4-氯-2-氟-N-乙酰基苯胺 | 59280-70-5
中文名称
4-氯-2-氟-N-乙酰基苯胺
中文别名
4'-氯-2'-氟乙酰苯胺;2-氟-4-氯-N-乙酰苯胺;2-氟-4-氯乙酰苯胺
英文名称
4'-chloro-2'-fluoroacetanilide
英文别名
N-(4-chloro-2-fluorophenyl)acetamide
CAS
59280-70-5
化学式
C8H7ClFNO
mdl
MFCD00042520
分子量
187.601
InChiKey
GVRKNSAEOVXHOS-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:154-156 °C(lit.)
-
沸点:312.8±32.0 °C(Predicted)
-
密度:1.3020 (estimate)
-
稳定性/保质期:
避免接触氧化物
计算性质
-
辛醇/水分配系数(LogP):1.9
-
重原子数:12
-
可旋转键数:1
-
环数:1.0
-
sp3杂化的碳原子比例:0.125
-
拓扑面积:29.1
-
氢给体数:1
-
氢受体数:2
安全信息
-
危险等级:IRRITANT
-
危险品标志:Xi,T
-
危险类别码:R23/24/25
-
WGK Germany:3
-
海关编码:2924299090
-
安全说明:S36/37/39,S45
-
危险性防范说明:P264,P280,P302+P352+P332+P313+P362+P364,P305+P351+P338+P337+P313
-
危险性描述:H315,H319
SDS
| Name: | 4 -Chloro-2 -Fluoroacetanilide 99+%(TLC) Material Safety Data Sheet |
| Synonym: | N-(4-Chloro-2-Fluorophenyl)Acetamid |
| CAS: | 59280-70-5 |
Synonym:N-(4-Chloro-2-Fluorophenyl)Acetamid
Section 2 - COMPOSITION, INFORMATION ON INGREDIENTS
| CAS# | Chemical Name | content | EINECS# |
| 59280-70-5 | 4'-Chloro-2'-Fluoroacetanilide | 99% | unlisted |
Risk Phrases: 23/24/25
Section 3 - HAZARDS IDENTIFICATION
EMERGENCY OVERVIEW
Toxic by inhalation, in contact with skin and if swallowed.
Potential Health Effects
Eye:
May cause eye irritation.
Skin:
May cause skin irritation.
Ingestion:
May cause irritation of the digestive tract. May cause cardiac disturbances. May cause central nervous system depression.
Inhalation:
May cause respiratory tract irritation. May cause cardiac abnormalities. Inhalation at high concentrations may cause CNS depression and asphixiation.
Chronic:
No information found.
Section 4 - FIRST AID MEASURES
Eyes: Immediately flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid.
Skin:
Get medical aid. Flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes. Wash clothing before reuse.
Ingestion:
Never give anything by mouth to an unconscious person. Get medical aid. Do NOT induce vomiting. If conscious and alert, rinse mouth and drink 2-4 cupfuls of milk or water.
Inhalation:
Remove from exposure and move to fresh air immediately. If breathing is difficult, give oxygen. Get medical aid. Do NOT use mouth-to-mouth resuscitation. If breathing has ceased apply artificial respiration using oxygen and a suitable mechanical device such as a bag and a mask.
Notes to Physician:
Section 5 - FIRE FIGHTING MEASURES
General Information:
As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear. During a fire, irritating and highly toxic gases may be generated by thermal decomposition or combustion. Containers may explode when heated. Non-combustible, substance itself does not burn but may decompose upon heating to produce irritating, corrosive and/or toxic fumes. Runoff from fire control or dilution water may cause pollution.
Extinguishing Media:
Use water spray to cool fire-exposed containers. Use agent most appropriate to extinguish fire. For small fires, use dry chemical, carbon dioxide, or water spray. For large fires, use dry chemical, carbon dioxide, alcohol-resistant foam, or water spray.
