lithium dodecyl sulphate | 2044-56-6
中文名称
——
中文别名
——
英文名称
lithium dodecyl sulphate
英文别名
lithium dodecyl sulfate;lithium dodecylsulfate;lithium lauryl sulphate;LDS;lithium lauryl sulfate;lithium dodecylsulphate;lithium;dodecyl sulfate
CAS
2044-56-6
化学式
C12H25O4S*Li
mdl
——
分子量
272.335
InChiKey
YFVGRULMIQXYNE-UHFFFAOYSA-M
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
沸点:199℃[at 101 325 Pa]
-
密度:1.168[at 20℃]
-
溶解度:H2O: 0.1 M , 20 °C, 澄清,无色
计算性质
-
辛醇/水分配系数(LogP):0.39
-
重原子数:18
-
可旋转键数:12
-
环数:0.0
-
sp3杂化的碳原子比例:1.0
-
拓扑面积:74.8
-
氢给体数:0
-
氢受体数:4
安全信息
-
TSCA:Yes
-
危险等级:4.1
-
危险品标志:Xi
-
安全说明:S26,S36
-
危险类别码:R36/37/38
-
WGK Germany:3
-
海关编码:29209010
-
危险品运输编号:UN 1325
-
危险标志:GHS02,GHS05,GHS07
-
危险性描述:H228,H315,H318,H335
-
危险性防范说明:P210,P261,P280,P305 + P351 + P338
-
包装等级:II
-
危险类别:4.1
SDS
制备方法与用途
概述
目前,十二烷基硫酸锂主要作为生物科研用试剂使用,但由于用量较小,国内产品市场占有率不高。国内鲜有关于十二烷基硫酸锂的生产报道,大规模生产的厂家也未见报道。常规工艺路线包括脂肪醇磺化、中和、沉降、喷雾干燥等步骤,但受限于原料及生产工艺,其产品纯度低、杂质含量高。主要杂质包括同系物、硫酸锂、氯化锂以及未反应的脂肪醇,这限制了产品的使用范围。
提纯方法- 溶解:将粉状十二烷基硫酸锂加入到装有无水乙醇的溶解罐中,搅拌使其完全溶解,在40-60℃下保温30-60分钟。
- 脱色:将步骤(1)所得液体转入脱色过滤器进行脱色和初滤,以吸附溶液中的少量颜色及颗粒较大的机械杂质、无机盐等。
- 过滤:依次通过精密过滤器Ⅰ、精密过滤器Ⅱ和精密过滤器Ⅲ进行三级过滤。其中,精密过滤器Ⅰ的过滤精度为10μm(压力≤0.4MPa),可以过滤掉直径大于10μm的颗粒杂质;精密过滤器Ⅱ的过滤精度为5μm(压力≤0.3MPa),可过滤掉直径大于5μm的颗粒杂质;精密过滤器Ⅲ的过滤精度为0.45μm(压力≤0.3MPa),可以过滤掉直径大于0.45μm的颗粒杂质。
- 精馏浓缩:将过滤后的滤液转入精馏罐进行精馏。精馏过程中,溶剂通过回流器进入冷凝器,待精馏罐中物料含醇量小于10%时停止精馏,收集冷凝器中的冷凝液,并将精馏罐中剩余物料转入离心机甩干5-10分钟。此过程可除去易溶于溶剂的杂质如未磺化的脂肪醇等,离心结束后物料接近干燥,溶剂含量约为2%,且不板结,呈湿流沙状。精馏过程中循环水温度控制在60℃左右,精馏罐压力为0.08MPa。
- 干燥:将离心甩干后的物料送入干燥器进行干燥处理。控制循环水温度在约60℃,真空度约为0.08MPa,干燥时间根据物料的干燥程度决定。待物料完全干燥后称重、装袋。最终产品中十二烷基硫酸钠含量达到99.4%。
阴离子去垢剂(Anionic detergent),尤其在低温条件下适用于变性聚丙烯酰胺凝胶电泳。
反应信息
-
作为反应物:描述:参考文献:名称:在胶束溶液中酸催化十二烷基硫酸盐和十二烷基二乙氧基硫酸盐表面活性剂水解的动力学。第1部分:酸和表面活性剂浓度以及抗衡离子的性质和浓度的影响摘要:在浓缩的表面活性剂溶液(0.035–0.6 mol dm –3)中研究了酸催化十二烷基硫酸钠(SDS)和十二烷基二乙氧基硫酸钠(SDE 2 S)水解的速率。初始速率的酸浓度依赖性显示出“饱和”效应,而高于cmc的表面活性剂浓度增加会导致k 2,obs值最大,超过此值它们会急剧下降。根据胶束反应的离子交换假相模型讨论了这些结果。抗衡离子的性质会影响k 2,obs,其值遵循序列NH 4 > Li> Na镁 这归因于具有氢离子的胶束的斯特恩层中这些阳离子的离子交换常数K H / X的差异。保持总抗衡离子浓度和表面活性剂抗衡离子浓度与氢离子浓度之比恒定(为30,即0.6:0.02 mol dm –3),很大程度上消除了k 2,obs随表面活性剂浓度增加而降低的趋势,因为可以从伪相离子交换模型中获得。但是,理论和实验之间的定量一致性不太好,这至少部分是由于在高离子强度溶液中偏离理想状态的缘故。DOI:10.1039/f19837900953
-
