P-丁基氧基苯甲醛 | 50262-49-2
中文名称
P-丁基氧基苯甲醛
中文别名
——
英文名称
4-(n-Butyryloxy)benzaldehyd
英文别名
4-ethylacetoxy benzaldehyde;4-formylphenyl butyrate;4-butyroxybenzaldehyde;Butanoic acid, 4-formylphenyl ester;(4-formylphenyl) butanoate
CAS
50262-49-2
化学式
C11H12O3
mdl
MFCD00021058
分子量
192.214
InChiKey
DAFTVVBQFUITOL-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
LogP:2.284 (est)
计算性质
-
辛醇/水分配系数(LogP):2
-
重原子数:14
-
可旋转键数:5
-
环数:1.0
-
sp3杂化的碳原子比例:0.272
-
拓扑面积:43.4
-
氢给体数:0
-
氢受体数:3
SDS
上下游信息
-
下游产品
中文名称 英文名称 CAS号 化学式 分子量 —— 4-(hydroxymethyl)phenyl butyrate —— C11H14O3 194.23
反应信息
-
作为反应物:参考文献:名称:Formulating a new basis for the treatment against botulinum neurotoxin intoxication: 3,4-Diaminopyridine prodrug design and characterization摘要:Botulism is a disease characterized by neuromuscular paralysis and is produced from botulinum neurotoxins (BoNTs) found within the Gram positive bacterium Clostridium botulinum. This bacteria produces the most deadliest toxin known, with lethal doses as low as 1 ng/kg. Due to the relative ease of production and transport, the use of these agents as potential bioterrorist weapons has become of utmost concern. No small molecule therapies against BoNT intoxication have been approved to date. However, 3,4-diaminopyridine (3,4-DAP), a potent reversible inhibitor of voltage-gated potassium channels, is an effective cholinergic agonist used in the treatment of neuromuscular degenerative disorders that require cholinergic enhancement. 3,4-DAP has also been shown to facilitate recovery of neuromuscular action potential post botulinum intoxication by blocking K(+) channels. Unfortunately, 3,4-DAP displays toxicity largely due to blood-brain-barrier (BBB) penetration. As a dual-action prodrug approach to cholinergic enhancement we have designed carbamate and amide conjugates of 3,4-DAP. The carbamate prodrug is intended to be a slowly reversible inhibitor of acetylcholinesterase (AChE) along the lines of the stigmines thereby allowing increased persistence of released acetylcholine within the synaptic cleft. As a secondary activity, cleavage of the carbamate prodrug by AChE will afford the localized release of 3,4-DAP, which in turn, will enhance the pre-synaptic release of additional acetylcholine. Being a competitive inhibitor with respect to acetylcholine, the activity of the prodrug will be greatest at the synaptic junctions most depleted of acetylcholine. Here we report upon the synthesis and biochemical characterization of three new classes of prodrugs intended to limit previously reported stability and toxicity issues. Of the prodrugs examined, compound 32, demonstrated the most clinically relevant half-life of 2.76 h, while selectively inhibiting AChE over butyrylcholinesterase-a plasma-based high activity esterase. Future in vivo studies could provide validation of prodrug 32 as a potential treatment against BoNT intoxication as well as other neuromuscular disorders. (C) 2011 Elsevier Ltd. All rights reserved.DOI:10.1016/j.bmc.2011.09.019
-
作为产物:参考文献:名称:Formulating a new basis for the treatment against botulinum neurotoxin intoxication: 3,4-Diaminopyridine prodrug design and characterization摘要:Botulism is a disease characterized by neuromuscular paralysis and is produced from botulinum neurotoxins (BoNTs) found within the Gram positive bacterium Clostridium botulinum. This bacteria produces the most deadliest toxin known, with lethal doses as low as 1 ng/kg. Due to the relative ease of production and transport, the use of these agents as potential bioterrorist weapons has become of utmost concern. No small molecule therapies against BoNT intoxication have been approved to date. However, 3,4-diaminopyridine (3,4-DAP), a potent reversible inhibitor of voltage-gated potassium channels, is an effective cholinergic agonist used in the treatment of neuromuscular degenerative disorders that require cholinergic enhancement. 3,4-DAP has also been shown to facilitate recovery of neuromuscular action potential post botulinum intoxication by blocking K(+) channels. Unfortunately, 3,4-DAP displays toxicity largely due to blood-brain-barrier (BBB) penetration. As a dual-action prodrug approach to cholinergic enhancement we have designed carbamate and amide conjugates of 3,4-DAP. The carbamate prodrug is intended to be a slowly reversible inhibitor of acetylcholinesterase (AChE) along the lines of the stigmines thereby allowing increased persistence of released acetylcholine within the synaptic cleft. As a secondary activity, cleavage of the carbamate prodrug by AChE will afford the localized release of 3,4-DAP, which in turn, will enhance the pre-synaptic release of additional acetylcholine. Being a competitive inhibitor with respect to acetylcholine, the activity of the prodrug will be greatest at the synaptic junctions most depleted of acetylcholine. Here we report upon the synthesis and biochemical characterization of three new classes of prodrugs intended to limit previously reported stability and toxicity issues. Of the prodrugs examined, compound 32, demonstrated the most clinically relevant half-life of 2.76 h, while selectively inhibiting AChE over butyrylcholinesterase-a plasma-based high activity esterase. Future in vivo studies could provide validation of prodrug 32 as a potential treatment against BoNT intoxication as well as other neuromuscular disorders. (C) 2011 Elsevier Ltd. All rights reserved.DOI:10.1016/j.bmc.2011.09.019
文献信息
-
BISPHOSPHONATE COMPOUNDS申请人:Ebetino Frank Hallock公开号:US20110098251A1公开(公告)日:2011-04-28Novel bisphosphonate cyclic acetal compounds are disclosed, as well as methods of preparing the compounds, pharmaceutical compositions including the compounds, and administration of the compounds in methods of treating bone metabolism disorders, such as abnormal calcium and phosphate metabolism.
