3,5-二叔丁基-4-羟基苯甲酸十六酯 | 67845-93-6
中文名称
3,5-二叔丁基-4-羟基苯甲酸十六酯
中文别名
紫外吸收剂UV-2908;3,5-二(1,1-二甲基乙基)-4-羟基苯甲酸十六酯;3,5-二叔丁基-4-羟基苯甲酸十六烷基酯;3,5-二叔丁基-4-羟基苯甲酸正十六酯
英文名称
n-hexadecyl 3,5-di-tert-butyl-4-hydroxybenzoate
英文别名
3,5-di-tert-butyl-4-hydroxybenzoic acid n-hexadecyl ester;hexadecyl 3,5-di-tert-butyl-4-hydroxybenzoate;cyasorb 2908;n-hexadecyl 3,5-di-tert.-butyl-4-hydroxybenzoate;hexadecyl 3,5-di-tert-butyl-4-hydroxy-benzoate;hexadecyl 3,5-di-tert.-butyl-4-hydroxybenzoate;hexadecyl 3,5-ditert-butyl-4-hydroxybenzoate
CAS
67845-93-6
化学式
C31H54O3
mdl
MFCD00180757
分子量
474.768
InChiKey
NZYMWGXNIUZYRC-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:59-61 °C(lit.)
-
沸点:539.2±50.0 °C(Predicted)
-
密度:0.936±0.06 g/cm3(Predicted)
-
溶解度:可溶于DMSO(少许)、乙酸乙酯(少许)
-
LogP:12.332
-
物理描述:DryPowder; DryPowder, PelletsLargeCrystals
计算性质
-
辛醇/水分配系数(LogP):12.5
-
重原子数:34
-
可旋转键数:19
-
环数:1.0
-
sp3杂化的碳原子比例:0.774
-
拓扑面积:46.5
-
氢给体数:1
-
氢受体数:3
安全信息
-
危险品标志:Xi
-
危险类别码:R36/37/38
-
WGK Germany:3
-
RTECS号:DG4436220
-
海关编码:2918290000
-
安全说明:S26,S36
-
危险性防范说明:P261,P305+P351+P338
-
危险性描述:H302,H315,H319,H335
-
储存条件:储存条件:室温、密封、干燥
SDS
Material Safety Data Sheet
Section 1. Identification of the substance
Product Name: 3,5-Di-tert-butyl-4-hydroxybenzoic acid hexadecyl ester
Synonyms:
Section 2. Hazards identification
Harmful by inhalation, in contact with skin, and if swallowed.
Section 3. Composition/information on ingredients.
Ingredient name: 3,5-Di-tert-butyl-4-hydroxybenzoic acid hexadecyl ester
CAS number: 67845-93-6
Section 4. First aid measures
Skin contact: Immediately wash skin with copious amounts of water for at least 15 minutes while removing
contaminated clothing and shoes. If irritation persists, seek medical attention.
Eye contact: Immediately wash skin with copious amounts of water for at least 15 minutes. Assure adequate
flushing of the eyes by separating the eyelids with fingers. If irritation persists, seek medical
attention.
Inhalation: Remove to fresh air. In severe cases or if symptoms persist, seek medical attention.
Ingestion: Wash out mouth with copious amounts of water for at least 15 minutes. Seek medical attention.
Section 5. Fire fighting measures
In the event of a fire involving this material, alone or in combination with other materials, use dry
powder or carbon dioxide extinguishers. Protective clothing and self-contained breathing apparatus
should be worn.
Section 6. Accidental release measures
Personal precautions: Wear suitable personal protective equipment which performs satisfactorily and meets local/state/national
standards.
Respiratory precaution: Wear approved mask/respirator
Hand precaution: Wear suitable gloves/gauntlets
Skin protection: Wear suitable protective clothing
Eye protection: Wear suitable eye protection
Methods for cleaning up: Mix with sand or similar inert absorbent material, sweep up and keep in a tightly closed container
for disposal. See section 12.
Environmental precautions: Do not allow material to enter drains or water courses.
Section 7. Handling and storage
Handling: This product should be handled only by, or under the close supervision of, those properly qualified
in the handling and use of potentially hazardous chemicals, who should take into account the fire,
health and chemical hazard data given on this sheet.
Store in closed vessels.
Storage:
Section 8. Exposure Controls / Personal protection
Engineering Controls: Use only in a chemical fume hood.
Personal protective equipment: Wear laboratory clothing, chemical-resistant gloves and safety goggles.
General hydiene measures: Wash thoroughly after handling. Wash contaminated clothing before reuse.
