2-甲氧基-1-萘腈 | 16000-39-8
中文名称
2-甲氧基-1-萘腈
中文别名
2-甲氧基-1-萘甲腈
英文名称
1-cyano-2-methoxynaphthalene
英文别名
2-methoxy-1-naphthonitrile;2-methoxynaphthalene-1-carbonitrile
CAS
16000-39-8
化学式
C12H9NO
mdl
——
分子量
183.21
InChiKey
KPIZWRFKCSLGQK-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:95-96 °C(lit.)
-
沸点:316.88°C (rough estimate)
-
密度:1.1261 (rough estimate)
-
稳定性/保质期:
遵照规定使用和储存,则不会分解。
计算性质
-
辛醇/水分配系数(LogP):2.9
-
重原子数:14
-
可旋转键数:1
-
环数:2.0
-
sp3杂化的碳原子比例:0.08
-
拓扑面积:33
-
氢给体数:0
-
氢受体数:2
安全信息
-
危险品标志:Xn
-
安全说明:S36
-
危险类别码:R20/21/22
-
WGK Germany:3
-
海关编码:2926909090
-
储存条件:存放于阴凉干燥处。
SDS
| Name: | 2-Methoxy-1-Naphthonitrile Material Safety Data Sheet |
| Synonym: | None known |
| CAS: | 16000-39-8 |
Synonym:None known
Section 2 - COMPOSITION, INFORMATION ON INGREDIENTS
| CAS# | Chemical Name | content | EINECS# |
| 16000-39-8 | 2-Methoxy-1-Naphthonitrile | ca 100 | 240-133-3 |
Risk Phrases: 20/21
Section 3 - HAZARDS IDENTIFICATION
EMERGENCY OVERVIEW
Harmful by inhalation and in contact with skin.The toxicological properties of this material have not been fully investigated.
Potential Health Effects
Eye:
May cause eye irritation.
Skin:
May cause skin irritation.
Ingestion:
May cause irritation of the digestive tract. The toxicological properties of this substance have not been fully investigated.
Inhalation:
May cause respiratory tract irritation. The toxicological properties of this substance have not been fully investigated.
Chronic:
No information found.
Section 4 - FIRST AID MEASURES
Eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid.
Skin:
Get medical aid. Flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes. Wash clothing before reuse.
Ingestion:
Never give anything by mouth to an unconscious person. Get medical aid. Do NOT induce vomiting. If conscious and alert, rinse mouth and drink 2-4 cupfuls of milk or water.
Inhalation:
Remove from exposure and move to fresh air immediately. If not breathing, give artificial respiration. If breathing is difficult, give oxygen. Get medical aid.
Notes to Physician:
Section 5 - FIRE FIGHTING MEASURES
General Information:
As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear. During a fire, irritating and highly toxic gases may be generated by thermal decomposition or combustion.
Extinguishing Media:
Use agent most appropriate to extinguish fire. Use water spray, dry chemical, carbon dioxide, or appropriate foam.
Section 6 - ACCIDENTAL RELEASE MEASURES
General Information: Use proper personal protective equipment as indicated in Section 8.
Spills/Leaks:
Vacuum or sweep up material and place into a suitable disposal container. Clean up spills immediately, observing precautions in the Protective Equipment section. Avoid generating dusty conditions.
Provide ventilation.
Section 7 - HANDLING and STORAGE
Handling:
Wash thoroughly after handling. Use with adequate ventilation.
Minimize dust generation and accumulation. Avoid contact with eyes, skin, and clothing. Keep container tightly closed. Avoid ingestion and inhalation.
Storage:
Store in a tightly closed container. Store in a cool, dry, well-ventilated area away from incompatible substances.
Section 8 - EXPOSURE CONTROLS, PERSONAL PROTECTION
Engineering Controls:
Facilities storing or utilizing this material should be equipped with an eyewash facility and a safety shower. Use adequate ventilation to keep airborne concentrations low.
Exposure Limits CAS# 16000-39-8: Personal Protective Equipment Eyes: Wear appropriate protective eyeglasses or chemical safety goggles as described by OSHA's eye and face protection regulations in 29 CFR 1910.133 or European Standard EN166.
Skin:
Wear appropriate protective gloves to prevent skin exposure.
Clothing:
Wear appropriate protective clothing to prevent skin exposure.
Respirators:
A respiratory protection program that meets OSHA's 29 CFR 1910.134 and ANSI Z88.2 requirements or European Standard EN 149 must be followed whenever workplace conditions warrant respirator use.