Section 6 - ACCIDENTAL RELEASE MEASURES
General Information: Use proper personal protective equipment as indicated in Section 8.
Spills/Leaks:
Vacuum or sweep up material and place into a suitable disposal container. Clean up spills immediately, observing precautions in the Protective Equipment section. Avoid generating dusty conditions.
Provide ventilation.
Section 7 - HANDLING and STORAGE
Handling:
Wash thoroughly after handling. Use with adequate ventilation.
Minimize dust generation and accumulation. Avoid contact with eyes, skin, and clothing. Keep container tightly closed. Avoid ingestion and inhalation.
Storage:
Store in a tightly closed container. Store in a cool, dry, well-ventilated area away from incompatible substances.
Section 8 - EXPOSURE CONTROLS, PERSONAL PROTECTION
Engineering Controls:
Facilities storing or utilizing this material should be equipped with an eyewash facility and a safety shower. Use adequate ventilation to keep airborne concentrations low.
Exposure Limits CAS# 59280-70-5: Personal Protective Equipment Eyes: Wear appropriate protective eyeglasses or chemical safety goggles as described by OSHA's eye and face protection regulations in 29 CFR 1910.133 or European Standard EN166.
Skin:
Wear appropriate protective gloves to prevent skin exposure.
Clothing:
Wear appropriate protective clothing to prevent skin exposure.
Respirators:
Follow the OSHA respirator regulations found in 29 CFR 1910.134 or European Standard EN 149. Use a NIOSH/MSHA or European Standard EN 149 approved respirator if exposure limits are exceeded or if irritation or other symptoms are experienced.
Section 9 - PHYSICAL AND CHEMICAL PROPERTIES
Physical State: Crystalline powder
Color: slightly yellow
Odor: None reported.
pH: Not available.
Vapor Pressure: Not available.
Viscosity: Not available.
Boiling Point: Not available.
Freezing/Melting Point: 156.00 - 158.00 deg C
Autoignition Temperature: Not applicable.
Flash Point: Not applicable.
Explosion Limits, lower: Not available.
Explosion Limits, upper: Not available.
Decomposition Temperature:
Solubility in water: insoluble
Specific Gravity/Density:
Molecular Formula: C8H7ClFNO
Molecular Weight: 187.60
Section 10 - STABILITY AND REACTIVITY
Chemical Stability:
Stable under normal temperatures and pressures.
Conditions to Avoid:
Incompatible materials, dust generation, excess heat, strong oxidants.
Incompatibilities with Other Materials:
Oxidizing agents.
Hazardous Decomposition Products:
Hydrogen chloride, nitrogen oxides, carbon monoxide, irritating and toxic fumes and gases, carbon dioxide, hydrogen fluoride gas, nitrogen.
Hazardous Polymerization: Has not been reported.
Section 11 - TOXICOLOGICAL INFORMATION
RTECS#:
CAS# 59280-70-5 unlisted.
LD50/LC50:
Not available.
Carcinogenicity:
4'-Chloro-2'-Fluoroacetanilide - Not listed by ACGIH, IARC, or NTP.
Section 12 - ECOLOGICAL INFORMATION
Section 13 - DISPOSAL CONSIDERATIONS
Dispose of in a manner consistent with federal, state, and local regulations.
Section 14 - TRANSPORT INFORMATION
IATA
Shipping Name: TOXIC SOLID, ORGANIC, N.O.S.*
Hazard Class: 6.1
UN Number: 2811
Packing Group: III
IMO
Shipping Name: TOXIC SOLID, ORGANIC, N.O.S.
Hazard Class: 6.1
UN Number: 2811
Packing Group: III
RID/ADR
Shipping Name: TOXIC SOLID, ORGANIC, N.O.S.
Hazard Class: 6.1
UN Number: 2811
Packing group: III
Section 15 - REGULATORY INFORMATION
European/International Regulations
European Labeling in Accordance with EC Directives
Hazard Symbols: T
Risk Phrases:
R 23/24/25 Toxic by inhalation, in contact with skin
and if swallowed.