Section 9. Physical and chemical properties
Appearance: Not specified
Boiling point: No data
No data
Melting point:
Flash point: No data
Density: No data
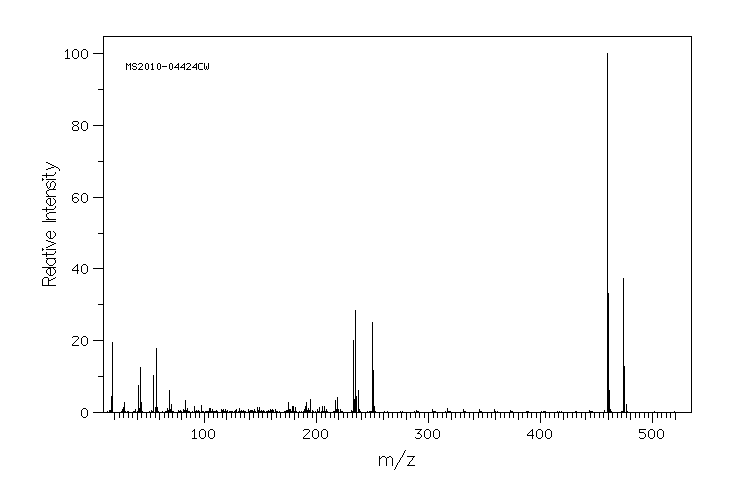
Molecular formula: C31H54O3
Molecular weight: 474.8
Section 10. Stability and reactivity
Conditions to avoid: Heat, flames and sparks.
Materials to avoid: Oxidizing agents.
Possible hazardous combustion products: Carbon monoxide.
Section 11. Toxicological information
No data.
Section 12. Ecological information
No data.
Section 13. Disposal consideration
Arrange disposal as special waste, by licensed disposal company, in consultation with local waste
disposal authority, in accordance with national and regional regulations.
Section 14. Transportation information
Non-harzardous for air and ground transportation.
Section 15. Regulatory information
No chemicals in this material are subject to the reporting requirements of SARA Title III, Section
302, or have known CAS numbers that exceed the threshold reporting levels established by SARA
Title III, Section 313.
SECTION 16 - ADDITIONAL INFORMATION
N/A
Section 1. Identification of the substance
Product Name: 3,5-Di-tert-butyl-4-hydroxybenzoic acid hexadecyl ester
Synonyms:
Section 2. Hazards identification
Harmful by inhalation, in contact with skin, and if swallowed.
Section 3. Composition/information on ingredients.
Ingredient name: 3,5-Di-tert-butyl-4-hydroxybenzoic acid hexadecyl ester
CAS number: 67845-93-6
Section 4. First aid measures
Skin contact: Immediately wash skin with copious amounts of water for at least 15 minutes while removing
contaminated clothing and shoes. If irritation persists, seek medical attention.
Eye contact: Immediately wash skin with copious amounts of water for at least 15 minutes. Assure adequate
flushing of the eyes by separating the eyelids with fingers. If irritation persists, seek medical
attention.
Inhalation: Remove to fresh air. In severe cases or if symptoms persist, seek medical attention.
Ingestion: Wash out mouth with copious amounts of water for at least 15 minutes. Seek medical attention.
Section 5. Fire fighting measures
In the event of a fire involving this material, alone or in combination with other materials, use dry
powder or carbon dioxide extinguishers. Protective clothing and self-contained breathing apparatus
should be worn.
Section 6. Accidental release measures
Personal precautions: Wear suitable personal protective equipment which performs satisfactorily and meets local/state/national
standards.
Respiratory precaution: Wear approved mask/respirator
Hand precaution: Wear suitable gloves/gauntlets
Skin protection: Wear suitable protective clothing
Eye protection: Wear suitable eye protection
Methods for cleaning up: Mix with sand or similar inert absorbent material, sweep up and keep in a tightly closed container
for disposal. See section 12.
Environmental precautions: Do not allow material to enter drains or water courses.
Section 7. Handling and storage
Handling: This product should be handled only by, or under the close supervision of, those properly qualified
in the handling and use of potentially hazardous chemicals, who should take into account the fire,
health and chemical hazard data given on this sheet.
Store in closed vessels.
Storage:
Section 8. Exposure Controls / Personal protection
Engineering Controls: Use only in a chemical fume hood.
Personal protective equipment: Wear laboratory clothing, chemical-resistant gloves and safety goggles.
General hydiene measures: Wash thoroughly after handling. Wash contaminated clothing before reuse.
Section 9. Physical and chemical properties
Appearance: Not specified
Boiling point: No data
No data
Melting point:
Flash point: No data
Density: No data
Molecular formula: C31H54O3
Molecular weight: 474.8
Section 10. Stability and reactivity
Conditions to avoid: Heat, flames and sparks.
