4-二甲基磺酰基苯甲酸 | 1206-37-7
中文名称
4-二甲基磺酰基苯甲酸
中文别名
——
英文名称
4-(N,N-dimethylsulfamoyl)benzoic acid
英文别名
4-(dimethylaminosulfonyl)benzoic acid;4-dimethylsulfamoylbenzoic acid;4-[(Dimethylamino)sulfonyl]benzoic acid;4-(dimethylsulfamoyl)benzoic acid
CAS
1206-37-7
化学式
C9H11NO4S
mdl
MFCD00721975
分子量
229.257
InChiKey
DGVPSAQTQGLBAM-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
计算性质
-
辛醇/水分配系数(LogP):0.6
-
重原子数:15
-
可旋转键数:3
-
环数:1.0
-
sp3杂化的碳原子比例:0.222
-
拓扑面积:83.1
-
氢给体数:1
-
氢受体数:5
安全信息
-
海关编码:2935009090
-
危险性防范说明:P261,P264,P270,P271,P280,P301+P312,P302+P352,P304+P340,P305+P351+P338,P330,P332+P313,P337+P313,P362,P403+P233,P405,P501
-
危险性描述:H302,H315,H319,H335
SDS
Material Safety Data Sheet
Section 1. Identification of the substance
Product Name: 4-[(Dimethylamino)sulfonyl]benzoic acid
Synonyms:
Section 2. Hazards identification
Harmful by inhalation, in contact with skin, and if swallowed.
Section 3. Composition/information on ingredients.
Ingredient name: 4-[(Dimethylamino)sulfonyl]benzoic acid
CAS number: 1206-37-7
Section 4. First aid measures
Skin contact: Immediately wash skin with copious amounts of water for at least 15 minutes while removing
contaminated clothing and shoes. If irritation persists, seek medical attention.
Eye contact: Immediately wash skin with copious amounts of water for at least 15 minutes. Assure adequate
flushing of the eyes by separating the eyelids with fingers. If irritation persists, seek medical
attention.
Inhalation: Remove to fresh air. In severe cases or if symptoms persist, seek medical attention.
Ingestion: Wash out mouth with copious amounts of water for at least 15 minutes. Seek medical attention.
Section 5. Fire fighting measures
In the event of a fire involving this material, alone or in combination with other materials, use dry
powder or carbon dioxide extinguishers. Protective clothing and self-contained breathing apparatus
should be worn.
Section 6. Accidental release measures
Personal precautions: Wear suitable personal protective equipment which performs satisfactorily and meets local/state/national
standards.
Respiratory precaution: Wear approved mask/respirator
Hand precaution: Wear suitable gloves/gauntlets
Skin protection: Wear suitable protective clothing
Eye protection: Wear suitable eye protection
Methods for cleaning up: Mix with sand or similar inert absorbent material, sweep up and keep in a tightly closed container
for disposal. See section 12.
Environmental precautions: Do not allow material to enter drains or water courses.
Section 7. Handling and storage
Handling: This product should be handled only by, or under the close supervision of, those properly qualified
in the handling and use of potentially hazardous chemicals, who should take into account the fire,
health and chemical hazard data given on this sheet.
Store in closed vessels.
Storage:
Section 8. Exposure Controls / Personal protection
Engineering Controls: Use only in a chemical fume hood.
Personal protective equipment: Wear laboratory clothing, chemical-resistant gloves and safety goggles.
General hydiene measures: Wash thoroughly after handling. Wash contaminated clothing before reuse.
Section 9. Physical and chemical properties
Appearance: Not specified
Boiling point: No data
No data
Melting point:
Flash point: No data
Density: No data
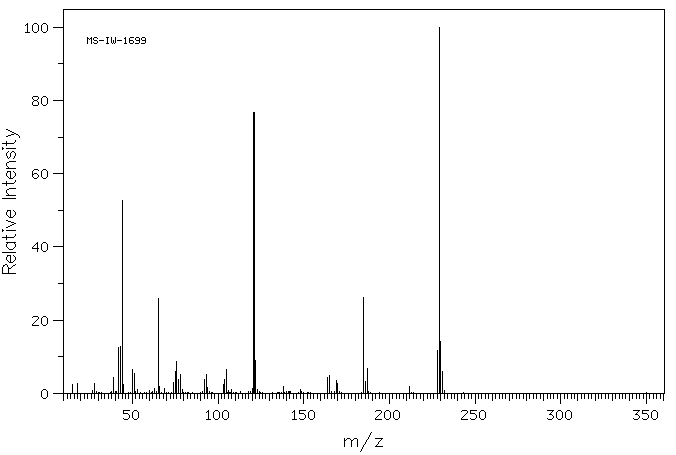
Molecular formula: C9H11NO4S
Molecular weight: 229.3
Section 10. Stability and reactivity
Conditions to avoid: Heat, flames and sparks.