作为产物:描述:参考文献:名称:有序系统中的电化学。2.硝基苯与阴离子,阳离子和非离子胶束之间相互作用的电化学和光谱检查摘要:DOI:10.1021/j100211a017
-
作为试剂:描述:lithium nitrate 、 Iron(III) nitrate nonahydrate 、 manganese (II) nitrate tetrahydrate 、 nickel(II) nitrate hexahydrate 、 alkaline earth salt of/the/ methylsulfuric acid 在 lithium dodecyl sulphate 、 正丁醇 作用下, 以 环己烷 、 水 为溶剂, 反应 18.0h, 生成参考文献:名称:Porous lithium rich Li 1.2 Mn 0.54 Ni 0.22 Fe 0.04 O 2 prepared by microemulsion route as a high capacity and high rate capability positive electrode material摘要:A porous layered composite of Li2MnO3 and LiMn0.35Ni0.55Fe0.1O2 (composition:Li1.2Mn0.54Ni0.22Fe0.04O2) is prepared by inverse microemulsion method and studied as a positive electrode material. The precursor is heated at several temperatures between 500 and 900 degrees C. The X-ray diffraction, scanning electron microscopy, and transmission electron microscopy studies suggested that well crystalline submicronsized particles are obtained. The product samples possess mesoporosity with broadly distributed pores around 10 similar to 50 nm diameter. Pore volume and surface area decrease by increasing the temperature of preparation. However, the electrochemical activity of the composite samples increases with an increase in temperature. The discharge capacity values of the samples prepared at 900 degrees C are about 186 mAh g(-1) at a specific current of 25 mA g(-1) with an excellent cycling stability. The composite sample also possesses high rate capability. The high rate capability is attributed to the porous nature of the material. (C) 2014 Elsevier Ltd. All rights reserved.DOI:10.1016/j.electacta.2014.07.155
文献信息
-
TARGETED IONOPHORE-BASED METAL DELIVERY申请人:The Regents of the University of California公开号:US20200113937A1公开(公告)日:2020-04-16The present disclosure provides ionophore compounds, which are useful for facilitating delivery of a metal ion to a cell, tissue or organ of interest. The present disclosure provides compositions comprising the subject ionophore compounds. The present disclosure provides methods of delivering a metal ion intracellularly to a target cell. The present disclosure also provides methods of treating a condition associated with a metal deficiency in an individual.
