3-(2,3,4-三甲氧基苯基)丙酸 | 33130-04-0
中文名称
3-(2,3,4-三甲氧基苯基)丙酸
中文别名
——
英文名称
3-(2,3,4-trimethoxyphenyl)propanoic acid
英文别名
——
CAS
33130-04-0
化学式
C12H16O5
mdl
MFCD00002773
分子量
240.256
InChiKey
QOPNYPCVRBRZOP-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:70-73℃
计算性质
-
辛醇/水分配系数(LogP):1.5
-
重原子数:17
-
可旋转键数:6
-
环数:1.0
-
sp3杂化的碳原子比例:0.416
-
拓扑面积:65
-
氢给体数:1
-
氢受体数:5
安全信息
-
海关编码:2918990090
-
危险性防范说明:P233,P260,P261,P264,P271,P280,P302+P352,P304,P304+P340,P305+P351+P338,P312,P321,P332+P313,P337+P313,P340,P362,P403,P403+P233,P405,P501
-
危险性描述:H315,H319,H335
-
储存条件:室温
SDS
| Name: | 3-(2 3 4-TRIMETHOXYPHENYL)PROPIONIC ACID 98+% Material Safety Data Sheet |
| Synonym: | 2,3,4-Trimethoxyhydrocinnamic aci |
| CAS: | 33130-04-0 |
Synonym:2,3,4-Trimethoxyhydrocinnamic aci
Section 2 - COMPOSITION, INFORMATION ON INGREDIENTS
| CAS# | Chemical Name | content | EINECS# |
| 33130-04-0 | 3-(2,3,4-TRIMETHOXYPHENYL)PROPIONIC AC | 251-389-0 |
Risk Phrases: None Listed.
Section 3 - HAZARDS IDENTIFICATION
EMERGENCY OVERVIEW
Not available.
Potential Health Effects
The toxicological properties of this material have not been investigated. Use appropriate procedures to prevent opportunities for direct contact with the skin or eyes and to prevent inhalation.
Section 4 - FIRST AID MEASURES
Eyes: Immediately flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids.
Skin:
Flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes.
Ingestion:
Do NOT induce vomiting. If conscious and alert, rinse mouth and drink 2-4 cupfuls of milk or water.
Inhalation:
Remove from exposure and move to fresh air immediately.
Notes to Physician:
Section 5 - FIRE FIGHTING MEASURES
General Information:
As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear. During a fire, irritating and highly toxic gases may be generated by thermal decomposition or combustion.
Extinguishing Media:
In case of fire, use water, dry chemical, chemical foam, or alcohol-resistant foam.
Section 6 - ACCIDENTAL RELEASE MEASURES
General Information: Use proper personal protective equipment as indicated in Section 8.
Spills/Leaks:
Clean up spills immediately, observing precautions in the Protective Equipment section. Sweep up, then place into a suitable container for disposal.
Section 7 - HANDLING and STORAGE
Handling:
Wash thoroughly after handling. Remove contaminated clothing and wash before reuse. Avoid contact with eyes, skin, and clothing. Avoid ingestion and inhalation.
Storage:
Store in a cool, dry place. Keep container closed when not in use.
Section 8 - EXPOSURE CONTROLS, PERSONAL PROTECTION
Engineering Controls:
Use adequate general or local exhaust ventilation to keep airborne concentrations below the permissible exposure limits. Use process enclosure, local exhaust ventilation, or other engineering controls to control airborne levels.
Exposure Limits CAS# 33130-04-0: Personal Protective Equipment Eyes: Not available.
Skin:
Wear appropriate protective gloves and clothing to prevent skin exposure.
Clothing:
Wear appropriate protective clothing to minimize contact with skin.
Respirators:
Wear a NIOSH/MSHA or European Standard EN 149 approved full-facepiece airline respirator in the positive pressure mode with emergency escape provisions.
Section 9 - PHYSICAL AND CHEMICAL PROPERTIES
Physical State: Not available.
Color: Not available.
Odor: None reported.
pH: Not available.
