3,4-二甲基苯乙酸 | 22618-23-1
中文名称
3,4-二甲基苯乙酸
中文别名
——
英文名称
3,4-dimethylphenyl acetate
英文别名
(3,4-dimethylphenyl) acetate
CAS
22618-23-1
化学式
C10H12O2
mdl
MFCD00225308
分子量
164.204
InChiKey
ZFIOOTIKKICLOR-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:22°C
-
沸点:235°C (estimate)
-
密度:1.0326 (rough estimate)
-
LogP:2.9
计算性质
-
辛醇/水分配系数(LogP):2.9
-
重原子数:12
-
可旋转键数:2
-
环数:1.0
-
sp3杂化的碳原子比例:0.3
-
拓扑面积:26.3
-
氢给体数:0
-
氢受体数:2
安全信息
-
海关编码:2915390090
SDS
上下游信息
反应信息
-
作为反应物:描述:参考文献:名称:Vene,J. et al., Bulletin de la Societe Chimique de France, 1963, p. 1813 - 1817摘要:DOI:
-
作为产物:参考文献:名称:FISCHER, ALFRED;IBRAHIM, PRABHA N., TETRAHEDRON, 46,(1990) N, C. 2737-2746摘要:DOI:
文献信息
-
AlCl<sub>3</sub> · 6H<sub>2</sub>O/KI/CH<sub>3</sub>CN/H<sub>2</sub>O: An Efficient and Versatile System for Chemoselective C–O Bond Cleavage and Formation of Halides and Carbonyl Compounds from Alcohols in Hydrated Media作者:Dilip Konwar、Saikat Das Sharma、Prodip Kumar Gogoi、Pranjal GogoiDOI:10.1080/00397910500516316日期:2006.5versatile system, cleaves the C–O bonds of esters, acetals, ethers, and oxathiolanes to the corresponding acids, alcohols, and carbonyl compounds chemoselectively at 80 °C in hydrated media with good yields. This system also converts the alcohols (primary/secondary) to halides and oxidizes the alcohols (primary/secondary) to the corresponding carbonyl compounds in the presence of DMSO.
-
Highly Efficient and Convenient Methods for the Direct Conversion of Aryl Silyl Ethers and Aryl Acetates into Aryl Alkyl Ethers作者:Takeshi Oriyama、Kojiro Noda、Kaori YatabeDOI:10.1055/s-1997-3267日期:1997.6Direct conversion of aryl trialkylsilyl ethers and aryl acetates into the corresponding alkyl-protected phenols such as benzyl and allyl ethers can be conveniently performed by the reaction with alkyl halide in the presence of cesium fluoride and sodium methoxide, respectively, in a one-pot procedure.
-
N-ACYL ANTHRANILIC ACID DERIVATIVE OR SALT THEREOF申请人:Yokotani Junichi公开号:US20110275797A1公开(公告)日:2011-11-10An N-acyl anthranilic acid derivative represented by general formula (1) or a salt thereof is useful for prevention or treatment of diseases associated with excessive production of collagen. (In the formula, R 1 represents a carboxyl group or the like; R 2 represents a hydrogen atom or the like; R 3 represents an optionally substituted aryl group or the like; X 1 represents a carbonyl group; X 2 represents a bonding hand; X 3 represents a bonding hand; X 4 represents a bonding hand or the like; and A represents an optionally substituted phenyl group or the like.)