-
[EN] HAT INHIBITORS AND METHODS FOR THEIR USE<br/>[FR] INHIBITEURS DE HAT ET PROCÉDÉS POUR LEUR UTILISATION申请人:ABBVIE INC公开号:WO2016044777A1公开(公告)日:2016-03-24Compounds having a structure of Formula I or a stereoisomer, tautomer or pharmaceutically acceptable salt thereof, wherein R1, R2a, R2b, R3a, R3b, R3c, R4a, R4b, R5, R6, Z and X are as defined herein are provided. Pharmaceutical compositions comprising such compounds and methods for treating various HAT-related conditions or diseases, including cancer, by administration of such compounds are also provided.提供具有公式I结构或其立体异构体、互变异构体或药学上可接受的盐的化合物,其中R1、R2a、R2b、R3a、R3b、R3c、R4a、R4b、R5、R6、Z和X如本文所定义。还提供包含这些化合物的药物组合物以及通过给予这些化合物治疗各种HAT相关病症或疾病,包括癌症的方法。
-
[EN] SPIROCYCLIC HAT INHIBITORS AND METHODS FOR THEIR USE<br/>[FR] INHIBITEURS DE HAT SPIROCYCLIQUES ET LEURS PROCÉDÉS D'UTILISATION申请人:ABBVIE INC公开号:WO2016044770A1公开(公告)日:2016-03-24Compounds having a structure of Formula (IX) or a stereoisomer, tautomer or pharmaceutically acceptable salt thereof, wherein R1, R2a, R2b, R3a, R3b, R4a, R4b, Q1----Q2, R6, R7, A, B, W, x, and y are as defined herein and are provided. Pharmaceutical compositions comprising such compounds and methods for treating various HAT-related conditions or diseases, including cancer, by administration of such compounds are also provided.具有式(IX)的结构或其立体异构体、互变异构体或药学上可接受的盐的化合物,其中R1、R2a、R2b、R3a、R3b、R4a、R4b、Q1----Q2、R6、R7、A、B、W、x和y如本文所定义,并提供。还提供了包括这些化合物的药物组合物和通过给予这些化合物治疗各种HAT相关疾病或疾病,包括癌症的方法。
-
Inhibitors of nedd8-activating enzyme申请人:IP Gesellschaft für Management mbH公开号:EP2764866A1公开(公告)日:2014-08-13The invention relates to an administration unit comprising crystalline form I of (1 S,2S,4R)-4-[(6-[(1R,2S)-5-chloro-2methoxy-2,3-dihydro-1H-inden-1-yl]amino}pyrimidin-4-yl)oxy]-2-hydroxycyclopentyl}methyl sulfamate (I-216) hydrochloride salt and to a packaging comprising the administration unit according to the invention.
-
[EN] TETRAHYDROISOQUINOLINES AS SELECTIVE NADPH OXIDASE 2 INHIBITORS<br/>[FR] TÉTRAHYDROISOQUINOLINES UTILES COMME INHIBITEURS SÉLECTIFS DE NADPH OXIDASE 2申请人:UNIV PITTSBURGH公开号:WO2014179592A1公开(公告)日:2014-11-06Embodiments of bridged tetrahydroisoquinolines and methods for their use in selectively inhibiting nicotinamide adenine dinucleotide phosphate (NADPH) oxidase 2 are disclosed. The disclosed compounds have a structure according to general formula I or a pharmaceutically acceptable salt thereof: wherein "−−−−−−−" represents a single or double bond, R1 is hydrogen, halogen, lower aliphatic, substituted or unsubstituted aryl, or substituted or unsubstituted heteroaryl; Ra is hydrogen, -CH2R2, R 3, or -SO2R 4; R 2 is lower aliphatic, substituted or unsubstituted aryl, or substituted or unsubstituted heteroaryl; R3 is substituted or unsubstituted aryl, or substituted or unsubstituted heteroaryl; R4 is lower aliphatic, or substituted or unsubstituted aryl; and R5 is hydrogen, halogen, or lower aliphatic.
