5-乙酰基噻吩-2-甲酸 | 4066-41-5
中文名称
5-乙酰基噻吩-2-甲酸
中文别名
5-乙酰基噻吩-2-羧酸,5-乙酰基硫代苯-2-羧酸;5-乙酰基噻吩-2-羧酸;5-乙酰-噻吩-2-羧酸;5-乙酰基硫代苯-2-羧酸;5-乙酰基硫代苯-2-羧酸, 98+%
英文名称
5-acetylthiophene-2-carboxylic acid
英文别名
5-acetyl-2-thiophenecarboxylic acid;2-Acetylthiophen-5-carbonsaeure;5-Acetyl-thiophen-2-carbonsaeure
CAS
4066-41-5
化学式
C7H6O3S
mdl
MFCD00055512
分子量
170.189
InChiKey
LIKIMWYKJUFVJP-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:210-212°C
-
沸点:385.2±27.0 °C(Predicted)
-
密度:1.383
-
溶解度:溶于甲醇
-
稳定性/保质期:
在常温常压下保持稳定
计算性质
-
辛醇/水分配系数(LogP):1.5
-
重原子数:11
-
可旋转键数:2
-
环数:1.0
-
sp3杂化的碳原子比例:0.142
-
拓扑面积:82.6
-
氢给体数:1
-
氢受体数:4
安全信息
-
危险品标志:Xi
-
危险类别码:R36/37/38
-
海关编码:2934999090
-
安全说明:S26,S36/37/39
-
危险性防范说明:P261,P305+P351+P338
-
危险性描述:H315,H319,H335
-
储存条件:避光、存于通风干燥处,并密封保存。
SDS
Material Safety Data Sheet
Section 1. Identification of the substance
5-Acetylthiophene-2-carboxylic acid
Product Name:
Synonyms:
Section 2. Hazards identification
Harmful by inhalation, in contact with skin, and if swallowed.
H315: Causes skin irritation
H319: Causes serious eye irritation
H335: May cause respiratory irritation
P261: Avoid breathing dust/fume/gas/mist/vapours/spray
Wear protective gloves/protective clothing/eye protection/face protection
P280:
P305+P351+P338: IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses if present
and easy to do – continue rinsing
P304+P340: IF INHALED: Remove victim to fresh air and keep at rest in a position comfortable for breathing
P405: Store locked up
Section 3. Composition/information on ingredients.
5-Acetylthiophene-2-carboxylic acid
Ingredient name:
CAS number: 4066-41-5
Section 4. First aid measures
Immediately wash skin with copious amounts of water for at least 15 minutes while removing
Skin contact:
contaminated clothing and shoes. If irritation persists, seek medical attention.
Eye contact: Immediately wash skin with copious amounts of water for at least 15 minutes. Assure adequate
flushing of the eyes by separating the eyelids with fingers. If irritation persists, seek medical
attention.
Inhalation: Remove to fresh air. In severe cases or if symptoms persist, seek medical attention.
Wash out mouth with copious amounts of water for at least 15 minutes. Seek medical attention.
Ingestion:
Section 5. Fire fighting measures
In the event of a fire involving this material, alone or in combination with other materials, use dry
powder or carbon dioxide extinguishers. Protective clothing and self-contained breathing apparatus
should be worn.
Section 6. Accidental release measures
Personal precautions: Wear suitable personal protective equipment which performs satisfactorily and meets local/state/national
standards.
Respiratory precaution: Wear approved mask/respirator
Hand precaution: Wear suitable gloves/gauntlets
Skin protection: Wear suitable protective clothing
Eye protection: Wear suitable eye protection
Methods for cleaning up: Mix with sand or similar inert absorbent material, sweep up and keep in a tightly closed container
for disposal. See section 12.
Environmental precautions: Do not allow material to enter drains or water courses.
Section 7. Handling and storage
Handling: This product should be handled only by, or under the close supervision of, those properly qualified
in the handling and use of potentially hazardous chemicals, who should take into account the fire,
health and chemical hazard data given on this sheet.
Storage: Store in closed vessels.
Section 8. Exposure Controls / Personal protection
Engineering Controls: Use only in a chemical fume hood.
Personal protective equipment: Wear laboratory clothing, chemical-resistant gloves and safety goggles.
General hydiene measures: Wash thoroughly after handling. Wash contaminated clothing before reuse.
Section 9. Physical and chemical properties
Not specified
Appearance:
Boiling point: No data
Melting point: No data
Flash point: No data
Density: No data
Molecular formula: C7H6O3S
Molecular weight: 170.2
Section 10. Stability and reactivity
Conditions to avoid: Heat, flames and sparks.