-
Synthesis and anti-inflammatory activity of 2-oxo-2H-chromenyl and 2H-chromenyl-5-oxo-2,5-dihydrofuran-3-carboxylates作者:Siva Hariprasad Kurma、Shailaja Karri、Madhusudana Kuncha、Ramakrishna Sistla、China Raju BhimapakaDOI:10.1016/j.bmcl.2020.127341日期:2020.82-oxo-2H-chromenyl-5-oxo-2,5-dihydrofuran-3-carboxylates (5a-n) and 2H-chromenyl-5-oxo-2,5-dihydrofuran-3-carboxylates (8a-p). All the prepared compounds were screened for anti-inflammatory activity. In vitro anti-inflammatory activity data demonstrated that the compounds 5g, 5i, 5k-l and 8f are effective among the tested compounds against TNF-α (1.108 ± 0.002, 0.423 ± 0.022, 0.047 ± 0.001, 0.070 ± 0.0024-氯-2-氧代-2 H-亚甲基-3-甲醛(3a-g)和4-氯-2 H-亚甲基-3-甲醛(7a-h)与活化炔烃(4a-b)的环加成反应提供了2-oxo-2 H -chromenyl-5-oxo-2,5-dihydrofuran-3-羧酸盐(5a-n)和2 H -chromenyl-5-oxo-2,5-dihydrofuran-3-羧酸盐(8a -p)。筛选所有制备的化合物的抗炎活性。体外抗炎活性数据表明化合物5g,5i,5k-1和8f与标准化合物泼尼松龙(0.033±0.002 µM)相比,被测化合物对TNF-α(1.108±0.002,0.423±0.022,0.047±0.001,0.070±0.002和0.142±0.001 µM)有效。根据体外结果,已选择了三种化合物(5i,5k和8f)用于体内实验,在LPS诱导的小鼠模型中,就抗炎活性而言,这些化合物被确定为更好的化合物。化合物5
-
An efficient method to prepare aryl acetates by the carbonylation of aryl methyl ethers or phenols作者:Dejin Zhang、Guoqiang Yang、Junping Xiong、Jia Liu、Xingbang Hu、Zhibing ZhangDOI:10.1039/d0nj05050d日期:——Synthesis of valuable chemicals