表征谱图
-
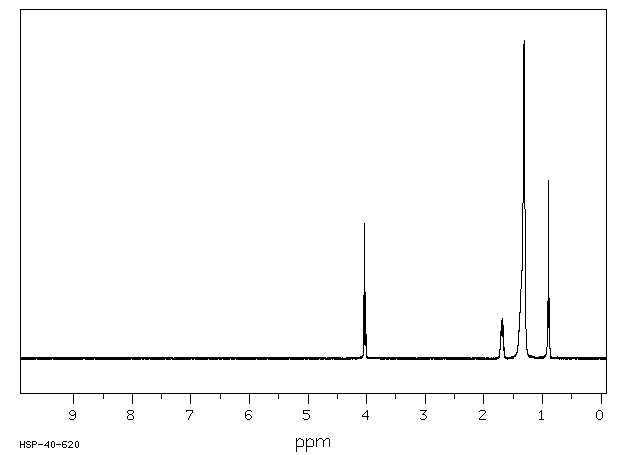
氢谱1HNMR
-
质谱MS
-
碳谱13CNMR
-
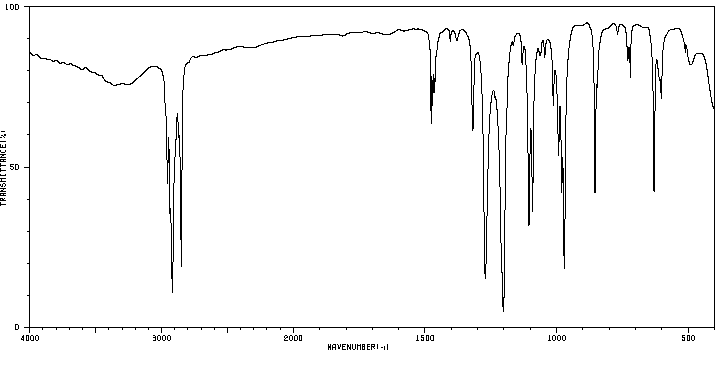
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(R)-3-BOC-5-甲基-1,2,3-氧杂噻唑烷-2,2-二氧化物
(4S)-4-叔丁基-1,2,3-氧杂噻唑烷-2,2-二氧化物-3-羧酸叔丁酯
(4S)-4-i-丙基-1,2,3-氧代噻唑烷-2,2-二氧化物-3-羧酸叔丁酯
(4R)-4-叔丁基-1,2,3-氧杂噻唑烷-2,2-二氧化物-3-羧酸叔丁酯
鲸蜡硬脂醇硫酸酯钠盐
高香草酸硫酸盐
鞘氨醇
非氯双链季铵盐
靛草素
阿维巴坦中间体5
间硝基苯基甲基硫酸盐
镧硫酸二乙酯
镁己基硫酸盐
铵硫酸癸酯盐
铵硫酸甲酯盐
铵己基硫酸盐
铵二十烷基硫酸盐
铵二十二烷基硫酸酯盐
铵2,3-二溴丙基硫酸盐
铝十二烷基硫酸盐
钾十八烷基硫酸酯盐
钾二十二烷基硫酸酯盐
钾p-氨基苯基硫酸盐
钾(4-硝基苯基)硫酸盐
钠硫酸丙酯盐
钠癸烷-2-基硫酸盐
钠氨基甲酰(羟基)氨基磺酸
钠二十六烷基硫酸酯盐
钠乙酰基硫酸盐
钠[(2-羟基乙基)锍二基]二-2,1-乙二基二硫酸盐
钠2-己氧乙基硫酸盐
钙十三烷基硫酸盐
钙二(甲基氨基磺酸)
酸性甘油-1,3-二硫酸盐
酸式硫酸三乙基锡
酚酞二硫酸酯钾盐
酒石酸去甲肾上腺素杂质3钠盐
邻苯二酚-4-磺酸铵
辛基硫酸钠
辛基硫酸酯钾盐
辛基硫酸氢酯
辛基-2-硫酸酯
莎拉西娅根茎提取物
草莓酸
苯肾上腺素O-芳基硫酸盐
苯肼,硫酸盐
苯氧基磺酰基7-甲基辛酸酯
苯基硫酸钠盐
苯基2,2,2-三氯乙基硫酸盐
脲硫酸盐(1:1)