Materials to avoid: Oxidizing agents.
Possible hazardous combustion products: Carbon monoxide.
Section 11. Toxicological information
No data.
Section 12. Ecological information
No data.
Section 13. Disposal consideration
Arrange disposal as special waste, by licensed disposal company, in consultation with local waste
disposal authority, in accordance with national and regional regulations.
Section 14. Transportation information
Non-harzardous for air and ground transportation.
Section 15. Regulatory information
No chemicals in this material are subject to the reporting requirements of SARA Title III, Section
302, or have known CAS numbers that exceed the threshold reporting levels established by SARA
Title III, Section 313.
SECTION 16 - ADDITIONAL INFORMATION
N/A
制备方法与用途
紫外线吸收剂
紫外线吸收剂 UV-2908
UV-2908 是一种优良的光稳定剂,挥发性低、着色少且化学稳定性好,耐酸碱性强。它适用于 PP 和 PE 等聚烯烃塑料,通常用量为 0.25% 至 0.1%,与紫外线吸收剂、受阻胺光稳定剂及硫酯类物质具有良好的相容性。根据 FDA 批准,其在聚烯烃中的最大使用量可达 0.5%。
用途紫外线吸收剂 UV-2908
UV-2908 属于受阻羟基苯甲酸酯类光稳定剂,特别适用于保护聚烯烃、聚碳酸酯以及含有阻燃剂和硫酯的体系。它可以单独使用,也可以与其他类型的紫外线稳定剂或抗氧化剂结合使用,以产生良好的协同效应。建议其用量在 0.25% 至 1% 之间,具体根据最终应用的基材及性能要求而定。
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 3,5-二叔丁基-4-羟基苯甲酸 3,5-Di-tert-butyl-4-hydroxybenzoic acid 1421-49-4 C15H22O3 250.338
反应信息
-
作为产物:描述:1-十六烷醇 、 3,5-二叔丁基-4-羟基苯甲酸 反应 5.0h, 以92.5%的产率得到3,5-二叔丁基-4-羟基苯甲酸十六酯参考文献:名称:酸性离子液体催化合成3,5-二叔丁基-4-羟基 苯甲酸正十六酯的方法摘要:本发明公开了一种酸性离子液体催化合成3,5‑二叔丁基‑4‑羟基苯甲酸正十六酯的方法,属于化工领域,以解决现有方法工艺流程复杂,且后处理麻烦,对环境造成污染的问题。本发明方法利用酸性双核功能化离子液体作为催化剂,以3,5‑二叔丁基‑4‑羟基苯甲酸和正十六醇作为原料,利用抽真空脱水,控制反应温度100~160℃,反应时间3~8小时,反应结束后,降温,静置分层,下层为离子液体,分离后重复使用;上层倒入甲醇溶液中,出现大量结晶,过滤得滤饼,即为目标产物。本发明以离子液体作为一种绿色催化剂,在很多酸催化反应中表现出高效、高选择性、反应条件温和、简单可控、环境友好、催化效率高,后处理简单等优点。公开号:CN108218699B
文献信息
-
OXIME ESTER PHOTOINITIATORS申请人:BASF SE公开号:US20180208583A1公开(公告)日:2018-07-26Compounds of the formulae (I) or (II) wherein X is A is O, S, NR 5 or CR 16 R 17 ; R 1 is for example hydrogen or C 1 -C 20 alkyl R 2 is for example hydrogen, C 1 -C 20 alkyl or C 6 -C 20 aryl R 5 for example is C 1 -C 20 alkyl; R 7 , R 8 , R 9 , R 10 and R 11 for example independently of each other are hydrogen. C 1 -C 20 alkyl, halogen, CN or NO 2 ; Ar 1 is for example unsubstituted or substituted C 6 -C 20 aryl, C 3 -C 20 heteroaryl, C 6 -C 20 aroyl, C 3 -C 20 heteroarylcarbonyl or or Ar 1 is Ar 2 is for example phenylene, all of which are unsubstituted or substituted M is for example unsubstituted or substituted C 1 -C 20 alkylene Y is a direct bond, O, S, NR 5 or CO; Z 1 is for example O or S; Z 2 is a direct bond, O, S or NR 5 ; and Q is CO or a direct bond.公式(I)或(II)的化合物 其中 X是 A是O, S, NR 5 或CR 16 R 17 ; R 1 例如是氢或C 1 -C 20 烷基 R 2 例如是氢, C 1 -C 20 烷基或C 6 -C 20 芳基 R 5 例如是C 1 -C 20 烷基; R 7 , R 8 , R 9 , R 10 和R 11 例如彼此独立是氢. C 1 -C 20 烷基, 卤素, CN或NO 2 ; Ar 1 例如是不取代或取代的C 6 -C 20 芳基, C 3 -C 20 杂芳基, C 6 -C 20 芳酰基, C 3 -C 20 杂芳基甲酰基或 或Ar 1 是 Ar 2 例如是苯基, 所有这些都不取代或取代 M例如是不取代或取代的C 1 -C 20 亚烷基 Y是直接键, O, S, NR 5 或CO; Z 1 例如是O或S; Z 2 是直接键, O, S或NR 5 ; 和 Q是CO或直接键。