Materials to avoid: Oxidizing agents.
Possible hazardous combustion products: Carbon monoxide, sulfur oxides.
Section 11. Toxicological information
No data.
Section 12. Ecological information
No data.
Section 13. Disposal consideration
Arrange disposal as special waste, by licensed disposal company, in consultation with local waste
disposal authority, in accordance with national and regional regulations.
Section 14. Transportation information
Non-harzardous for air and ground transportation.
Section 15. Regulatory information
No chemicals in this material are subject to the reporting requirements of SARA Title III, Section
302, or have known CAS numbers that exceed the threshold reporting levels established by SARA
Title III, Section 313.
SECTION 16 - ADDITIONAL INFORMATION
N/A
Section 1. Identification of the substance
5-Acetylthiophene-2-carboxylic acid
Product Name:
Synonyms:
Section 2. Hazards identification
Harmful by inhalation, in contact with skin, and if swallowed.
H315: Causes skin irritation
H319: Causes serious eye irritation
H335: May cause respiratory irritation
P261: Avoid breathing dust/fume/gas/mist/vapours/spray
Wear protective gloves/protective clothing/eye protection/face protection
P280:
P305+P351+P338: IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses if present
and easy to do – continue rinsing
P304+P340: IF INHALED: Remove victim to fresh air and keep at rest in a position comfortable for breathing
P405: Store locked up
Section 3. Composition/information on ingredients.
5-Acetylthiophene-2-carboxylic acid
Ingredient name:
CAS number: 4066-41-5
Section 4. First aid measures
Immediately wash skin with copious amounts of water for at least 15 minutes while removing
Skin contact:
contaminated clothing and shoes. If irritation persists, seek medical attention.
Eye contact: Immediately wash skin with copious amounts of water for at least 15 minutes. Assure adequate
flushing of the eyes by separating the eyelids with fingers. If irritation persists, seek medical
attention.
Inhalation: Remove to fresh air. In severe cases or if symptoms persist, seek medical attention.
Wash out mouth with copious amounts of water for at least 15 minutes. Seek medical attention.
Ingestion:
Section 5. Fire fighting measures
In the event of a fire involving this material, alone or in combination with other materials, use dry
powder or carbon dioxide extinguishers. Protective clothing and self-contained breathing apparatus
should be worn.
Section 6. Accidental release measures
Personal precautions: Wear suitable personal protective equipment which performs satisfactorily and meets local/state/national
standards.
Respiratory precaution: Wear approved mask/respirator
Hand precaution: Wear suitable gloves/gauntlets
Skin protection: Wear suitable protective clothing
Eye protection: Wear suitable eye protection
Methods for cleaning up: Mix with sand or similar inert absorbent material, sweep up and keep in a tightly closed container
for disposal. See section 12.
Environmental precautions: Do not allow material to enter drains or water courses.
Section 7. Handling and storage
Handling: This product should be handled only by, or under the close supervision of, those properly qualified
in the handling and use of potentially hazardous chemicals, who should take into account the fire,
health and chemical hazard data given on this sheet.
Storage: Store in closed vessels.
Section 8. Exposure Controls / Personal protection
Engineering Controls: Use only in a chemical fume hood.
Personal protective equipment: Wear laboratory clothing, chemical-resistant gloves and safety goggles.
General hydiene measures: Wash thoroughly after handling. Wash contaminated clothing before reuse.
Section 9. Physical and chemical properties
Not specified
Appearance:
Boiling point: No data
Melting point: No data
Flash point: No data
Density: No data
Molecular formula: C7H6O3S
Molecular weight: 170.2
Section 10. Stability and reactivity
Conditions to avoid: Heat, flames and sparks.
Materials to avoid: Oxidizing agents.
Possible hazardous combustion products: Carbon monoxide, sulfur oxides.
Section 11. Toxicological information
No data.
Section 12. Ecological information
No data.
Section 13. Disposal consideration
Arrange disposal as special waste, by licensed disposal company, in consultation with local waste
disposal authority, in accordance with national and regional regulations.
Section 14. Transportation information
Non-harzardous for air and ground transportation.
Section 15. Regulatory information
No chemicals in this material are subject to the reporting requirements of SARA Title III, Section
302, or have known CAS numbers that exceed the threshold reporting levels established by SARA
Title III, Section 313.