from lignin based compounds is critical for the application of biomass. Here, we develop a method of preparing aryl acetates by the carbonylation of aryl methyl ethers or phenols under low CO pressure. Good to excellent yields of aryl acetates were obtained using different substrates, and a possible reaction mechanism was proposed by conducting a series of control experiments
表征谱图
-
氢谱1HNMR
-
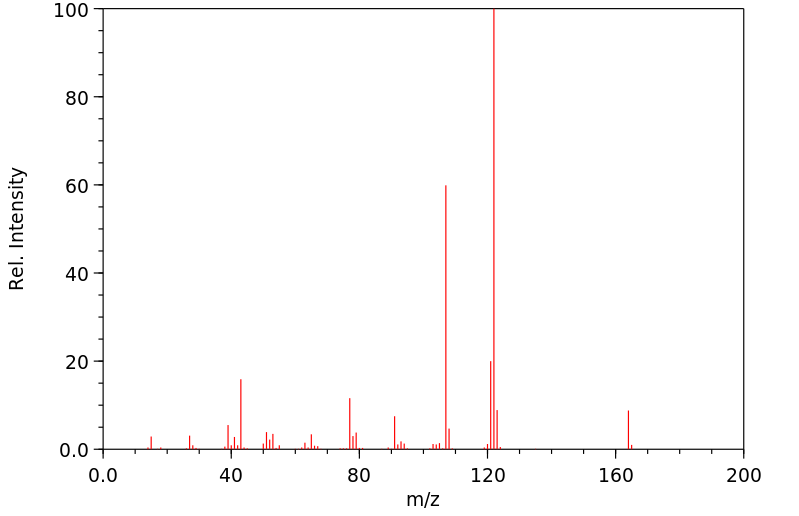
质谱MS
-
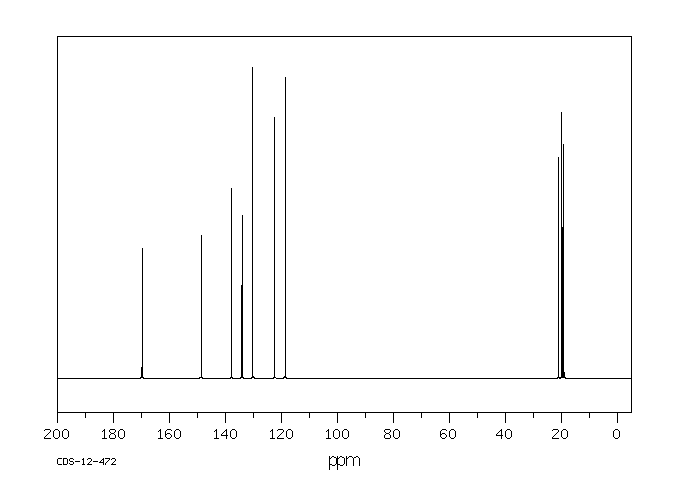
碳谱13CNMR
-
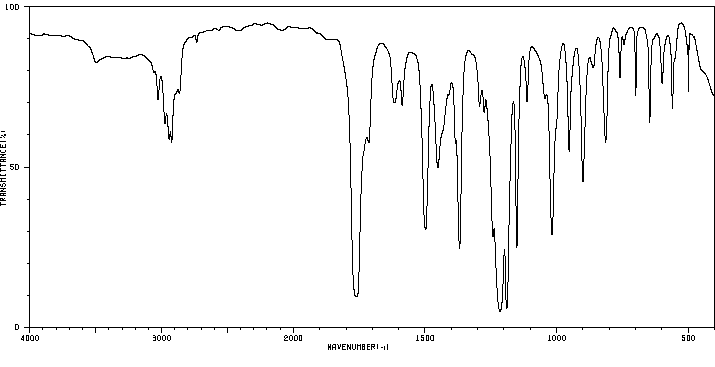
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
马来酰亚胺四聚乙二醇CH2CH2COOPFPESTER
马来酰亚胺六聚乙二醇CH2CH2COOPFPESTER
马来酰亚胺-酰胺-PEG8-四氟苯酚酯
马来酰亚胺-四聚乙二醇-五氟苯酯
马来酰亚胺-三聚乙二醇-五氟苯酚酯
靛酚乙酸酯
阿立哌唑标准品002
间硝基苯基戊酸酯
间氯苯乙酸乙酯
间乙酰苯甲酸
钾4-乙酰氧基苯磺酸酯
酚醛乙酸酯
邻苯二酚二乙酸酯
邻甲苯基环己甲酸酯
邻甲氧基苯乙酸酯
辛酸苯酯
辛酸对甲苯酚酯
辛酸五氯苯基酯
辛酸-(3-氯-苯基酯)
辛酰溴苯腈
苯酰胺,3,4-二(乙酰氧基)-N-[6-氨基-1,2,3,4-四氢-1-(4-甲氧苯基)-3-甲基-2,4-二羰基-5-嘧啶基]-
苯酚-乳酸
苯酚,4-异氰基-,乙酸酯(ester)
苯酚,4-[(四氢-2H-吡喃-2-基)氧代]-,乙酸酯
苯酚,3-(1,1-二甲基乙基)-,乙酸酯
苯酚,2-溴-3-(二溴甲基)-5-甲氧基-,乙酸酯
苯甲醇,4-(乙酰氧基)-3,5-二甲氧基-
苯甲酸,4-(乙酰氧基)-2-氟-
苯氧基氯乙酸苯酯
苯基金刚烷-1-羧酸酯
苯基氰基甲酸酯
苯基庚酸酯
苯基庚-6-炔酸酯
苯基己酸酯
苯基呋喃-2-羧酸酯
苯基吡啶-2-羧酸酯
苯基十一碳-10-烯酸酯
苯基乙醛酸酯
苯基乙酸酯-d5
苯基丙二酸单苯酯
苯基丙-2-炔酸酯
苯基丁-2,3-二烯酸酯
苯基4-乙基环己烷羧酸
苯基3-乙氧基-3-亚氨基丙酸盐
苯基2-(苯磺酰基)乙酸酯
苯基2-(4-甲氧基苯基)乙酸酯
苯基2-(2-甲氧基苯基)乙酸酯
苯基2-(2-甲基苯基)乙酸酯
苯基-乙酸-(2-甲酰基-苯基酯)
苯基-乙酸-(2-环己基-苯基酯)