4-(三氟甲基)苯甲酸乙酯 | 583-02-8
中文名称
4-(三氟甲基)苯甲酸乙酯
中文别名
4-三氟甲基苯甲酸乙酯
英文名称
ethyl 4-(trifluoromethyl)benzoate
英文别名
ethyl p-trifluoromethylbenzoate
CAS
583-02-8
化学式
C10H9F3O2
mdl
MFCD00013563
分子量
218.175
InChiKey
ZQDADDSPMCHZPX-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
沸点:90 °C
-
密度:1.230±0.06 g/cm3(Predicted)
-
闪点:78-80°C/5mm
-
稳定性/保质期:
计算性质
-
辛醇/水分配系数(LogP):3.5
-
重原子数:15
-
可旋转键数:3
-
环数:1.0
-
sp3杂化的碳原子比例:0.3
-
拓扑面积:26.3
-
氢给体数:0
-
氢受体数:5
安全信息
-
危险品标志:F
-
安全说明:S26,S36
-
危险类别码:R36/37/38
-
海关编码:2916399090
-
危险性防范说明:P261,P305+P351+P338
-
危险性描述:H302,H315,H319,H335
-
储存条件:贮存: 将密封器皿密封后,放置于阴凉、干燥处保存。
SDS
| Name: | Ethyl 4-(trifluoromethyl)benzoate 97% Material Safety Data Sheet |
| Synonym: | |
| CAS: | 583-02-8 |
Synonym:
Section 2 - COMPOSITION, INFORMATION ON INGREDIENTS
| CAS# | Chemical Name | content | EINECS# |
| 583-02-8 | Ethyl 4-(trifluoromethyl)benzoate | 97% | unlisted |
Risk Phrases: None Listed.
Section 3 - HAZARDS IDENTIFICATION
EMERGENCY OVERVIEW
Not available.
Potential Health Effects
Eye:
May cause eye irritation.
Skin:
May cause skin irritation. May be harmful if absorbed through the skin.
Ingestion:
May cause irritation of the digestive tract. May be harmful if swallowed.
Inhalation:
May cause respiratory tract irritation. May be harmful if inhaled.
Chronic:
Not available.
Section 4 - FIRST AID MEASURES
Eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid.
Skin:
Get medical aid. Flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes.
Ingestion:
Get medical aid. Wash mouth out with water.
Inhalation:
Remove from exposure and move to fresh air immediately.
Notes to Physician:
Treat symptomatically and supportively.
Section 5 - FIRE FIGHTING MEASURES
General Information:
As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear.
Extinguishing Media:
Use water spray, dry chemical, carbon dioxide, or chemical foam.
Section 6 - ACCIDENTAL RELEASE MEASURES
General Information: Use proper personal protective equipment as indicated in Section 8.
Spills/Leaks:
Absorb spill with inert material (e.g. vermiculite, sand or earth), then place in suitable container.
Section 7 - HANDLING and STORAGE
Handling:
Avoid breathing dust, vapor, mist, or gas. Avoid contact with skin and eyes.
Storage:
Store in a cool, dry place. Store in a tightly closed container.
Section 8 - EXPOSURE CONTROLS, PERSONAL PROTECTION
Engineering Controls:
Use adequate ventilation to keep airborne concentrations low.
Exposure Limits CAS# 583-02-8: Personal Protective Equipment Eyes: Not available.
Skin:
Wear appropriate protective gloves to prevent skin exposure.
Clothing:
Wear appropriate protective clothing to prevent skin exposure.
Respirators:
Follow the OSHA respirator regulations found in 29 CFR 1910.134 or European Standard EN 149. Use a NIOSH/MSHA or European Standard EN 149 approved respirator if exposure limits are exceeded or if irritation or other symptoms are experienced.
Section 9 - PHYSICAL AND CHEMICAL PROPERTIES
Physical State: Liquid
Color: colorless
Odor: Not available.
pH: Not available.
Vapor Pressure: Not available.
Viscosity: Not available.
