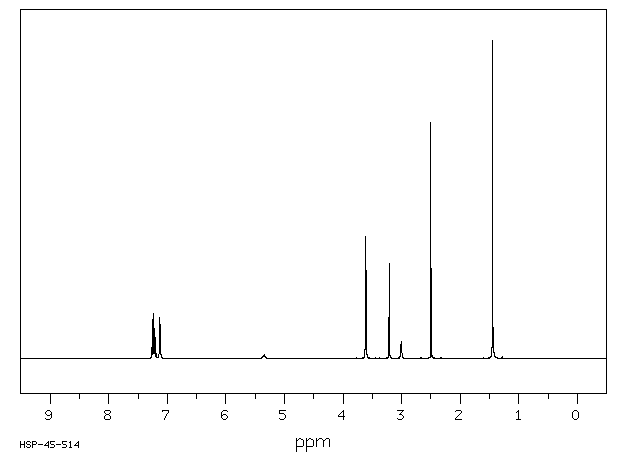
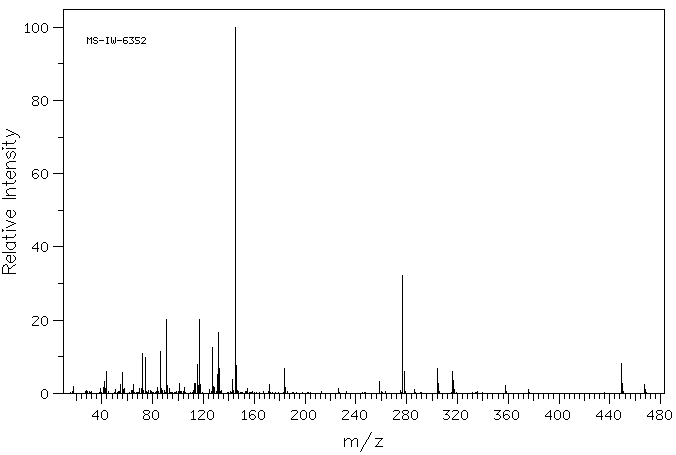
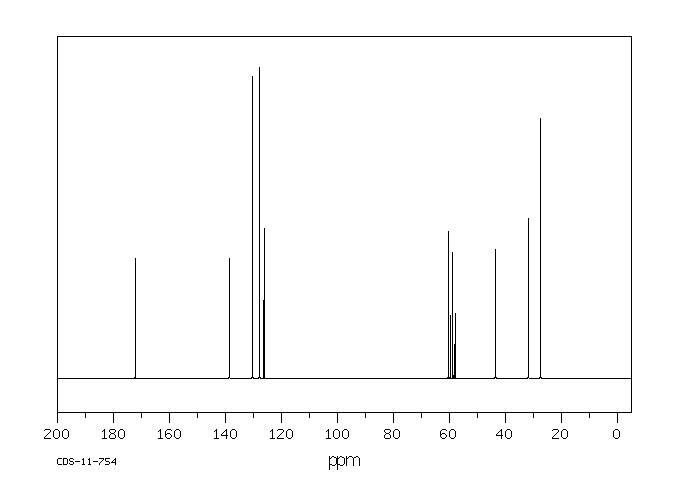
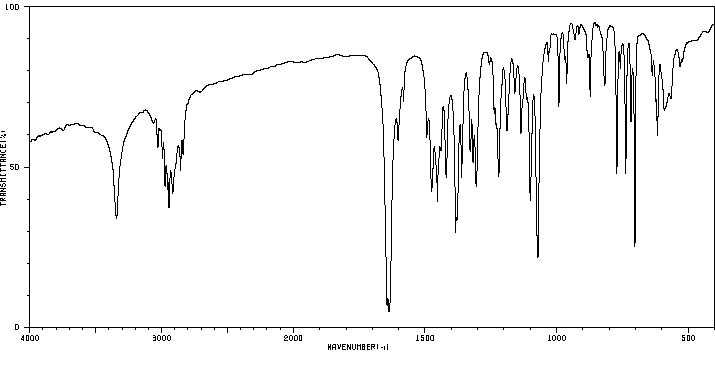
代谢
奥昔他卡因在肝脏中被迅速且广泛地代谢。代谢后,会形成如beta-羟基-美芬特明和beta-羟基-芬特明等初级代谢物。这些主要代谢物在血浆中的含量微不足道。
Oxetacaine is rapidly and extensively metabolized hepatically. After metabolism, there is a formation of primary metabolites such as beta-hydroxy-mephentermine and beta-hydroxy-phentermine. The major metabolites are found in the plasma in insignificant amounts.
来源:DrugBank