Safety Phrases:
S 28A After contact with skin, wash immediately with
plenty of water.
S 36/37/39 Wear suitable protective clothing, gloves
and eye/face protection.
S 37 Wear suitable gloves.
S 45 In case of accident or if you feel unwell, seek
medical advice immediately (show the label where
possible).
WGK (Water Danger/Protection)
CAS# 59280-70-5: No information available.
Canada
None of the chemicals in this product are listed on the DSL/NDSL list.
CAS# 59280-70-5 is not listed on Canada's Ingredient Disclosure List.
US FEDERAL
TSCA
CAS# 59280-70-5 is not listed on the TSCA inventory.
It is for research and development use only.
SECTION 16 - ADDITIONAL INFORMATION
N/A
制备方法与用途
用途:用于制造精细化工中间体。
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 2-氟乙酰苯胺 N-(2-fluorophenyl)acetamide 399-31-5 C8H8FNO 153.156 4-氯-2-氟苯胺 2-fluoro-4-chloroaniline 57946-56-2 C6H5ClFN 145.564 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 —— 5-acetyloamino-2-chloro-4-fluoro-thiophenol 95635-53-3 C8H7ClFNOS 219.667 —— N-(4-chloro-2-fluoro-3-(hydroxymethyl)phenyl)acetamide 1442470-80-5 C9H9ClFNO2 217.627 4-氯-2-氟苯胺 2-fluoro-4-chloroaniline 57946-56-2 C6H5ClFN 145.564 —— N-(4-chloro-2-fluoro-5-nitrophenyl)acetamide 95635-45-3 C8H6ClFN2O3 232.599 —— bis-(5-acetamino-2-chloro-4-fluorobenzene)disulfide 145977-41-9 C16H12Cl2F2N2O2S2 437.318 —— N-(4-chloro-3-(cyanomethyl)-2-fluorophenyl)acetamide 1442470-84-9 C10H8ClFN2O 226.638
反应信息
-
作为反应物:描述:参考文献:名称:Herbicidal aryl triazolinones摘要:公式为##STR1##的除草化合物,其中,例如,X是Br、Cl或F;Y是Cl或Br,R.sup.2是CHF.sub.2,R.sup.3是CH.sub.3,R是较低的烷基,R.sup.1是H、Na、较低的烷基或--SO.sub.2R。公开号:US04818275A1
-
作为产物:参考文献:名称:N-氟苯磺酰亚胺铜催化苯胺区域选择性卤化摘要:已开发出具有优异区域选择性和单取代的铜催化苯胺直接氯化和溴化。该协议提供了在温和条件下使用廉价的 KCl 或 NaBr 作为卤素源从容易获得的芳烃中获得广泛的芳烃氯化物和溴化物的途径。DOI:10.1002/ejoc.202200399
-