-
[EN] STERICALLY HINDERED AMINE STABILIZERS<br/>[FR] STABILISANTS D'AMINES À ENCOMBREMENT STÉRIQUE申请人:BASF SE公开号:WO2010142572A1公开(公告)日:2010-12-16The instant invention pertains to hindered amine compounds having at least two nitrogen atoms with different basicity. One part is substituted on the N-atom by alkoxy moieties and the other part is substituted on the N-atom by a hydroxy-alkyl moiety. These materials are particularly effective in stabilizing polymers, especially thermoplastic polyolefins, against the deleterious effects of oxidative, thermal and actinic radiation. The compounds are also particularly effective in stabilizing acid catalyzed and ambient cured coatings systems.这项瞬时发明涉及具有至少两个氮原子且碱度不同的受阻胺化合物。其中一部分在N原子上被烷氧基团取代,另一部分在N原子上被一个羟基-烷基基团取代。这些材料在稳定聚合物方面特别有效,尤其是热塑性聚烯烃,可以抵御氧化、热和光辐射的有害影响。这些化合物在稳定酸催化和环境固化的涂层系统方面也特别有效。
-
[EN] LIQUID PHOSPHITE COMPOSITION DERIVED FROM META-CRESOLS<br/>[FR] COMPOSITION LIQUIDE DE PHOSPHITES ISSUE DE MÉTA-CRÉSOLS申请人:CHEMTURA CORP公开号:WO2011014211A1公开(公告)日:2011-02-03A composition at least two different phosphites, one of which is derived from an alkylated m-cresol, wherein the composition is a liquid at ambient conditions. The other phosphites may be derived from alkylated cresol, alkylated phenol or other alkylated hydroxyaryl compounds. The cresol may be mono-alkylated or di-alkylated with a C1-C18 alkyl group.
-
4-Formyl amino-n-methylpiperidine derivatives, the use thereof as stabilisers and organic material stabilised therewith申请人:——公开号:US20030083406A1公开(公告)日:2003-05-01The present invention relates to 4-formylamino-N-methylpiperidine derivatives of the formula (I) 1 where the variables are as defined in the Description, to a process for preparing these piperidine derivatives, to the use of these piperidine derivatives of the invention, or prepared according to the invention, for stabilizing organic material, in particular for stabilizing plastics or coating materials, and also to the use of these piperidine derivatives of the invention, or prepared according to the invention, as light stabilizers or stabilizers for wood surfaces. The present invention further relates to stabilized organic material which comprises these piperidine derivatives of the invention or prepared according to the invention.
-
[EN] OXIME ESTER PHOTOINITIATORS<br/>[FR] PHOTOAMORCEURS À BASE D'ESTER D'OXIME申请人:BASF SE公开号:WO2015036910A1公开(公告)日:2015-03-19Disclosed are oxime ester compounds which have specific benzo (unsaturated 5-membered ring)-carbonyl group and their use as photoinitiators in photopolymerizable compositions, in particular in photoresist formulations for display applications, e.g. liquid crystal display (LCD), organic light emitting diode (OLED) and touch panel.