Materials to avoid: Oxidizing agents.
Possible hazardous combustion products: Carbon monoxide, nitrogen oxides, sulfur oxides.
Section 11. Toxicological information
No data.
Section 12. Ecological information
No data.
Section 13. Disposal consideration
Arrange disposal as special waste, by licensed disposal company, in consultation with local waste
disposal authority, in accordance with national and regional regulations.
Section 14. Transportation information
Non-harzardous for air and ground transportation.
Section 15. Regulatory information
No chemicals in this material are subject to the reporting requirements of SARA Title III, Section
302, or have known CAS numbers that exceed the threshold reporting levels established by SARA
Title III, Section 313.
SECTION 16 - ADDITIONAL INFORMATION
N/A
Section 1. Identification of the substance
Product Name: 4-[(Dimethylamino)sulfonyl]benzoic acid
Synonyms:
Section 2. Hazards identification
Harmful by inhalation, in contact with skin, and if swallowed.
Section 3. Composition/information on ingredients.
Ingredient name: 4-[(Dimethylamino)sulfonyl]benzoic acid
CAS number: 1206-37-7
Section 4. First aid measures
Skin contact: Immediately wash skin with copious amounts of water for at least 15 minutes while removing
contaminated clothing and shoes. If irritation persists, seek medical attention.
Eye contact: Immediately wash skin with copious amounts of water for at least 15 minutes. Assure adequate
flushing of the eyes by separating the eyelids with fingers. If irritation persists, seek medical
attention.
Inhalation: Remove to fresh air. In severe cases or if symptoms persist, seek medical attention.
Ingestion: Wash out mouth with copious amounts of water for at least 15 minutes. Seek medical attention.
Section 5. Fire fighting measures
In the event of a fire involving this material, alone or in combination with other materials, use dry
powder or carbon dioxide extinguishers. Protective clothing and self-contained breathing apparatus
should be worn.
Section 6. Accidental release measures
Personal precautions: Wear suitable personal protective equipment which performs satisfactorily and meets local/state/national
standards.
Respiratory precaution: Wear approved mask/respirator
Hand precaution: Wear suitable gloves/gauntlets
Skin protection: Wear suitable protective clothing
Eye protection: Wear suitable eye protection
Methods for cleaning up: Mix with sand or similar inert absorbent material, sweep up and keep in a tightly closed container
for disposal. See section 12.
Environmental precautions: Do not allow material to enter drains or water courses.
Section 7. Handling and storage
Handling: This product should be handled only by, or under the close supervision of, those properly qualified
in the handling and use of potentially hazardous chemicals, who should take into account the fire,
health and chemical hazard data given on this sheet.
Store in closed vessels.
Storage:
Section 8. Exposure Controls / Personal protection
Engineering Controls: Use only in a chemical fume hood.
Personal protective equipment: Wear laboratory clothing, chemical-resistant gloves and safety goggles.
General hydiene measures: Wash thoroughly after handling. Wash contaminated clothing before reuse.
Section 9. Physical and chemical properties
Appearance: Not specified
Boiling point: No data
No data
Melting point:
Flash point: No data
Density: No data
Molecular formula: C9H11NO4S
Molecular weight: 229.3
Section 10. Stability and reactivity
Conditions to avoid: Heat, flames and sparks.
Materials to avoid: Oxidizing agents.
Possible hazardous combustion products: Carbon monoxide, nitrogen oxides, sulfur oxides.
Section 11. Toxicological information
No data.
Section 12. Ecological information
No data.
Section 13. Disposal consideration
Arrange disposal as special waste, by licensed disposal company, in consultation with local waste
disposal authority, in accordance with national and regional regulations.
