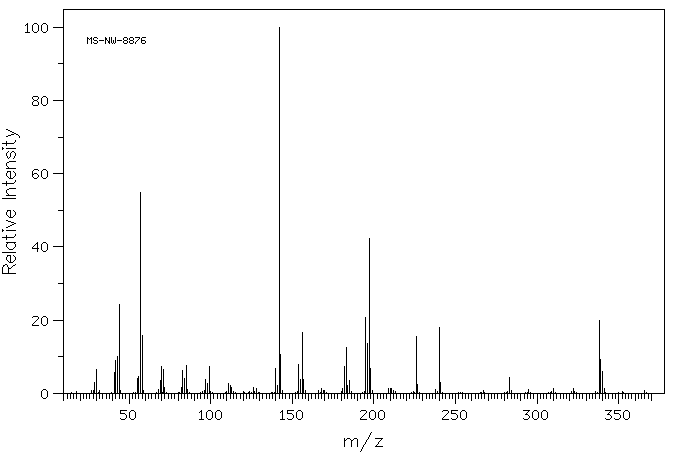
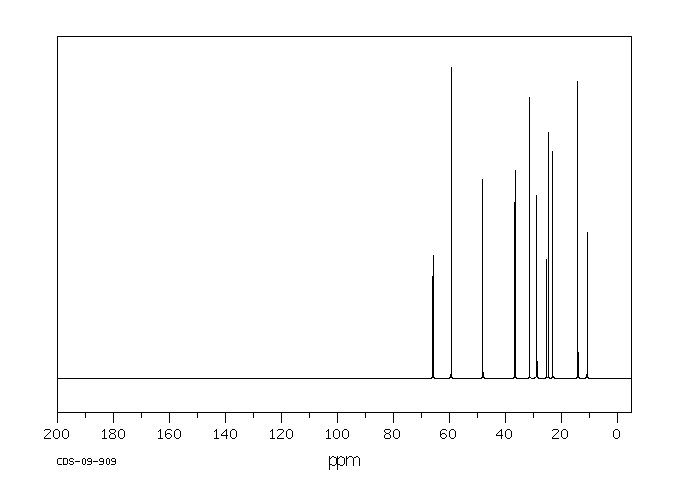
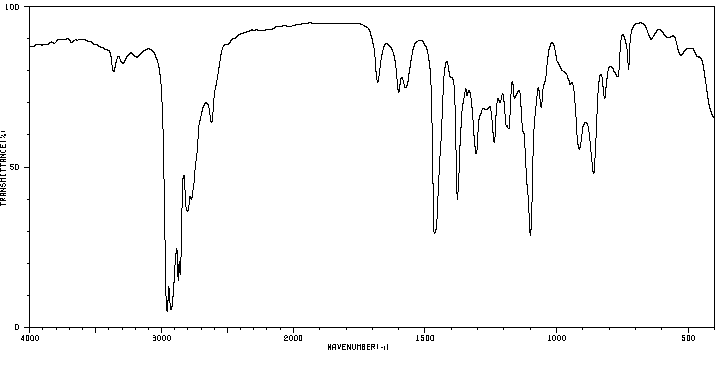
毒理性
/SRP:/ 立即急救:确保已经进行了充分的中毒物清除。如果患者停止呼吸,开始人工呼吸,最好使用需求阀复苏器、袋阀面罩装置或口袋面罩,按训练操作。如有必要,执行心肺复苏。立即用缓慢流动的水冲洗受污染的眼睛。不要催吐。如果发生呕吐,让患者前倾或将其置于左侧(如果可能的话,头部向下),以保持呼吸道畅通并防止吸入。保持患者安静,维持正常体温。寻求医疗帮助。 /毒物A和B/
/SRP:/ Immediate first aid: Ensure that adequate decontamination has been carried out. If patient is not breathing, start artificial respiration, preferably with a demand valve resuscitator, bag-valve-mask device, or pocket mask, as trained. Perform CPR if necessary. Immediately flush contaminated eyes with gently flowing water. Do not induce vomiting. If vomiting occurs, lean patient forward or place on the left side (head-down position, if possible) to maintain an open airway and prevent aspiration. Keep patient quiet and maintain normal body temperature. Obtain medical attention. /Poisons A and B/
来源:Hazardous Substances Data Bank (HSDB)