-
[2+2] Photodimerization of Stilbazoles Promoted by Oxalic Acid in Suspension作者:Thanh Binh Nguyen、Tuan Minh Nguyen、Pascal RetailleauDOI:10.1002/chem.201905597日期:2020.4.9waxy or even insoluble stilbazoles. Moreover, the oxalic acid loading could be lowered to sub-stoichiometric amounts. When further optimizations were needed, our strategy was found to be highly flexible to identify other oligocarboxylic acids as alternative additive to improve or even overturn regioselectivity. Oxalic acid and other oligocarboxylic acids were found to be capable of orienting more than
-
TEMPLATED MOLECULES AND METHODS FOR USING SUCH MOLECULES申请人:NUEVOLUTION A/S公开号:US20150315567A1公开(公告)日:2015-11-05The present invention relates to a method for synthesising templated molecules. In one aspect of the invention, the templated molecules are linked to the template which templated the synthesis thereof. The intion allows the generation of libraries which can be screened for e.g. therapeutic activity.本发明涉及一种合成模板分子的方法。在本发明的一个方面中,模板分子与模板连接在一起,从而模板化其合成。该方法允许生成图书馆,可用于筛选例如治疗活性。
-
Preparation of amino acids from unsaturated hydantoins申请人:STAUFFER CHEMICAL COMPANY公开号:EP0177072A2公开(公告)日:1986-04-09Amino acids can be easily prepared by reducing unsaturated hydantoins to the corresponding saturated hydantoins by hydrogenating the unsaturated hydantoin using either Raney Nickel catalyst in the presence of more than a stoichiometric amount of caustic or by using zinc and hydrochloric acid followed by hydrolyzing the resultant composition with at least 3 molar equivalents of an alkali metal hydroxide to produce a racemate of an alpha amino acid. The amino acid in suitable derivative form can then be resolved particularly using a two-phase solvent system. The residual isomer of the amino acid remaining after the resolution process can then be racemized using either pyridoxal-5-phosphate or an aliphatic acid in combination with an aldehyde or a ketone. By these procedures, it is possible to obtain high yields of amino acids.
-
Promotion of raney nickel catalyst申请人:STAUFFER CHEMICAL COMPANY公开号:EP0210683A1公开(公告)日:1987-02-04There is disclosed a number of processes for the promotion of the Raney Nickel catalyzed hydrogenation of carbon-carbon double bonds. (a) One process uses tertiary amines to promote the Raney Nickel catalyzed hydrogenation. (b) Another process uses acetylene and acetylene derivatives to promote the Raney Nickel catalyzed hydrogenation. The promotion of Raney Nickel catalyst is particularly suited for the reduction of unsaturated hydantoins to saturated hydantoins and also for the reduction of cyclic and acyclic olefins and diolefins to the corresponding cyclic and acyclic alkanes.