作为试剂:描述:、 2-氟乙酰苯胺 、 氯 、 在 4-氯-2-氟-N-乙酰基苯胺 、 ice 、 甲醇 作用下, 以 溶剂黄146 为溶剂, 反应 5.0h, 以to yield 119 parts of 4'-chloro-2'-fluroacetanilide as white crystals melting at 152°-155° C.的产率得到参考文献:名称:Herbicidal 2-substituted aryl-4,5,6,7-tetrahydro-2H-isoindole-1,3-diones摘要:本发明涉及除草的2-取代芳基-4,5,6,7-四氢-2H-异吲哚-1,3-二酮。这些化合物可用于某些作物的选择性除草或全面植被控制。公开号:US04032326A1
文献信息
-
IDO INHIBITORS申请人:BRISTOL-MYERS SQUIBB COMPANY公开号:US20160289171A1公开(公告)日:2016-10-06There are disclosed compounds that modulate or inhibit the enzymatic activity of indoleamine 2,3-dioxygenase (IDO), pharmaceutical compositions containing said compounds and methods of treating proliferative disorders, such as cancer, viral infections and/or inflammatory disorders utilizing the compounds of the invention.已披露的化合物可调节或抑制吲哌酮胺2,3-二氧化酶(IDO)的酶活性,含有该化合物的药物组合物以及利用本发明的化合物治疗增殖性疾病,如癌症、病毒感染和/或炎症性疾病的方法。
-
[EN] CYCLOPENTYLBENZAMIDE DERIVATIVES AND THEIR USE FOR THE TREATMENT OF PSYCHOTIC AND COGNITIVE DISORDERS<br/>[FR] DÉRIVÉS DE CYCLOPENTYLBENZAMIDE ET LEUR UTILISATION POUR LE TRAITEMENT DE TROUBLES PSYCHOTIQUES OU COGNITIFS申请人:TAKEDA CAMBRIDGE LTD公开号:WO2015055994A1公开(公告)日:2015-04-23The present invention provides compounds of formula (I) and pharmaceutically acceptable salts thereof, wherein n, L, X, Ra, Rb, R1, R2 and R3 their preparation, pharmaceutical compositions containing them and their use in therapy.本发明提供了式(I)的化合物及其药用盐,其中n,L,X,Ra,Rb,R1,R2和R3,它们的制备,含有它们的药物组合物以及它们在治疗中的用途。
-
HETEROCYCLIC COMPOUND申请人:Takeda Pharmaceutical Company Limited公开号:US20190169166A1公开(公告)日:2019-06-06Provided is a heterocyclic compound that may have a GCN2 inhibitory action, and is expected to be useful for the prophylaxis or treatment of GCN2 associated diseases including cancer and the like. A compound represented by the formula (I): wherein each symbol is as described in the DESCRIPTION, or a salt thereof.提供的是一种可能具有GCN2抑制作用的杂环化合物,预计可用于预防或治疗与GCN2相关的疾病,包括癌症等。一种由以下式(I)表示的化合物: 其中每个符号如描述中所述,或其盐。
-