Section 14. Transportation information
Non-harzardous for air and ground transportation.
Section 15. Regulatory information
No chemicals in this material are subject to the reporting requirements of SARA Title III, Section
302, or have known CAS numbers that exceed the threshold reporting levels established by SARA
Title III, Section 313.
SECTION 16 - ADDITIONAL INFORMATION
N/A
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 —— 4-<(Dimethylamino)sulfonyl>benzoic Acid Ethyl Ester 83112-37-2 C11H15NO4S 257.31 对羧基苯磺酰胺 4-(aminosulfonyl)-benzoic acid 138-41-0 C7H7NO4S 201.203 N,N-二甲基对甲苯磺酰胺 N,N,4-trimethylbenzenesulfonamide 599-69-9 C9H13NO2S 199.274 4-氰基-N,N-二甲基苯磺酰胺 4-cyano-N,N-dimethylbenzenesulfonamide 63877-94-1 C9H10N2O2S 210.257 4-氯-N,N-二甲基苯磺酰胺 4-chloro-N,N-dimethylbenzenesulfonamide 7463-22-1 C8H10ClNO2S 219.692 4-(氯磺酰)苯甲酸 p-carboxyphenylsulfonyl chloride 10130-89-9 C7H5ClO4S 220.633 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 —— 4-<(Dimethylamino)sulfonyl>benzoic Acid Ethyl Ester 83112-37-2 C11H15NO4S 257.31 —— 4-hydroxymethyl-benzenesulfonic acid dimethylamide 58804-18-5 C9H13NO3S 215.273 4-(二甲基氨基磺酰基)苯甲酰氯 4-(N,N-dimethylaminosulfonyl)benzoyl chloride 29171-70-8 C9H10ClNO3S 247.702 —— 4-dimethylsulfamoyl-benzoic acid methylamide 857486-81-8 C10H14N2O3S 242.299 —— 4-<(Dimethylamino)sulfonyl>benzoic Acid Hydrazide 53554-96-4 C9H13N3O3S 243.287
反应信息
-
作为反应物:描述:4-二甲基磺酰基苯甲酸 在 盐酸 、 一水合肼 作用下, 反应 15.0h, 生成 N,N-dimethyl-4-(5-phenyl-1,3,4-oxadiazol-2-yl)benzenesulfonamide参考文献:名称:Peet, Norton P.; Sunder, Shyam, Journal of Heterocyclic Chemistry, 1984, vol. 21, p. 1807 - 1816摘要:DOI:
-
作为产物:描述:乙基4-(氯磺酰基)苯甲酸酯 在 lithium hydroxide monohydrate 、 三乙胺 作用下, 以 甲醇 、 二氯甲烷 、 水 为溶剂, 反应 7.0h, 生成 4-二甲基磺酰基苯甲酸参考文献:名称:治疗耐多药结核病的噻吩苯磺酰胺衍生物的鉴定摘要:通过探索先导化合物2,3-二取代噻吩25a和297F的构效关系,设计并合成了一系列噻吩苯磺酰胺衍生物作为抗结核药物,对药物敏感和临床分离的耐药结核病表现出有效的抗分枝杆菌活性。特别是,与先导化合物相比,化合物17b具有改善的活性(最低抑制浓度为 0.023 μg/mL),在巨噬细胞中显示出良好的细胞内抗分枝杆菌活性,减少了 1.29 log 10 CFU。成药性评价表明,化合物17b具有良好的肝细胞稳定性、低细胞毒性和低hERG通道抑制作用。此外,化合物17b在急性结核病小鼠模型中表现出适度的体内功效。此外,分子对接研究阐明了化合物17b在DprE1活性位点的结合模式。因此,化合物17b可能成为进一步研究的有前景的抗结核先导药物。DOI:10.1016/j.ejmech.2022.114145
文献信息
-
Aroyl disulfides as promoter-modifiers for copolymerization of butadiene and styrene作者:Robert L. Frank、James R. Blegen、Archie DeutschmanDOI:10.1002/pol.1948.120030108日期:1948.2veratroyl, p-bromobenzoyl, p-phenylbenzoyl, p-benzoylbenzoyl, p-methylsulfonylbenzoyl, p(N,N-dimethylsulfonamido)-benzoyl and -naphthoyl disulfides are promoters of the emulsion copolymerization of butadiene and styrene, but exhibit little effect on the polymer properties. Furoyl, phenyl, p-chlorophenyl and p-bromophenyl disulfides are neither promoters nor modifiers.