表征谱图
-
氢谱1HNMR
-
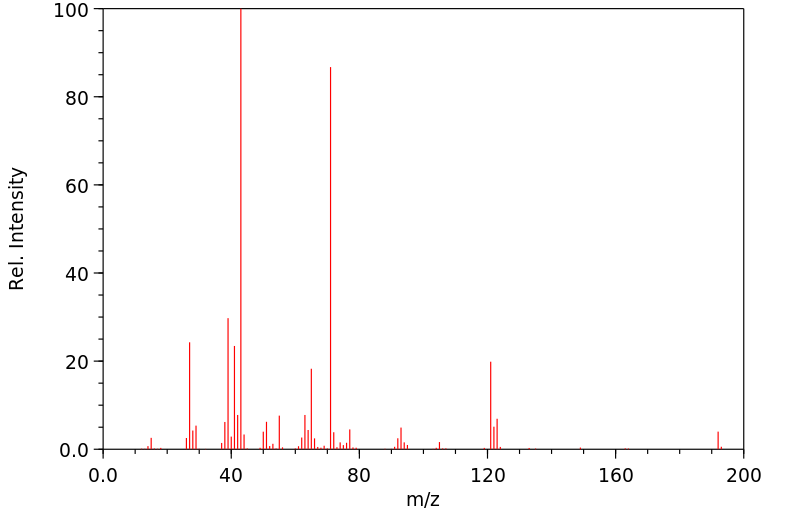
质谱MS
-
碳谱13CNMR
-
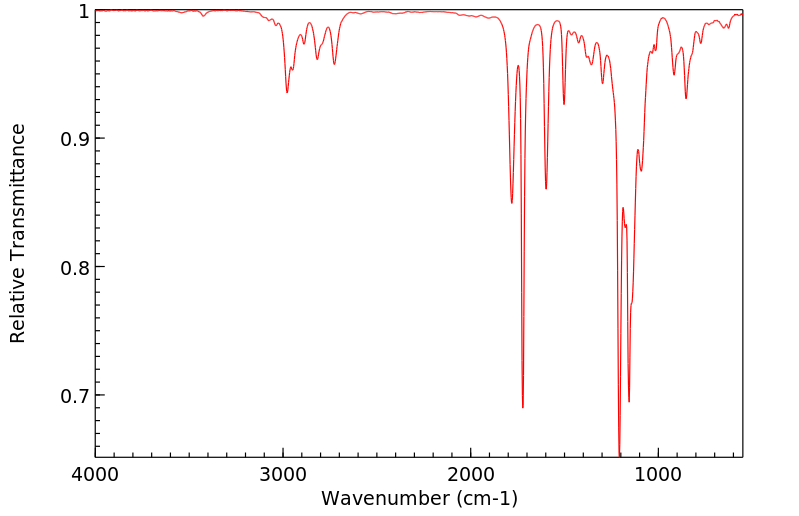
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
马来酰亚胺四聚乙二醇CH2CH2COOPFPESTER
马来酰亚胺六聚乙二醇CH2CH2COOPFPESTER
马来酰亚胺-酰胺-PEG8-四氟苯酚酯
马来酰亚胺-四聚乙二醇-五氟苯酯
马来酰亚胺-三聚乙二醇-五氟苯酚酯
靛酚乙酸酯
阿立哌唑标准品002
间硝基苯基戊酸酯
间氯苯乙酸乙酯
间乙酰苯甲酸
钾4-乙酰氧基苯磺酸酯
酚醛乙酸酯
邻苯二酚二乙酸酯
邻甲苯基环己甲酸酯
邻甲氧基苯乙酸酯
辛酸苯酯
辛酸对甲苯酚酯
辛酸五氯苯基酯
辛酸-(3-氯-苯基酯)
辛酰溴苯腈
苯酰胺,3,4-二(乙酰氧基)-N-[6-氨基-1,2,3,4-四氢-1-(4-甲氧苯基)-3-甲基-2,4-二羰基-5-嘧啶基]-
苯酚-乳酸
苯酚,4-异氰基-,乙酸酯(ester)
苯酚,4-[(四氢-2H-吡喃-2-基)氧代]-,乙酸酯
苯酚,3-(1,1-二甲基乙基)-,乙酸酯
苯酚,2-溴-3-(二溴甲基)-5-甲氧基-,乙酸酯
苯甲醇,4-(乙酰氧基)-3,5-二甲氧基-
苯甲酸,4-(乙酰氧基)-2-氟-
苯氧基氯乙酸苯酯
苯基金刚烷-1-羧酸酯
苯基氰基甲酸酯
苯基庚酸酯
苯基庚-6-炔酸酯
苯基己酸酯
苯基呋喃-2-羧酸酯
苯基吡啶-2-羧酸酯
苯基十一碳-10-烯酸酯
苯基乙醛酸酯
苯基乙酸酯-d5
苯基丙二酸单苯酯
苯基丙-2-炔酸酯
苯基丁-2,3-二烯酸酯
苯基4-乙基环己烷羧酸
苯基3-乙氧基-3-亚氨基丙酸盐
苯基2-(苯磺酰基)乙酸酯
苯基2-(4-甲氧基苯基)乙酸酯
苯基2-(2-甲氧基苯基)乙酸酯
苯基2-(2-甲基苯基)乙酸酯
苯基-乙酸-(2-甲酰基-苯基酯)
苯基-乙酸-(2-环己基-苯基酯)