Discovery of 3-[(4,5,7-Trifluorobenzothiazol-2-yl)methyl]indole-<i>N</i>-acetic Acid (Lidorestat) and Congeners as Highly Potent and Selective Inhibitors of Aldose Reductase for Treatment of Chronic Diabetic Complications作者:Michael C. Van Zandt、Michael L. Jones、David E. Gunn、Leo S. Geraci、J. Howard Jones、Diane R. Sawicki、Janet Sredy、Jorge L. Jacot、A. Thomas DiCioccio、Tatiana Petrova、Andre Mitschler、Alberto D. PodjarnyDOI:10.1021/jm0492094日期:2005.5.1Recent efforts to identify treatments for chronic diabetic complications have resulted in the discovery of a novel series of highly potent and selective 3-[(benzothiazol-2-yl)methyl]indole-N-alkanoic acid aldose reductase inhibitors. The lead candidate, 3-[(4,5,7-trifluorobenzothiazol-2-yl)methyl]indole-N-acetic acid (lidorestat, 9) inhibits aldose reductase with an IC(50) of 5 nM, while being 5400最近鉴定慢性糖尿病并发症的治疗方法的努力导致发现了一系列新型的高效和选择性的3-[((苯并噻唑-2-基)甲基]吲哚-N-链烷酸醛糖还原酶抑制剂。主要候选化合物3-[((4,5,7-三氟苯并噻唑-2-基)甲基]吲哚-N-乙酸(利多司他,9)抑制醛糖还原酶,IC(50)为5 nM,是5400倍对醛还原酶的活性较低,醛还原酶是一种与活性醛解毒有关的酶。在为期5天的STZ诱导的糖尿病大鼠模型中,它可降低神经和晶状体山梨糖醇的水平,ED(50)分别为1.9和4.5 mg / kg / d po。在3个月的糖尿病干预模型中(糖尿病1个月,随后2个月以5 mg / kg / d po进行药物治疗),相对于糖尿病对照,它可以使多元醇正常化,并减少运动神经传导速度不足59%。它具有良好的药代动力学特征(F,82%; t(1/2),5.6 h; Vd,0.694 L / kg),并且在目标组织中的良好药物渗透
-
N-arylsulfonamide- and pyrrolidinecarboxylic acid intermediates, and their use for the preparation of herbicidal 1,3-dioxo-1h-pyrrolo[1,2-c]imidazole derivatives申请人:Degussa AG公开号:US06384234B1公开(公告)日:2002-05-07This invention is directed to compounds and intermediates of Formulae 6 and 3, useful in preparing herbicidal sulfonamides of Formula 1, including all geometric and stereoisomers thereof, and agricultural salts thereof. wherein the substituents are defined in the specification.这项发明涉及到公式6和3的化合物和中间体,用于制备公式1的除草磺胺类化合物,包括其所有的几何和立体异构体,以及农业盐。其中取代基在说明书中有定义。
表征谱图
-
氢谱1HNMR
-
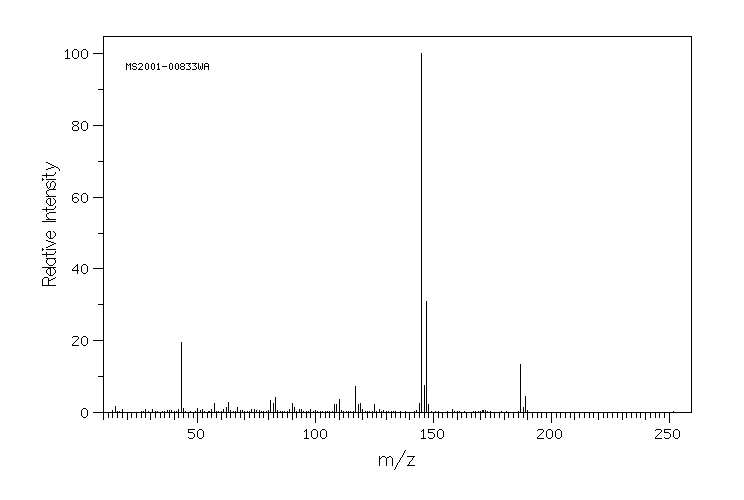
质谱MS
-
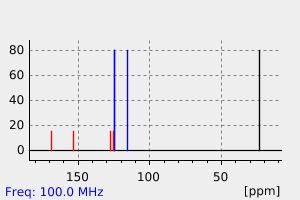
碳谱13CNMR
-
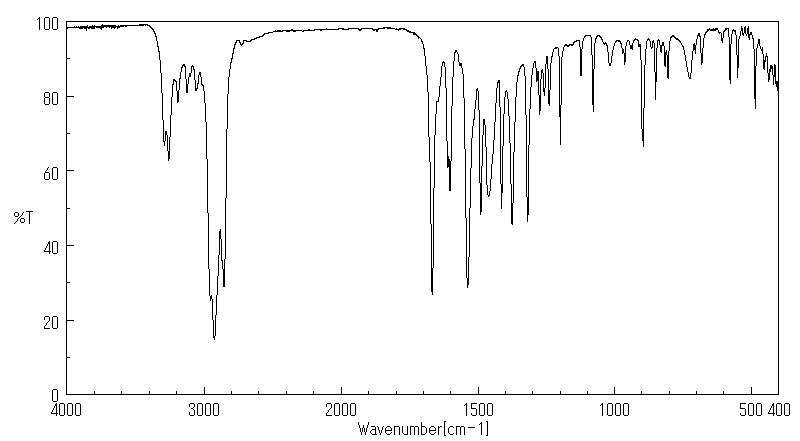