-
Pyridyl inhibitors of hedgehog signalling申请人:Gunzner L. Janet公开号:US20060063779A1公开(公告)日:2006-03-23The invention provides novel inhibitors of hedgehog signaling that are useful as a therapeutic agents for treating malignancies where the compounds have the general formula I: wherein A, X, Y R 1 , R 2 , R 3 , R 4 , m and n are as described herein.这项发明提供了一种新型的刺刺信号抑制剂,可用作治疗恶性肿瘤的治疗剂,其中化合物具有一般式I: 其中A、X、Y、R1、R2、R3、R4、m和n如本文所述。
-
Discovery of novel thieno[2,3-d]pyrimidin-4-yl hydrazone-based inhibitors of Cyclin D1-CDK4: Synthesis, biological evaluation and structure–activity relationships. Part 2作者:Takao Horiuchi、Motoko Nagata、Mayumi Kitagawa、Kouichi Akahane、Kouichi UotoDOI:10.1016/j.bmc.2009.10.039日期:2009.12The design, synthesis and evaluation of novel thieno[2,3-d]pyrimidin-4-yl hydrazone analogues as cyclin-dependent kinase 4 (CDK4) inhibitor are described. Focusing on the optimization of the heteroaryl moiety at the hydrazone with substituted phenyl groups, 4-[(methylamino)methyl]benzaldehyde (22) and 5-isoindolinecarbaldehyde (24) (6-tert-butylthieno[2,3-d]pyrimidin-4-yl)hydrazone derivatives have
-
Substituted Dihydroimidazoles and their Use in the Treatment of Tumors申请人:Chamoin Sylvie公开号:US20100075966A1公开(公告)日:2010-03-25The invention relates to dihydroimidazoles of formula rac-(I), wherein the radicals and symbols are as defined herein; their use as inhibitors of the interaction of the MDM2 protein with a p53-like peptide, new pharmaceutical formulations comprising said compounds, said compounds for use in the therapeutic treatment of warm-blooded animals, especially humans, their use in the treatment of proliferative diseases or for the manufacture of pharmaceutical formulations useful in the treatment of proliferative diseases that respond to modulation of the interaction of the MDM2 protein with a p53-like peptide, a pharmaceutical formulation e.g. useful in the treatment of proliferative diseases that respond to modulation of the interaction of the MDM2 protein with a p53-like peptide comprising said compound, methods of treatment comprising administration of said compounds to a warm-blooded animal, and/or processes for the manufacture of said compounds.
-