Boiling Point: 78 - 80 deg C @5mmHg
Freezing/Melting Point: Not available.
Autoignition Temperature: Not available.
Flash Point: Not available.
Explosion Limits, lower: Not available.
Explosion Limits, upper: Not available.
Decomposition Temperature:
Solubility in water:
Specific Gravity/Density:
Molecular Formula: C10H9F3O2
Molecular Weight: 218
Section 10 - STABILITY AND REACTIVITY
Chemical Stability:
Not available.
Conditions to Avoid:
Incompatible materials.
Incompatibilities with Other Materials:
Strong oxidizing agents, acids, bases.
Hazardous Decomposition Products:
Carbon monoxide, carbon dioxide, fluorine, hydrogen fluoride gas.
Hazardous Polymerization: Has not been reported
Section 11 - TOXICOLOGICAL INFORMATION
RTECS#:
CAS# 583-02-8 unlisted.
LD50/LC50:
Not available.
Carcinogenicity:
Ethyl 4-(trifluoromethyl)benzoate - Not listed by ACGIH, IARC, or NTP.
Section 12 - ECOLOGICAL INFORMATION
Section 13 - DISPOSAL CONSIDERATIONS
Dispose of in a manner consistent with federal, state, and local regulations.
Section 14 - TRANSPORT INFORMATION
IATA
No information available.
IMO
No information available.
RID/ADR
No information available.
Section 15 - REGULATORY INFORMATION
European/International Regulations
European Labeling in Accordance with EC Directives
Hazard Symbols: Not available.
Risk Phrases:
Safety Phrases:
S 24/25 Avoid contact with skin and eyes.
WGK (Water Danger/Protection)
CAS# 583-02-8: No information available.
Canada
None of the chemicals in this product are listed on the DSL/NDSL list.
CAS# 583-02-8 is not listed on Canada's Ingredient Disclosure List.
US FEDERAL
TSCA
CAS# 583-02-8 is not listed on the TSCA inventory.
It is for research and development use only.
SECTION 16 - ADDITIONAL INFORMATION
N/A
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 4-(三氟甲基)苯甲酸甲酯 methyl 4-(trifluoromethyl)benzoate 2967-66-0 C9H7F3O2 204.149 4-三氟甲基苯甲酸 4-trifluoromethylbenzoic acid 455-24-3 C8H5F3O2 190.122 苯甲酸乙酯 benzoic acid ethyl ester 93-89-0 C9H10O2 150.177 对三氟甲基苯甲醛 4-Trifluoromethylbenzaldehyde 455-19-6 C8H5F3O 174.122 4-(三氟甲基)苄醇 4-(trifluoromethyl)benzylic alcohol 349-95-1 C8H7F3O 176.138 4-氯苯甲酸乙酯 ethyl 4-chlorobenzoate 7335-27-5 C9H9ClO2 184.622 苯佐卡因 p-aminoethylbenzoate 94-09-7 C9H11NO2 165.192 4-溴苯甲酸乙酯 Ethyl 4-bromobenzoate 5798-75-4 C9H9BrO2 229.073 对碘苯甲酸乙酯 4-iodobenzoic acid ethyl ester 51934-41-9 C9H9IO2 276.074 4-三氟甲基苯甲酰氯 4-trifluoromethyl-phenyl acetyl chloride 329-15-7 C8H4ClF3O 208.567 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 —— ethyl 4-(difluoromethyl)benzoate 64747-71-3 C10H10F2O2 200.185 4-(氟甲基)苯甲酸乙酯 4-carbethoxybenzyl fluoride 86239-04-5 C10H11FO2 182.195 4-三氟甲基苯甲酸 4-trifluoromethylbenzoic acid 455-24-3 C8H5F3O2 190.122 —— ethyl 2-bromo-4-(trifluoromethyl)benzoate —— C10H8BrF3O2 297.072 —— ethyl 2-chloro-4-(trifluoromethyl)benzoate 192805-87-1 C10H8ClF3O2 252.621 —— ethyl 4-(trifluoromethyl)salicylate 773128-58-8 C10H9F3O3 234.175 对三氟甲基苯甲醛 4-Trifluoromethylbenzaldehyde 455-19-6 C8H5F3O 174.122 4-(三氟甲基)苄醇 4-(trifluoromethyl)benzylic alcohol 349-95-1 C8H7F3O 176.138
反应信息
-
