1,9-dichlorophenazine | 6479-79-4
中文名称
——
中文别名
——
英文名称
1,9-dichlorophenazine
英文别名
1,9-dichloro-phenazine;1,9-Dichlor-phenazin;1,9-Dichlorphenazin
CAS
6479-79-4
化学式
C12H6Cl2N2
mdl
——
分子量
249.099
InChiKey
DCKUXYHWYKPULG-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:208-209 °C
-
沸点:420.9±15.0 °C(Predicted)
-
密度:1.482±0.06 g/cm3(Predicted)
计算性质
-
辛醇/水分配系数(LogP):3.9
-
重原子数:16
-
可旋转键数:0
-
环数:3.0
-
sp3杂化的碳原子比例:0.0
-
拓扑面积:25.8
-
氢给体数:0
-
氢受体数:2
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 —— 1,9-dichloro-phenazine-5-oxide 18258-49-6 C12H6Cl2N2O 265.098
反应信息
-
作为反应物:描述:参考文献:名称:Tschernezkii, Zhurnal Obshchei Khimii, 1957, vol. 27, p. 2833,2835; engl. Ausg. S. 2869, 2871摘要:DOI:
-
作为产物:参考文献:名称:Otomasu, Pharmaceutical Bulletin, 1955, vol. 3, p. 365,368摘要:DOI:
文献信息
-
Enantiomerically pure chiral phenazino-crown ethers: synthesis, preliminary circular dichroism spectroscopic studies and complexes with the enantiomers of 1-arethyl ammonium salts作者:Erika Samu、Péter Huszthy、László Somogyi、Miklós HollósiDOI:10.1016/s0957-4166(99)00287-6日期:1999.7Enantiomerically pure chiral crown ethers containing the phenazine unit [(R,R)-2–(S,S)-8] were prepared by two types of cyclization reactions. Ligands (R,R)-2, (R,R)-3, (S,S)-4, (R,R)-5, (R,R)-6 and (R,R)-7 were prepared from phenazine-1,9-diol 9 and the appropriate ditosylates (S,S)-10–(S,S)-15 in weak basic conditions with complete inversion of configuration. Ligands (S,S)-2, (S,S)-7 and (S,S)-8通过两种环化反应制备了含有吩嗪单元[(R,R)-2-(S,S)-8 ]的对映体纯手性冠醚。配体(R,R) - 2,(R,R) - 3,(S,S) - 4,(R,R) - 5,(R,R) - 6和(R,R) - 7制备由吩嗪-1,9-二醇9和适当的二甲苯磺酸盐(S,S)-10 –(S,S)-15在弱的基本条件下,完全颠倒了配置。然而,配体(S,S)-2,(S,S)-7和(S,S)-8是由1,9-二氯吩嗪19和适当的二醇(S,S)-16-(S ,S)-18在牢固的基本条件下保持配置。已经通过CD光谱研究了α-(1-萘基)乙基高氯酸铵(NEA)和α-苯基乙基高氯酸铵(PEA)对大多数手性配体的对映体识别。
-
Tschernezkii; Serebrjanyi, Sb. Statei Obshch. Khim., 1953, p. 646,648作者:Tschernezkii、SerebrjanyiDOI:——日期:——
表征谱图
-
氢谱1HNMR
-
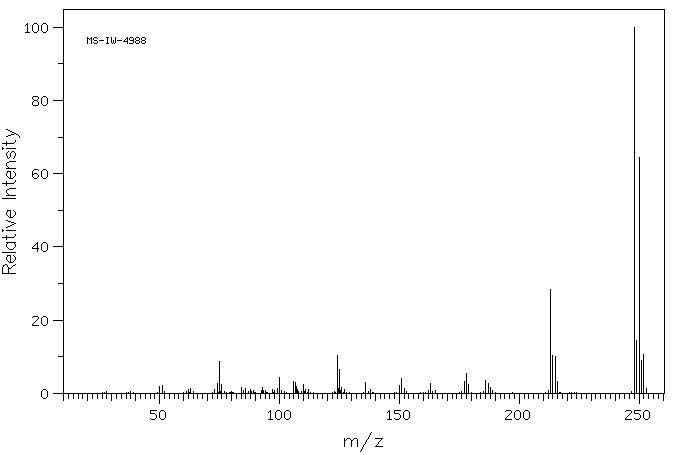
质谱MS
-
碳谱13CNMR
-
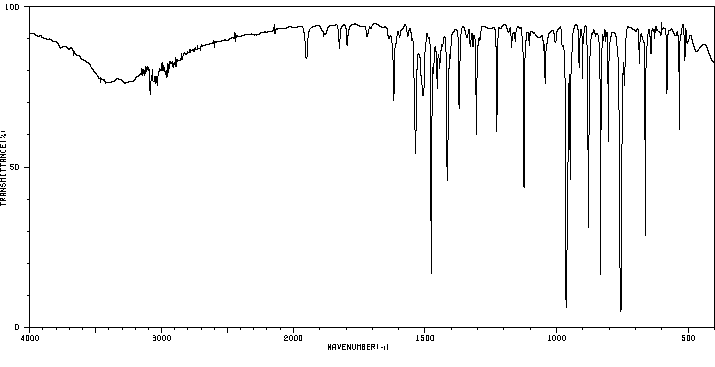
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(12羟基吲[2,1-b〕喹唑啉-6(12H)-酮)
黑暗猝灭剂BHQ-3,BHQ-3NHS
鸭嘴花酚碱
鸭嘴花碱酮;(S)-2,3-二氢-3,7-二羟基吡咯并[2,1-b]喹唑啉-9(1H)-酮
鸭嘴花碱酮
鸭嘴花碱盐酸盐
鲁米诺单钠盐
鲁米诺
骆驼蓬碱
颜料蓝64
颜料蓝60
顺式-卤夫酮
顺式-(喹喔啉-2-基)丙烯腈1,4-二氧化物
非奈利酮
青黛酮
雷替曲塞杂质1
阿法替尼杂质J
阿法替尼杂质I
阿法替尼杂质28
阿法替尼杂质18
阿法替尼杂质13
阿法替尼杂质
阿法替尼中间体
阿法替尼
阿法替尼
阿朴藏红
阿巴康唑
阿夫唑嗪杂质A
阿夫唑嗪杂质
阿夫唑嗪EP杂质C
阿夫唑嗪
阿喹司特
阿呋唑嗪杂质
阿呋唑嗪杂质
铜迈星
铁诱导细胞死亡激活剂
钠四丙基硼酸酯
酸性蓝98
酸性红101
酮色林醇
酞联氮基[2,3-b]酞嗪-5,14-二酮,7,12-二氢-
酞嗪-5-羧酸
酞嗪-2-氧化物
酚藏花红
酚嗪
酒石酸溴莫尼定
邻苯二甲酰肼
还原黄6GD
还原蓝6
达尼喹酮