Vapor Pressure: Not available.
Viscosity: Not available.
Boiling Point: Not available.
Freezing/Melting Point: 70.00 - 73.00 deg C
Autoignition Temperature: Not available.
Flash Point: Not available.
Explosion Limits, lower: Not available.
Explosion Limits, upper: Not available.
Decomposition Temperature:
Solubility in water:
Specific Gravity/Density:
Molecular Formula: C12H16O5
Molecular Weight: 240.26
Section 10 - STABILITY AND REACTIVITY
Chemical Stability:
Stable under normal temperatures and pressures.
Conditions to Avoid:
Incompatible materials, strong oxidants.
Incompatibilities with Other Materials:
Not available.
Hazardous Decomposition Products:
Carbon monoxide, irritating and toxic fumes and gases, carbon dioxide.
Hazardous Polymerization: Has not been reported.
Section 11 - TOXICOLOGICAL INFORMATION
RTECS#:
CAS# 33130-04-0 unlisted.
LD50/LC50:
Not available.
Carcinogenicity:
3-(2,3,4-TRIMETHOXYPHENYL)PROPIONIC ACID, 98+% - Not listed by ACGIH, IARC, or NTP.
Section 12 - ECOLOGICAL INFORMATION
Section 13 - DISPOSAL CONSIDERATIONS
Dispose of in a manner consistent with federal, state, and local regulations.
Section 14 - TRANSPORT INFORMATION
IATA
Not regulated as a hazardous material.
IMO
Not regulated as a hazardous material.
RID/ADR
Not regulated as a hazardous material.
Section 15 - REGULATORY INFORMATION
European/International Regulations
European Labeling in Accordance with EC Directives
Hazard Symbols: Not available.
Risk Phrases:
Safety Phrases:
S 24/25 Avoid contact with skin and eyes.
WGK (Water Danger/Protection)
CAS# 33130-04-0: No information available.
Canada
None of the chemicals in this product are listed on the DSL/NDSL list.
CAS# 33130-04-0 is not listed on Canada's Ingredient Disclosure List.
US FEDERAL
TSCA
CAS# 33130-04-0 is not listed on the TSCA inventory.
It is for research and development use only.
SECTION 16 - ADDITIONAL INFORMATION
N/A
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 2,3,4-三甲氧基苯甲醛 2,3,4-trimethoxybenzaldehyde 2103-57-3 C10H12O4 196.203 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 —— methyl 3-(2,3,4-trimethoxyphenyl)propanoate 76474-95-8 C13H18O5 254.283 —— 3-(2,3,4-trimethoxy-phenyl)-propionic acid ethyl ester 1057673-90-1 C14H20O5 268.31 3-(2,3,4-三甲氧基苯基)-1-丙醇 3-(2,3,4-trimethoxyphenyl)propanol 106800-17-3 C12H18O4 226.273 —— 3-(4-hydroxy-2,3-dimethoxyphenyl)propanol 106800-31-1 C11H16O4 212.246 —— 3-(3-hydroxy-2,4-dimethoxyphenyl)propanol 106800-32-2 C11H16O4 212.246 —— 3-(2-hydroxy-3,4-dimethoxyphenyl)propanol 106800-30-0 C11H16O4 212.246 —— 7,8-dimethoxychroman 106800-38-8 C11H14O3 194.23 —— 5,6,7-trimethoxy-3,4-dihydrocoumarin 67566-08-9 C12H14O5 238.24 2-(2,3,4-三甲氧基苯基)乙胺 2-(2,3,4-trimethoxyphenyl)ethylamine 3937-16-4 C11H17NO3 211.261 4,5,6-三甲氧基-2,3-二氢-1H-茚-1-酮 4,5,6-trimethoxy-2,3-dihydro-1H-inden-1-one 16718-42-6 C12H14O4 222.241 —— {3-[3-(2,3,4-Trimethoxy-phenyl)-propionylamino]-phenyl}-acetic acid methyl ester 864518-28-5 C21H25NO6 387.433 —— 2-bromo-4,5,6-trimethoxy-indan-1-one 111859-48-4 C12H13BrO4 301.137 - 1
- 2
反应信息
-
作为反应物:描述:参考文献:名称:Slotta; Heller, Chemische Berichte, 1930, vol. 63, p. 3029,3043摘要:DOI:
-
作为产物:描述:2,3,4-三甲氧基苯甲醛 在 哌啶 、 吡啶 、 palladium 10% on activated carbon 、 氢气 作用下, 以 乙醇 、 乙酸乙酯 为溶剂, 23.0~115.0 ℃ 、101.33 kPa 条件下, 反应 29.0h, 生成 3-(2,3,4-三甲氧基苯基)丙酸参考文献:名称:首次全合成外消旋螯合酸摘要:外消旋螯合酸全合成的关键步骤是铈催化的β-氧代酯的α-羟基化和氰化物催化的各中间产物环转化为δ-内酯结构。DOI:10.1002/ejoc.202101508
文献信息
-