作为反应物:描述:参考文献:名称:消除反应的同位素效应研究。六、2-芳乙基铵离子的双分子消除反应机理摘要:使用示踪剂和动力学同位素效应技术研究了乙醇中 2-芳基乙基三甲基铵离子与乙醇盐离子的消除反应机理。2-苯基乙基三甲基铵-2,2-d2 溴化物和 2-(对三氟甲基苯基)-乙基三甲基铵-2,2-d2 溴化物均不与溶剂交换,观察到的氮同位素效应分别为 1.3% 和 0.9%。消除了涉及两种盐的自由溶剂化碳负离子中间体的两步过程。当溶剂从乙醇变为水时,观察到氮同位素效应的大小仅发生轻微变化,这也排除了两性离子中间体,该中间体与通过去除 β-氢形成的乙醇分子特异性氢键合乙氧基离子。最后,使用 α- 和 β- 双氘化底物的示踪研究消除了涉及叶立德和卡宾中间体的不太可能的反应途径。结论是2-芳基醚的反应...DOI:10.1139/v70-017
-
作为产物:描述:参考文献:名称:Silver-Catalyzed C–H Trifluoromethylation of Arenes Using Trifluoroacetic Acid as the Trifluoromethylating Reagent摘要:Direct trifluoromethylation of arenes using TFA as the trifluoromethylating reagent was achieved with Ag as the catalyst. This reaction not only provides a new protocol for aryl CH trifluoromethylation, but the generation of CF3 from TFA may prove useful in other contexts and could potentially be extended to other trifluoromethylation reactions.DOI:10.1021/ol503189j
文献信息
-
A Bifunctional Copper Catalyst Enables Ester Reduction with H<sub>2</sub>: Expanding the Reactivity Space of Nucleophilic Copper Hydrides作者:Birte M. Zimmermann、Trung Tran Ngoc、Dimitrios-Ioannis Tzaras、Trinadh Kaicharla、Johannes F. TeichertDOI:10.1021/jacs.1c09626日期:2021.10.13activation of esters through hydrogen bonding and formation of nucleophilic copper(I) hydrides from H2, resulting in a catalytic hydride transfer to esters. The reduction step is further facilitated by a proton shuttle mediated by the guanidinium subunit. This bifunctional approach to ester reductions for the first time shifts the reactivity of generally considered “soft” copper(I) hydrides to previously
-
Practical in situ-generation of phosphinite ligands for palladium-catalyzed carbonylation of (hetero)aryl bromides forming esters作者:Lin Wang、Helfried Neumann、Anke Spannenberg、Matthias BellerDOI:10.1039/c7cc02828h日期:——An effective method for alkoxycarbonylation of (hetero)aryl bromides is developed in the presence of in situ-generated phosphinite ligands tBu2POR (R = nBu, nPr, Et or Me). For this purpose commercially available tBu2PCl was used as the pre-ligand in the presence of different alcohols. For the first time cross coupling reactions with two alcohols – one generating the ligand, the other used as substrate
-
Conversion of Aromatic Amino into Trifluoromethyl Groups through a Sandmeyer-Type Transformation作者:Jianbo Wang、Xi Wang、Yan Xu、Yujing Zhou、Yan ZhangDOI:10.1055/s-0033-1338659日期:——strategy for aromatic trifluoromethylation by converting amino into trifluoromethyl groups via a Sandmeyer-type reaction is reported. The transformation involves diazotization of the aromatic amines with tert-butyl nitrite and hydrochloric acid to form aryldiazonium chlorides, followed by trifluoromethylation with trifluoromethylsilver at low temperature. Various readily available aromatic amines are
-
Copper-Promoted Conversion of Aromatic Amines into Trifluoromethylated Arenes: One-Pot Sandmeyer Trifluoromethylation作者:Jianquan Hong、Guifu Wang、Lianguang Huo、Changge ZhengDOI:10.1002/cjoc.201700311日期:2017.11A simple copper‐promoted one‐pot Sandmeyer trifluoromethylation of aromatic amines with Langlois’ reagent has been demonstrated. The reaction is performed in mild reaction conditions under an air atmosphere with good substrate scope and functional group compatibility. It provides an alternative and straightforward synthetic approach to access a variety of trifluoromethylated arenes.