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(βS)-β-氨基-4-(4-羟基苯氧基)-3,5-二碘苯甲丙醇
(S,S)-邻甲苯基-DIPAMP
(S)-(-)-7'-〔4(S)-(苄基)恶唑-2-基]-7-二(3,5-二-叔丁基苯基)膦基-2,2',3,3'-四氢-1,1-螺二氢茚
(S)-盐酸沙丁胺醇
(S)-3-(叔丁基)-4-(2,6-二甲氧基苯基)-2,3-二氢苯并[d][1,3]氧磷杂环戊二烯
(S)-2,2'-双[双(3,5-三氟甲基苯基)膦基]-4,4',6,6'-四甲氧基联苯
(S)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(R)富马酸托特罗定
(R)-(-)-盐酸尼古地平
(R)-(-)-4,12-双(二苯基膦基)[2.2]对环芳烷(1,5环辛二烯)铑(I)四氟硼酸盐
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[((6-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(4-叔丁基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(3-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-4,7-双(3,5-二-叔丁基苯基)膦基-7“-[(吡啶-2-基甲基)氨基]-2,2”,3,3'-四氢1,1'-螺二茚满
(R)-3-(叔丁基)-4-(2,6-二苯氧基苯基)-2,3-二氢苯并[d][1,3]氧杂磷杂环戊烯
(R)-2-[((二苯基膦基)甲基]吡咯烷
(R)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(N-(4-甲氧基苯基)-N-甲基-3-(1-哌啶基)丙-2-烯酰胺)
(5-溴-2-羟基苯基)-4-氯苯甲酮
(5-溴-2-氯苯基)(4-羟基苯基)甲酮
(5-氧代-3-苯基-2,5-二氢-1,2,3,4-oxatriazol-3-鎓)
(4S,5R)-4-甲基-5-苯基-1,2,3-氧代噻唑烷-2,2-二氧化物-3-羧酸叔丁酯
(4S,4''S)-2,2''-亚环戊基双[4,5-二氢-4-(苯甲基)恶唑]
(4-溴苯基)-[2-氟-4-[6-[甲基(丙-2-烯基)氨基]己氧基]苯基]甲酮
(4-丁氧基苯甲基)三苯基溴化磷
(3aR,8aR)-(-)-4,4,8,8-四(3,5-二甲基苯基)四氢-2,2-二甲基-6-苯基-1,3-二氧戊环[4,5-e]二恶唑磷
(3aR,6aS)-5-氧代六氢环戊基[c]吡咯-2(1H)-羧酸酯
(2Z)-3-[[(4-氯苯基)氨基]-2-氰基丙烯酸乙酯
(2S,3S,5S)-5-(叔丁氧基甲酰氨基)-2-(N-5-噻唑基-甲氧羰基)氨基-1,6-二苯基-3-羟基己烷
(2S,2''S,3S,3''S)-3,3''-二叔丁基-4,4''-双(2,6-二甲氧基苯基)-2,2'',3,3''-四氢-2,2''-联苯并[d][1,3]氧杂磷杂戊环
(2S)-(-)-2-{[[[[3,5-双(氟代甲基)苯基]氨基]硫代甲基]氨基}-N-(二苯基甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[((1S,2S)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[[((1R,2R)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2-硝基苯基)磷酸三酰胺
(2,6-二氯苯基)乙酰氯
(2,3-二甲氧基-5-甲基苯基)硼酸
(1S,2S,3S,5S)-5-叠氮基-3-(苯基甲氧基)-2-[(苯基甲氧基)甲基]环戊醇
(1S,2S,3R,5R)-2-(苄氧基)甲基-6-氧杂双环[3.1.0]己-3-醇
(1-(4-氟苯基)环丙基)甲胺盐酸盐
(1-(3-溴苯基)环丁基)甲胺盐酸盐
(1-(2-氯苯基)环丁基)甲胺盐酸盐
(1-(2-氟苯基)环丙基)甲胺盐酸盐
(1-(2,6-二氟苯基)环丙基)甲胺盐酸盐
(-)-去甲基西布曲明
龙蒿油
龙胆酸钠
龙胆酸叔丁酯
龙胆酸
龙胆紫-d6
龙胆紫