[EN] BROMODOMAIN INHIBITORS<br/>[FR] INHIBITEURS DE BROMODOMAINE申请人:ABBVIE INC公开号:WO2014206150A1公开(公告)日:2014-12-31The present invention provides for compounds of formula (I). wherein Rx, Ry, Rx1, L1, G1, A1, A2, A3, and A4 have any of the values defined in the specification, and pharmaceutically acceptable salts thereof, that are useful as agents in the treatment of diseases and conditions, including inflammatory diseases, cancer, and AIDS. Also provided are pharmaceutical compositions comprised of one or more compounds of formula (I).本发明提供了式(I)的化合物。其中Rx、Ry、Rx1、L1、G1、A1、A2、A3和A4具有规范中定义的任何值,并且其药学上可接受的盐,可用作治疗疾病和病症的药剂,包括炎症性疾病、癌症和艾滋病。还提供了由一种或多种式(I)的化合物组成的药物组合物。
-
TUBULIN BINDING AGENTS申请人:The Provost, Fellows, Foundation Scholars, & the other Members of Board, of the College of the Holy公开号:US20150018566A1公开(公告)日:2015-01-15The invention provides combretastatin A-4 like compounds that are modified to have enhanced tubulin binding activity and in some embodiments the ability to promote accumulation in the vasculature undergoing angiogenesis (homing activity). The compounds are based on the combretastatin A-4 skeletal structure having a tubulin-binding pharmacophore comprising two fused rings (A and B rings) in which the B ring is substituted with (a) an aromatic ring structure (C ring) and (b) a second substituent/functional group that comes off the B ring. The aromatic ring structure is typically a six membered ring phenolic or aniline structure, or may also be a fused ring structure such as a substituted or unsubstituted naphthalene. The second substituent on the B ring may for example be a substituent which has been found to provide enhanced tubulin binding activity (for example a carbonyl group), or may be a substituent that facilitates functionalisation of the B ring (for example an hydroxyl or amine group), or it may be a binding agent for a target that is preferentially expressed on vasculature undergoing angiogenesis, and not expressed on quiescent vasculature.该发明提供了类似于康柏他定A-4的化合物,经过改良以增强微管结合活性,在某些实施例中还具有促进在进行血管生成的血管系统中积聚(归巢活性)的能力。这些化合物基于康柏他定A-4的骨架结构,其具有一个微管结合药效团,包括两个融合环(A环和B环),其中B环被取代为(a)芳香环结构(C环)和(b)从B环上脱落的第二取代基/官能团。芳香环结构通常是一个六元环酚或苯胺结构,也可以是一个融合环结构,如取代或未取代的萘。B环上的第二取代基可以是已知可提供增强微管结合活性的取代基(例如酰基),也可以是促进B环官能化的取代基(例如羟基或胺基),或者可以是一种针对在进行血管生成的血管系统上优先表达而在静止血管系统上不表达的靶标的结合剂。
-
Synthesis, biological evaluation and mechanism study of chalcone analogues as novel anti-cancer agents作者:Jie Chen、Jun Yan、Jinhui Hu、Yanqing Pang、Ling Huang、Xingshu LiDOI:10.1039/c5ra14888j日期:——A series of novel chalcone analogues were designed, synthesized and evaluated as anticancer agents. The results of antiproliferative activity tests showed that most of the analogues exhibited moderate to very good antiproliferative activities with GI50 values in the micromol to sub-micromol range. Especially compound 10a gave 0.026 μM to 0.035 μM GI50 for five cancer cell lines. The mechanistic studies
-
Efficient Method for Preparing Functionalized Benzosuberenes申请人:Pinney Kevin G.公开号:US20120130129A1公开(公告)日:2012-05-24The disclosed process can efficiently synthesize functionalized benzosuberenes. The process provides an improved method of production of benzosuberene and compounds containing a benzosuberene moiety, which is characterized by a ring closing methodology comprising reaction of a 5-phenylpentanoic acid with Eaton's reagent to form the benzosuberone. The process, optionally, further includes steps for adding a functional group at the ketone position.