-
Iron catalysed selective reduction of esters to alcohols作者:Sem Raj Tamang、Anthony F. Cozzolino、Michael FindlaterDOI:10.1039/c8ob02661k日期:——nature of the anionic complex was probed using EPR and IR experiments and is proposed to involve a dearomatized, radical, ligand scaffold. This complex is an active catalyst for the hydrosilylation of esters to afford alcohols; loadings as low as 1 mol% were employed.
表征谱图
-
氢谱1HNMR
-
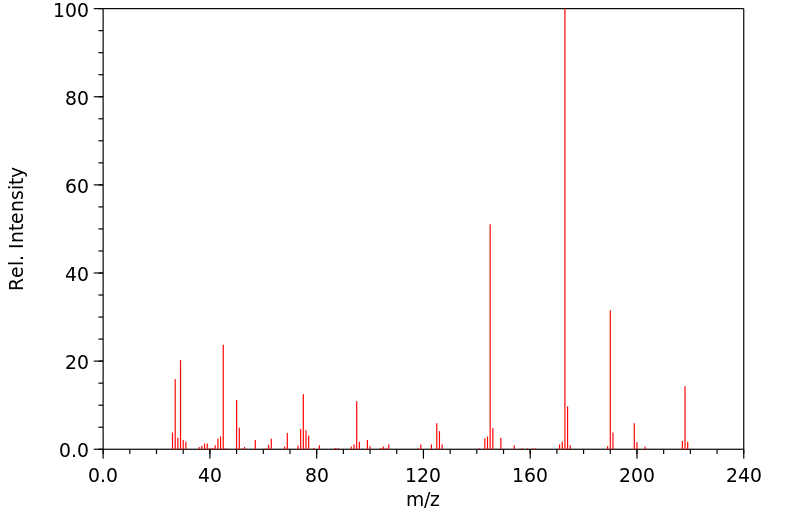
质谱MS
-
碳谱13CNMR
-
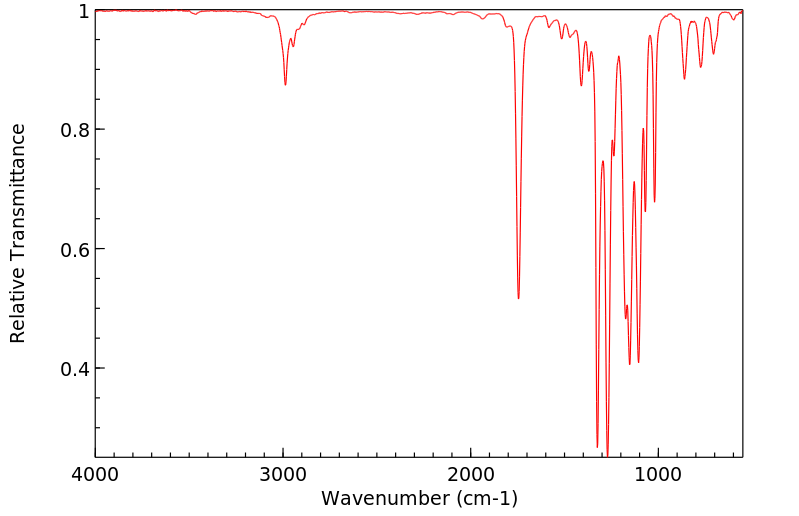
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(βS)-β-氨基-4-(4-羟基苯氧基)-3,5-二碘苯甲丙醇
(S,S)-邻甲苯基-DIPAMP
(S)-(-)-7'-〔4(S)-(苄基)恶唑-2-基]-7-二(3,5-二-叔丁基苯基)膦基-2,2',3,3'-四氢-1,1-螺二氢茚
(S)-盐酸沙丁胺醇
(S)-3-(叔丁基)-4-(2,6-二甲氧基苯基)-2,3-二氢苯并[d][1,3]氧磷杂环戊二烯
(S)-2,2'-双[双(3,5-三氟甲基苯基)膦基]-4,4',6,6'-四甲氧基联苯
(S)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(R)富马酸托特罗定
(R)-(-)-盐酸尼古地平
(R)-(-)-4,12-双(二苯基膦基)[2.2]对环芳烷(1,5环辛二烯)铑(I)四氟硼酸盐
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[((6-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(4-叔丁基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(3-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-4,7-双(3,5-二-叔丁基苯基)膦基-7“-[(吡啶-2-基甲基)氨基]-2,2”,3,3'-四氢1,1'-螺二茚满
