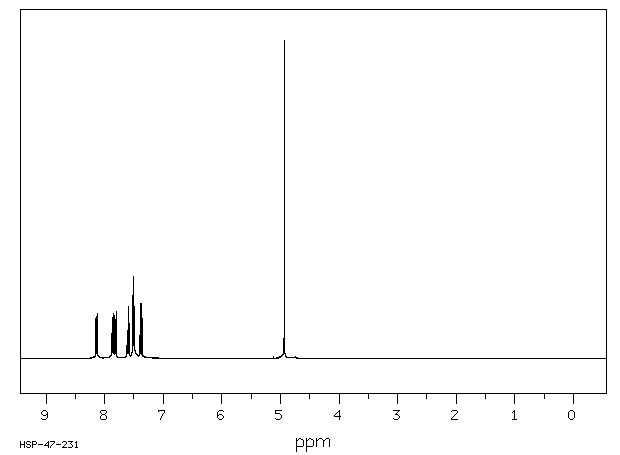
氢谱1HNMR
-
质谱MS
-
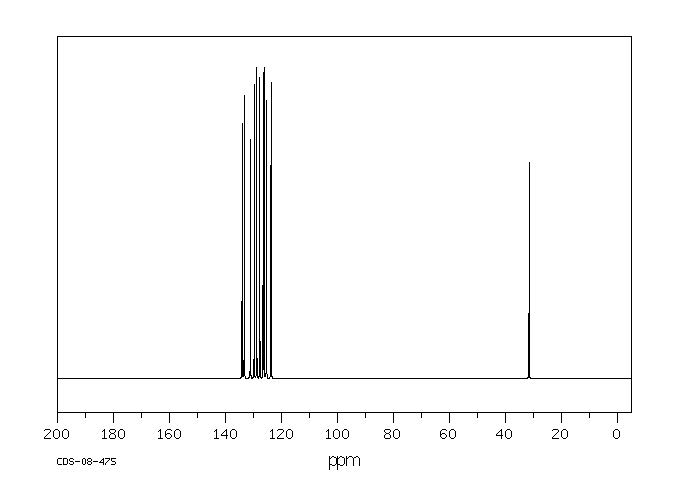
碳谱13CNMR
-
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(S)-溴烯醇内酯
(R)-3,3''-双([[1,1''-联苯]-4-基)-[1,1''-联萘]-2,2''-二醇
(3S,3aR)-2-(3-氯-4-氰基苯基)-3-环戊基-3,3a,4,5-四氢-2H-苯并[g]吲唑-7-羧酸
(3R,3’’R,4S,4’’S,11bS,11’’bS)-(+)-4,4’’-二叔丁基-4,4’’,5,5’’-四氢-3,3’’-联-3H-二萘酚[2,1-c:1’’,2’’-e]膦(S)-BINAPINE
(11bS)-2,6-双(3,5-二甲基苯基)-4-羟基-4-氧化物-萘并[2,1-d:1'',2''-f][1,3,2]二氧磷
(11bS)-2,6-双(3,5-二氯苯基)-4羟基-4-氧-二萘并[2,1-d:1'',2''-f][1,3,2]二氧磷杂七环
(11bR)-2,6-双[3,5-双(1,1-二甲基乙基)苯基]-4-羟基-4-氧化物-二萘并[2,1-d:1'',2''-f][1,3,2]二氧杂磷平
黄胺酸
马兜铃对酮
马休黄钠盐一水合物
马休黄
食品黄6号
食品红40铝盐色淀
飞龙掌血香豆醌
颜料黄101
颜料红70
颜料红63
颜料红53:3
颜料红5
颜料红48单钠盐
颜料红48:2
颜料红4
颜料红261
颜料红258
颜料红220
颜料红22
颜料红214
颜料红2
颜料红19
颜料红185
颜料红184
颜料红170
颜料红148
颜料红147
颜料红146
颜料红119
颜料红114
颜料红 9
颜料红 21
颜料橙7
颜料橙46
颜料橙38
颜料橙3
颜料橙22
颜料橙2
颜料橙17
颜料橙 5
颜料棕1
顺式-阿托伐醌-d5
雄甾烷-3,17-二酮