Section 9 - PHYSICAL AND CHEMICAL PROPERTIES
Physical State: Powder
Color: light tan
Odor: Not available.
pH: Not available.
Vapor Pressure: Not available.
Viscosity: Not available.
Boiling Point: Not available.
Freezing/Melting Point: 95.00 - 96.00 deg C
Autoignition Temperature: Not applicable.
Flash Point: Not applicable.
Explosion Limits, lower: Not available.
Explosion Limits, upper: Not available.
Decomposition Temperature:
Solubility in water:
Specific Gravity/Density:
Molecular Formula: C12H9NO
Molecular Weight: 183.21
Section 10 - STABILITY AND REACTIVITY
Chemical Stability:
Stable at room temperature in closed containers under normal storage and handling conditions.
Conditions to Avoid:
Incompatible materials, dust generation, excess heat.
Incompatibilities with Other Materials:
Strong acids, strong bases, strong oxidizing agents.
Hazardous Decomposition Products:
Carbon monoxide, oxides of nitrogen, irritating and toxic fumes and gases, carbon dioxide.
Hazardous Polymerization: Has not been reported
Section 11 - TOXICOLOGICAL INFORMATION
RTECS#:
CAS# 16000-39-8 unlisted.
LD50/LC50:
Not available.
Carcinogenicity:
2-Methoxy-1-Naphthonitrile - Not listed by ACGIH, IARC, or NTP.
Section 12 - ECOLOGICAL INFORMATION
Section 13 - DISPOSAL CONSIDERATIONS
Dispose of in a manner consistent with federal, state, and local regulations.
Section 14 - TRANSPORT INFORMATION
IATA
Shipping Name: TOXIC SOLID, ORGANIC, N.O.S.*
Hazard Class: 6.1
UN Number: 2811
Packing Group: III
IMO
Shipping Name: TOXIC SOLID, ORGANIC, N.O.S.
Hazard Class: 6.1
UN Number: 2811
Packing Group: III
RID/ADR
Shipping Name: TOXIC SOLID, ORGANIC, N.O.S.
Hazard Class: 6.1
UN Number: 2811
Packing group: III
Section 15 - REGULATORY INFORMATION
European/International Regulations
European Labeling in Accordance with EC Directives
Hazard Symbols: XN
Risk Phrases:
R 20/21 Harmful by inhalation and in contact with
skin.
Safety Phrases:
S 23 Do not inhale gas/fumes/vapour/spray.
S 28A After contact with skin, wash immediately with
plenty of water.
S 37 Wear suitable gloves.
S 45 In case of accident or if you feel unwell, seek
medical advice immediately (show the label where
possible).
WGK (Water Danger/Protection)
CAS# 16000-39-8: No information available.
Canada
None of the chemicals in this product are listed on the DSL/NDSL list.
CAS# 16000-39-8 is not listed on Canada's Ingredient Disclosure List.
US FEDERAL
TSCA
CAS# 16000-39-8 is not listed on the TSCA inventory.
It is for research and development use only.
SECTION 16 - ADDITIONAL INFORMATION
N/A
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 —— 1-cyano-2-hydroxynaphthalene 52805-47-7 C11H7NO 169.183 2-甲氧基-1-萘醛 2-methoxy-1-naphthaldehyde 5392-12-1 C12H10O2 186.21 —— 2-methoxy-1-naphthaldehydeoxime 99806-91-4 C12H11NO2 201.225 2-甲氧基-1-萘甲酸 2-methoxy-1-naphthoic acid 947-62-6 C12H10O3 202.21 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 —— 1-cyano-2-hydroxynaphthalene 52805-47-7 C11H7NO 169.183 2-甲氧基-1-甲基萘 2-methoxy-1-methylnaphthalene 1130-80-9 C12H12O 172.227 —— 2-Methoxy-naphthoesaeure-(1)-amid 876483-32-8 C12H11NO2 201.225
反应信息
-
作为反应物:描述:2-甲氧基-1-萘腈 在 18-冠醚-6 、 potassium tert-butylate 、 水 作用下, 以 1,4-二氧六环 为溶剂, 反应 16.0h, 以91%的产率得到1-cyano-2-hydroxynaphthalene参考文献:名称:通过C–OMe键裂解实现芳基甲基醚衍生物的无金属醚化摘要:为在温和的反应条件下通过无金属的C-OMe键裂解合成芳基烷基醚,开发了一种通用而有效的方案。该方法显示出广泛的甲氧基芳烃和醇,包括伯,仲和叔醇,以及天然产物,药物和生物活性醇。DFT计算和实验结果同时证实,钾离子通过与腈的结合在甲氧基的活化中起关键作用,并为S N Ar机制提供了支持。DOI:10.1021/acs.orglett.8b01696