-
[EN] BENZODIAZEPINONES AS FAK INHIBITORS FOR TREATMENT OF CANCER<br/>[FR] BENZODIAZÉPINONES À TITRE D'INHIBITEURS DE FAK POUR LE TRAITEMENT DU CANCER申请人:ABBOTT LAB公开号:WO2012045194A1公开(公告)日:2012-04-12Disclosed are compounds which inhibit the activity of focal adhesion kinase, compositions containing the compounds, and methods of treating diseases during which focal adhesion kinase is expressed.揭示了抑制细胞焦点粘附激酶活性的化合物,包含这些化合物的组合物,以及治疗焦点粘附激酶表达相关疾病的方法。
表征谱图
-
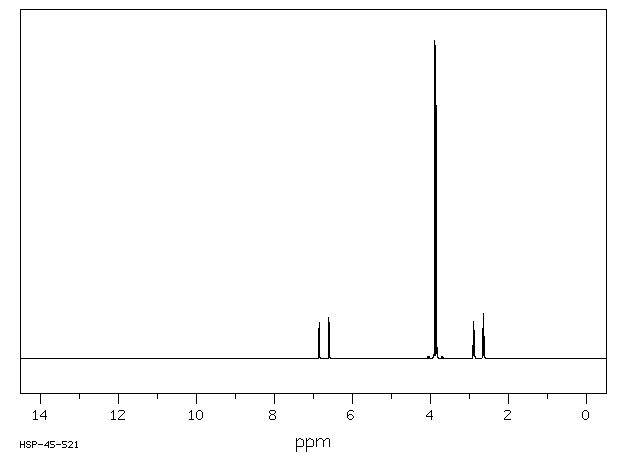
氢谱1HNMR
-
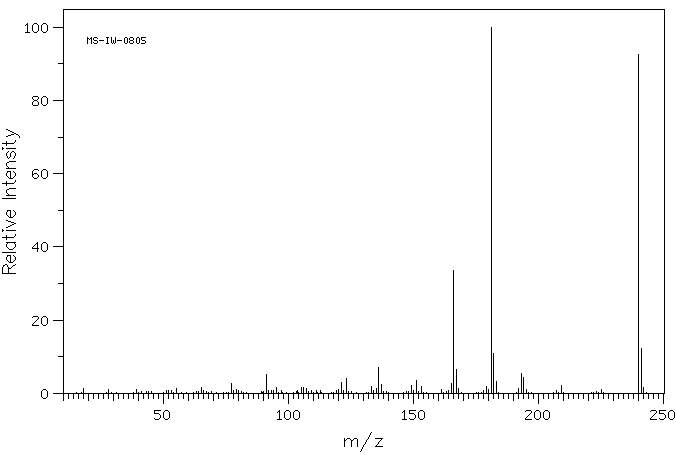
质谱MS
-
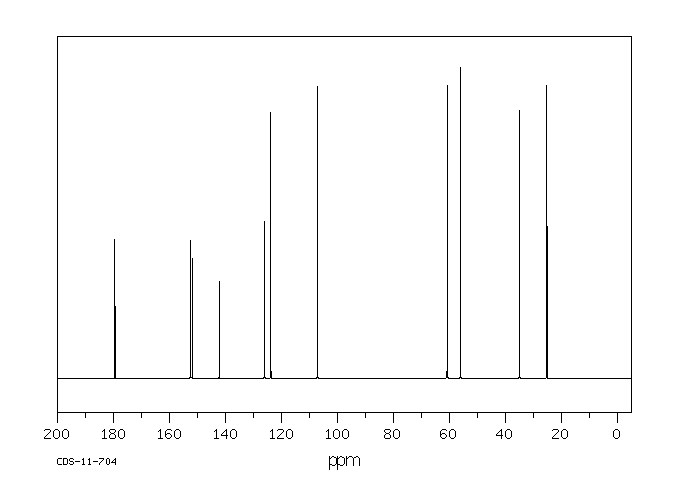
碳谱13CNMR
-
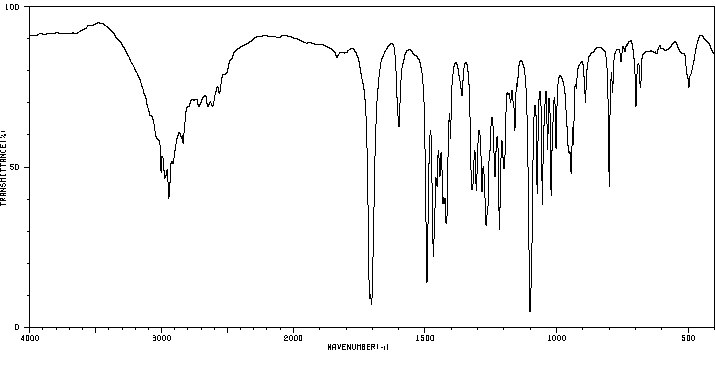