Phosphine Oxides from a Medicinal Chemist’s Perspective: Physicochemical and <i>in Vitro</i> Parameters Relevant for Drug Discovery作者:Peter Finkbeiner、Jörg P. Hehn、Christian GnammDOI:10.1021/acs.jmedchem.0c00407日期:2020.7.9physicochemical properties of a real drug scaffold is exemplified by a series of phosphorus-containing analogs of imatinib. We demonstrate that phosphine oxides are highly polar functional groups leading to high solubility and metabolic stability but occasionally at the cost of reduced permeability. We conclude that phosphine oxides and related phosphorus-containing functional groups are valuable polar
表征谱图
-
氢谱1HNMR
-
质谱MS
-
碳谱13CNMR
-
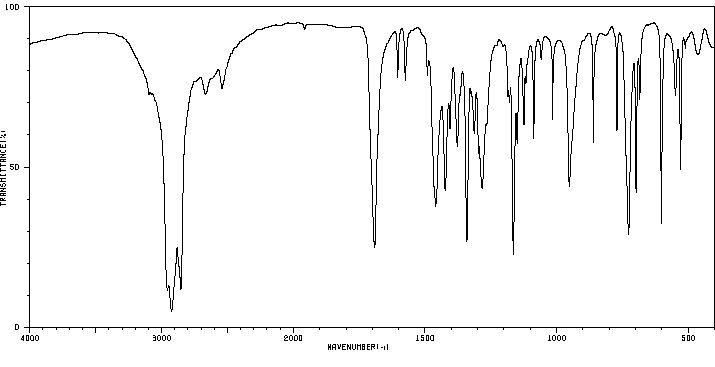
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(βS)-β-氨基-4-(4-羟基苯氧基)-3,5-二碘苯甲丙醇
(S,S)-邻甲苯基-DIPAMP
(S)-(-)-7'-〔4(S)-(苄基)恶唑-2-基]-7-二(3,5-二-叔丁基苯基)膦基-2,2',3,3'-四氢-1,1-螺二氢茚
(S)-盐酸沙丁胺醇
(S)-3-(叔丁基)-4-(2,6-二甲氧基苯基)-2,3-二氢苯并[d][1,3]氧磷杂环戊二烯
(S)-2,2'-双[双(3,5-三氟甲基苯基)膦基]-4,4',6,6'-四甲氧基联苯
(S)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(R)富马酸托特罗定
(R)-(-)-盐酸尼古地平
(R)-(-)-4,12-双(二苯基膦基)[2.2]对环芳烷(1,5环辛二烯)铑(I)四氟硼酸盐
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[((6-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(4-叔丁基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(3-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-4,7-双(3,5-二-叔丁基苯基)膦基-7“-[(吡啶-2-基甲基)氨基]-2,2”,3,3'-四氢1,1'-螺二茚满
(R)-3-(叔丁基)-4-(2,6-二苯氧基苯基)-2,3-二氢苯并[d][1,3]氧杂磷杂环戊烯
(R)-2-[((二苯基膦基)甲基]吡咯烷
(R)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(N-(4-甲氧基苯基)-N-甲基-3-(1-哌啶基)丙-2-烯酰胺)
(5-溴-2-羟基苯基)-4-氯苯甲酮
(5-溴-2-氯苯基)(4-羟基苯基)甲酮
(5-氧代-3-苯基-2,5-二氢-1,2,3,4-oxatriazol-3-鎓)
(4S,5R)-4-甲基-5-苯基-1,2,3-氧代噻唑烷-2,2-二氧化物-3-羧酸叔丁酯
(4S,4''S)-2,2''-亚环戊基双[4,5-二氢-4-(苯甲基)恶唑]
(4-溴苯基)-[2-氟-4-[6-[甲基(丙-2-烯基)氨基]己氧基]苯基]甲酮
(4-丁氧基苯甲基)三苯基溴化磷
(3aR,8aR)-(-)-4,4,8,8-四(3,5-二甲基苯基)四氢-2,2-二甲基-6-苯基-1,3-二氧戊环[4,5-e]二恶唑磷
(3aR,6aS)-5-氧代六氢环戊基[c]吡咯-2(1H)-羧酸酯
(2Z)-3-[[(4-氯苯基)氨基]-2-氰基丙烯酸乙酯
(2S,3S,5S)-5-(叔丁氧基甲酰氨基)-2-(N-5-噻唑基-甲氧羰基)氨基-1,6-二苯基-3-羟基己烷
(2S,2''S,3S,3''S)-3,3''-二叔丁基-4,4''-双(2,6-二甲氧基苯基)-2,2'',3,3''-四氢-2,2''-联苯并[d][1,3]氧杂磷杂戊环
(2S)-(-)-2-{[[[[3,5-双(氟代甲基)苯基]氨基]硫代甲基]氨基}-N-(二苯基甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[((1S,2S)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[[((1R,2R)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2-硝基苯基)磷酸三酰胺
(2,6-二氯苯基)乙酰氯
(2,3-二甲氧基-5-甲基苯基)硼酸
(1S,2S,3S,5S)-5-叠氮基-3-(苯基甲氧基)-2-[(苯基甲氧基)甲基]环戊醇
(1S,2S,3R,5R)-2-(苄氧基)甲基-6-氧杂双环[3.1.0]己-3-醇
(1-(4-氟苯基)环丙基)甲胺盐酸盐
(1-(3-溴苯基)环丁基)甲胺盐酸盐
(1-(2-氯苯基)环丁基)甲胺盐酸盐
(1-(2-氟苯基)环丙基)甲胺盐酸盐
(1-(2,6-二氟苯基)环丙基)甲胺盐酸盐
(-)-去甲基西布曲明
龙蒿油
龙胆酸钠
龙胆酸叔丁酯
龙胆酸
龙胆紫-d6
龙胆紫