-
作为产物:描述:参考文献:名称:使用固体负载的肼将醛完全转化为腈摘要:聚合物负载的肼试剂已被应用于将一系列醛类转化为腈类,为组合化学程序提供了一条清洁、有效的途径,以构建更加多样化的构建模块。DOI:10.1055/s-2002-25333
文献信息
-
Direct C–H Cyanation of Arenes via Organic Photoredox Catalysis作者:Joshua B. McManus、David A. NicewiczDOI:10.1021/jacs.6b12708日期:2017.3.1direct C-H functionalization of aromatic compounds are in demand for a variety of applications, including the synthesis of agrochemicals, pharmaceuticals, and materials. Herein, we disclose the construction of aromatic nitriles via direct C-H functionalization using an acridinium photoredox catalyst and trimethylsilyl cyanide under an aerobic atmosphere. The reaction proceeds at room temperature under
-
Phosphination of Phenol Derivatives and Applications to Divergent Synthesis of Phosphine Ligands作者:Chenchen Li、Kezhuo Zhang、Minghao Zhang、Wu Zhang、Wanxiang ZhaoDOI:10.1021/acs.orglett.1c03227日期:2021.11.19describe a general and efficient protocol for the synthesis of organophosphine compounds from phenols and phosphines (R2PH) via a metal-free C–O bond cleavage and C–P bond formation process. This approach exhibits broad substrate scope and excellent functional group tolerance. The synthetic utilities of this protocol were demonstrated by the synthesis of chiral ligands via the various transformations
-
Lewis Acid‐Mediated Cyanation of Phenols Using <i>N</i> ‐Cyano‐ <i>N</i> ‐phenyl‐ <i>p‐</i> toluenesulfonamide作者:Wu Zhang、Tao Li、Qingli Wang、Wanxiang ZhaoDOI:10.1002/adsc.201900813日期:2019.11.5Lewis acid‐mediated cyanation of phenol derivatives with N‐cyano‐N‐phenyl‐p‐toluenesulfonamide (NCTS) has been developed. The reaction proceeded efficiently with high regioselectivity to produce aromatic nitriles in moderate to excellent yields, which provides a direct and practical access to valuable products.
-
Azabicyclic compounds for the treatment of disease申请人:——公开号:US20030236270A1公开(公告)日:2003-12-25The invention provides compounds of Formula I: 1 wherein Azabicyclo is 2 W is a six-membered heterocyclic ring system having 1-2 nitrogen atoms or a 10-membered bicyclic-six-six-fused-ring system having up to two nitrogen atoms within either or both rings, provided that no nitrogen is at a bridge of the bicyclic-six-six-fused-ring system, and further having 1-2 substitutents independently selected from R 3 . These compounds may be in the form of pharmaceutical salts or compositions, may be in pure enantiomeric form or racemic mixtures, and are useful in pharmaceuticals to treat diseases or conditions in which &agr;7 is known to be involved.本发明提供了式I的化合物: 1 其中Azabicyclo是 2 W是一个六元杂环体系,具有1-2个氮原子,或一个10元双环六六并环体系,在任一或两个环中最多有两个氮原子,前提是该双环六六并环体系的桥上没有氮原子,并且还具有1-2个独立选自R 3 的取代基。 这些化合物可以是药物盐或组合物,可以是纯净的对映异构体形式或外消旋混合物,并且在制药中用于治疗那些已知涉及α7的疾病或状况。
-
Metal-Free One-Pot Conversion of Electron-Rich Aromatics into Aromatic Nitriles作者:Hideo Togo、Sousuke UshijimaDOI:10.1055/s-0029-1219575日期:2010.4Various electron-rich aromatics could be smoothly converted into the corresponding aromatic nitriles in good to moderate yields by treatment of electron-rich aromatics with POCl3 and DMF, followed by treatment with molecular iodine in aqueous ammonia. The present reaction is a novel metal-free one-pot method for the preparation of aromatic nitriles from electron-rich aromatics.