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
限制性核酸内切酶TAQⅠ(TTHHB8I)
阿明洛芬
阿拉洛芬
铁-N-(3-苯基戊二酰)去铁敏B
钙二[(2R)-2-羟基-3-苯丙酸酯]
酮洛芬相关物质C
酪泮酸钠
酪氨酸,3-羟基-b-亚甲基-
苯基丙酮酸缩氨基脲
苯基丁二酸
苯乙酸,a-甲基-4-(4,5,6,7-四氢-2-苯并噻唑基)-
苯丙酸钠盐
苯丙酸钙盐(2:1)
苯丙酸,加合N-环己基并环己胺(1:1)
苯丙酸,b-[[(苯基氨基)羰基]氨基]-
苯丙酸,b-[[(二乙胺基)硫代甲基]硫代]-
苯丙酸,a-[2-[甲基[2-(4-吗啉基)乙基]氨基]-2-羰基乙基]-,(R)-
苯丙酸,a-[(乙酰基硫代)甲基]-,(S)-
苯丙酸,4-羟基-b,2,6-三甲基-,(bR)-
苯丙酸,4-氯-a-(肟基)-
苯丙酸,3-硝基-b-(三氯甲锗烷基)-
苯丙酸,3-氯-a-羟基-
苯丙酸 羟基-4-甲氧基
苄氧羰基-DL-beta-苯丙氨酸
苄基马来酸
苄基丙二酸单酰肼
苄基丙二酸
苄基丁酸
艾司洛尔酸钠
艾司洛尔酸
胆影脒
羧基布洛芬
羟基布洛芬
美索洛芬
米格列奈
米格列奈
碘芬酸
碘番酸
碘泊酸钠
碘泊酸钙
硬脂酰胺丙基鲸蜡硬脂基二甲基铵甲苯磺酸盐
番石榴酸
甲酪氨酸
甲基多巴杂质A
甲基多巴EP杂质B
甲基多巴
甲基3-(4-苄氧基-2-甲基-苯基)丙酸酯
消旋甲酪氨酸
消旋布洛芬赖氨酸盐
消旋卡多曲二元酸杂质