表征谱图
-
氢谱1HNMR
-
质谱MS
-
碳谱13CNMR
-
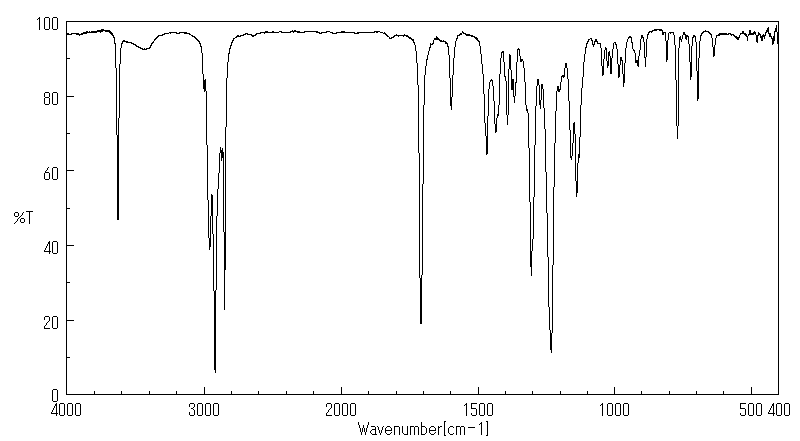
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(βS)-β-氨基-4-(4-羟基苯氧基)-3,5-二碘苯甲丙醇
(S,S)-邻甲苯基-DIPAMP
(S)-(-)-7'-〔4(S)-(苄基)恶唑-2-基]-7-二(3,5-二-叔丁基苯基)膦基-2,2',3,3'-四氢-1,1-螺二氢茚
(S)-盐酸沙丁胺醇
(S)-3-(叔丁基)-4-(2,6-二甲氧基苯基)-2,3-二氢苯并[d][1,3]氧磷杂环戊二烯
(S)-2,2'-双[双(3,5-三氟甲基苯基)膦基]-4,4',6,6'-四甲氧基联苯
(S)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(R)富马酸托特罗定
(R)-(-)-盐酸尼古地平
(R)-(-)-4,12-双(二苯基膦基)[2.2]对环芳烷(1,5环辛二烯)铑(I)四氟硼酸盐
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[((6-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(4-叔丁基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(3-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-4,7-双(3,5-二-叔丁基苯基)膦基-7“-[(吡啶-2-基甲基)氨基]-2,2”,3,3'-四氢1,1'-螺二茚满
(R)-3-(叔丁基)-4-(2,6-二苯氧基苯基)-2,3-二氢苯并[d][1,3]氧杂磷杂环戊烯
(R)-2-[((二苯基膦基)甲基]吡咯烷
(R)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(N-(4-甲氧基苯基)-N-甲基-3-(1-哌啶基)丙-2-烯酰胺)
(5-溴-2-羟基苯基)-4-氯苯甲酮
(5-溴-2-氯苯基)(4-羟基苯基)甲酮
(5-氧代-3-苯基-2,5-二氢-1,2,3,4-oxatriazol-3-鎓)
(4S,5R)-4-甲基-5-苯基-1,2,3-氧代噻唑烷-2,2-二氧化物-3-羧酸叔丁酯
(4S,4''S)-2,2''-亚环戊基双[4,5-二氢-4-(苯甲基)恶唑]
(4-溴苯基)-[2-氟-4-[6-[甲基(丙-2-烯基)氨基]己氧基]苯基]甲酮
(4-丁氧基苯甲基)三苯基溴化磷
(3aR,8aR)-(-)-4,4,8,8-四(3,5-二甲基苯基)四氢-2,2-二甲基-6-苯基-1,3-二氧戊环[4,5-e]二恶唑磷
(3aR,6aS)-5-氧代六氢环戊基[c]吡咯-2(1H)-羧酸酯
(2Z)-3-[[(4-氯苯基)氨基]-2-氰基丙烯酸乙酯
(2S,3S,5S)-5-(叔丁氧基甲酰氨基)-2-(N-5-噻唑基-甲氧羰基)氨基-1,6-二苯基-3-羟基己烷
(2S,2''S,3S,3''S)-3,3''-二叔丁基-4,4''-双(2,6-二甲氧基苯基)-2,2'',3,3''-四氢-2,2''-联苯并[d][1,3]氧杂磷杂戊环
(2S)-(-)-2-{[[[[3,5-双(氟代甲基)苯基]氨基]硫代甲基]氨基}-N-(二苯基甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[((1S,2S)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[[((1R,2R)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2-硝基苯基)磷酸三酰胺
(2,6-二氯苯基)乙酰氯
(2,3-二甲氧基-5-甲基苯基)硼酸
(1S,2S,3S,5S)-5-叠氮基-3-(苯基甲氧基)-2-[(苯基甲氧基)甲基]环戊醇
(1S,2S,3R,5R)-2-(苄氧基)甲基-6-氧杂双环[3.1.0]己-3-醇
(1-(4-氟苯基)环丙基)甲胺盐酸盐
(1-(3-溴苯基)环丁基)甲胺盐酸盐
(1-(2-氯苯基)环丁基)甲胺盐酸盐
(1-(2-氟苯基)环丙基)甲胺盐酸盐
(1-(2,6-二氟苯基)环丙基)甲胺盐酸盐
(-)-去甲基西布曲明
龙蒿油
龙胆酸钠
龙胆酸叔丁酯
龙胆酸
龙胆紫-d6
龙胆紫