表征谱图
-
氢谱1HNMR
-
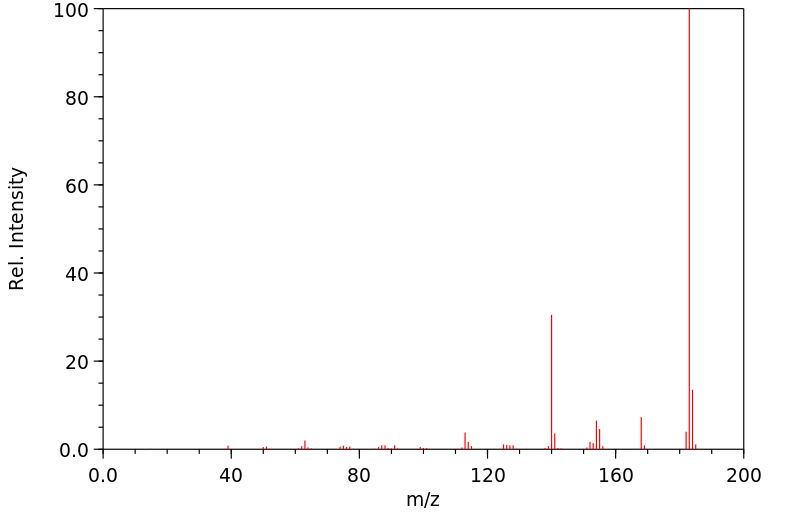
质谱MS
-
碳谱13CNMR
-
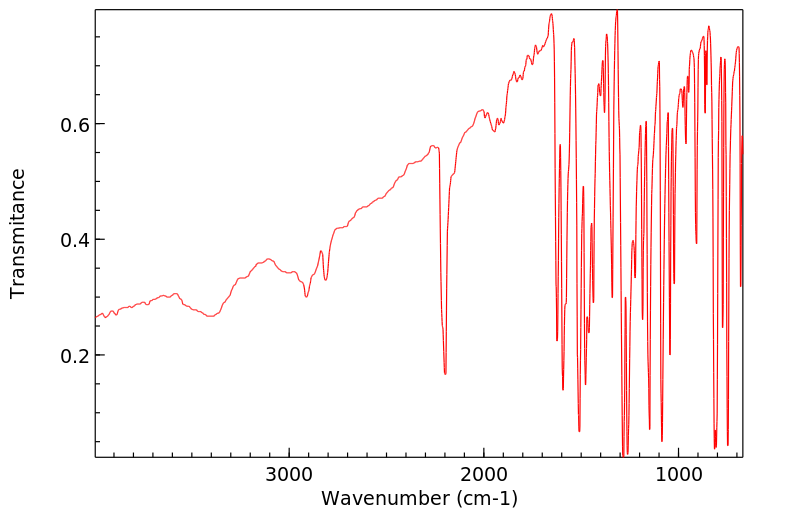
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(S)-溴烯醇内酯
(R)-3,3''-双([[1,1''-联苯]-4-基)-[1,1''-联萘]-2,2''-二醇
(3S,3aR)-2-(3-氯-4-氰基苯基)-3-环戊基-3,3a,4,5-四氢-2H-苯并[g]吲唑-7-羧酸
(3R,3’’R,4S,4’’S,11bS,11’’bS)-(+)-4,4’’-二叔丁基-4,4’’,5,5’’-四氢-3,3’’-联-3H-二萘酚[2,1-c:1’’,2’’-e]膦(S)-BINAPINE
(11bS)-2,6-双(3,5-二甲基苯基)-4-羟基-4-氧化物-萘并[2,1-d:1'',2''-f][1,3,2]二氧磷
(11bS)-2,6-双(3,5-二氯苯基)-4羟基-4-氧-二萘并[2,1-d:1'',2''-f][1,3,2]二氧磷杂七环
(11bR)-2,6-双[3,5-双(1,1-二甲基乙基)苯基]-4-羟基-4-氧化物-二萘并[2,1-d:1'',2''-f][1,3,2]二氧杂磷平
黄胺酸
马兜铃对酮
马休黄钠盐一水合物
马休黄
食品黄6号
食品红40铝盐色淀
飞龙掌血香豆醌
颜料黄101
颜料红70
颜料红63
颜料红53:3
颜料红5
颜料红48单钠盐
颜料红48:2
颜料红4
颜料红261
颜料红258
颜料红220
颜料红22
颜料红214
颜料红2
颜料红19
颜料红185
颜料红184
颜料红170
颜料红148
颜料红147
颜料红146
颜料红119
颜料红114
颜料红 9
颜料红 21
颜料橙7
颜料橙46
颜料橙38
颜料橙3
颜料橙22
颜料橙2
颜料橙17
颜料橙 5
颜料棕1
顺式-阿托伐醌-d5
雄甾烷-3,17-二酮