(R)-3-(叔丁基)-4-(2,6-二苯氧基苯基)-2,3-二氢苯并[d][1,3]氧杂磷杂环戊烯
(R)-2-[((二苯基膦基)甲基]吡咯烷
(R)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(N-(4-甲氧基苯基)-N-甲基-3-(1-哌啶基)丙-2-烯酰胺)
(5-溴-2-羟基苯基)-4-氯苯甲酮
(5-溴-2-氯苯基)(4-羟基苯基)甲酮
(5-氧代-3-苯基-2,5-二氢-1,2,3,4-oxatriazol-3-鎓)
(4S,5R)-4-甲基-5-苯基-1,2,3-氧代噻唑烷-2,2-二氧化物-3-羧酸叔丁酯
(4S,4''S)-2,2''-亚环戊基双[4,5-二氢-4-(苯甲基)恶唑]
(4-溴苯基)-[2-氟-4-[6-[甲基(丙-2-烯基)氨基]己氧基]苯基]甲酮
(4-丁氧基苯甲基)三苯基溴化磷
(3aR,8aR)-(-)-4,4,8,8-四(3,5-二甲基苯基)四氢-2,2-二甲基-6-苯基-1,3-二氧戊环[4,5-e]二恶唑磷
(3aR,6aS)-5-氧代六氢环戊基[c]吡咯-2(1H)-羧酸酯
(2Z)-3-[[(4-氯苯基)氨基]-2-氰基丙烯酸乙酯
(2S,3S,5S)-5-(叔丁氧基甲酰氨基)-2-(N-5-噻唑基-甲氧羰基)氨基-1,6-二苯基-3-羟基己烷
(2S,2''S,3S,3''S)-3,3''-二叔丁基-4,4''-双(2,6-二甲氧基苯基)-2,2'',3,3''-四氢-2,2''-联苯并[d][1,3]氧杂磷杂戊环
(2S)-(-)-2-{[[[[3,5-双(氟代甲基)苯基]氨基]硫代甲基]氨基}-N-(二苯基甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[((1S,2S)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[[((1R,2R)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2-硝基苯基)磷酸三酰胺
(2,6-二氯苯基)乙酰氯
(2,3-二甲氧基-5-甲基苯基)硼酸
(1S,2S,3S,5S)-5-叠氮基-3-(苯基甲氧基)-2-[(苯基甲氧基)甲基]环戊醇
(1S,2S,3R,5R)-2-(苄氧基)甲基-6-氧杂双环[3.1.0]己-3-醇
(1-(4-氟苯基)环丙基)甲胺盐酸盐
(1-(3-溴苯基)环丁基)甲胺盐酸盐
(1-(2-氯苯基)环丁基)甲胺盐酸盐
(1-(2-氟苯基)环丙基)甲胺盐酸盐
(1-(2,6-二氟苯基)环丙基)甲胺盐酸盐
(-)-去甲基西布曲明
龙蒿油
龙胆酸钠
龙胆酸叔丁酯
龙胆酸
龙胆紫-d6
龙胆紫