SECTION 16 - ADDITIONAL INFORMATION
N/A
制备方法与用途
化学性质
5-乙酰基噻吩-2-羧酸是一种浅黄色的结晶体,具有良好的热稳定性和化学稳定性,并能溶解于多种有机溶剂,如乙醇、乙醚和氯仿等。
生产方法
在氧(O₂)气氛下,将噻吩衍生物(0.1 mmol, 0.0140 g)、CeCl₃(5 mol%,0.0012 g)、CCl₃CH₂OH (1.0当量, 0.0149 g) 和 CH₃CN (2 mL) 加入反应管中。将混合物垂直放置于400 nm LED灯(10W) 下,然后在30°C的蓝光照射下搅拌24小时。通过薄层色谱(TLC)指示反应完成。之后,向混合物中加入乙酸乙酯和水进行三次萃取。最后,在减压下浓缩溶剂,并使用乙腈洗涤纯化残留物,从而得到5-乙酰基噻吩-2-羧酸。
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 2-乙酰-5-甲基噻吩 2-Acetyl-5-methylthiophen 13679-74-8 C7H8OS 140.206 —— 5-(α-methoxyimino)ethyl-2-thiophenecarboxylic acid 114774-09-3 C8H9NO3S 199.23 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 5-乙酰基-噻吩-2-羧酸甲酯 methyl 5-acetylthiophene-2-carboxylate 4101-81-9 C8H8O3S 184.216 —— 5-(2-Bromoacetyl)thiophene-2-carboxylic acid 96543-76-9 C7H5BrO3S 249.085 5-乙基噻吩-2-甲酸 5-ethyl-2-thiophenecarboxylic acid 23229-72-3 C7H8O2S 156.205 5-乙酰基噻吩-2-羧酸乙酯 ethyl 5-acetylthiophene-2-carboxylate 33148-82-2 C9H10O3S 198.243 5-(2-溴-乙酰基)-噻吩-2-羧酸甲酯 methyl 5-(2-bromoacetyl)thiophene-2-carboxylate 4192-32-9 C8H7BrO3S 263.112 —— tert-butyl-5-acetylthiophene-2-carboxylate 844502-29-0 C11H14O3S 226.296 —— 5-(2,2-dibromo-acetyl)-thiophene-2-carboxylic acid methyl ester 656227-47-3 C8H6Br2O3S 342.008 —— 5-(1-hydroxy-1-methyl-ethyl)thiophene-2-carboxylic acid 77756-27-5 C8H10O3S 186.232 —— 5-(3-dimethylaminoacryloyl)thiophene-2-carboxylic acid methyl ester 866523-73-1 C11H13NO3S 239.295 —— (E)-methyl 5-(3-(dimethylamino)acryloyl)thiophene-2-carboxylate 862698-96-2 C11H13NO3S 239.295 —— 5-[1-(hydroxyimino)ethyl]-2-thiophenecarboxylic acid 98279-55-1 C7H7NO3S 185.203 —— 5-(1-hydroxyimino-ethyl)-thiophene-2-carboxylic acid 98279-55-1 C7H7NO3S 185.203 —— (E)-5-(3-(2-chlorophenyl)acryloyl)thiophene-2-carboxylic acid 1076188-35-6 C14H9ClO3S 292.743 —— 5-acetylthiophene-2-carbonyl chloride 89692-69-3 C7H5ClO2S 188.635 5-乙酰基噻吩-2-甲酰氨 5-acetyl-2-thiopheneamide 68257-89-6 C7H7NO2S 169.204 - 1
- 2
反应信息
-
作为反应物:描述:参考文献:名称:Gol'dfarb et al., Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, 1959, p. 2021,2023; engl. Ausg. S. 1925, 1927摘要:DOI:
-
作为产物:描述:参考文献:名称:烷基芳烃的光诱导深度好氧氧化摘要:氧化是自然界和化学工业中生产含氧化学品的主要化学过程。目前,苯甲酸和苯多羧酸(BPCAs)的工业生产主要以聚烷基苯的深度氧化为主,由于形成消耗臭氧层的溴化溴和对生产设备的腐蚀危害,在环境和经济上都有些不利。 . 在这份报告中,开发了(多)烷基苯的光诱导深度有氧氧化为苯(多)羧酸。CeCl 3被证明是在醇存在下作为氢和电子穿梭的有效HAT(氢原子转移)催化剂。双氧 (O 2) 被发现是唯一的终端氧化剂。在大多数情况下,纯产品很容易通过简单的过滤分离,这意味着大规模实施的优势。该反应提供了从天然丰富的石油原料中生产有价值的精细化学品的理想方案。DOI:10.1007/s11426-021-1032-7
文献信息
-
Thienopyridones as AMPK activators for the treatment of diabetes and obesity申请人:Iyengar R. Rajesh公开号:US20050038068A1公开(公告)日:2005-02-17The present invention relates to compounds that activate AMP-activated protein kinase (AMPK), including the preparation of the compounds, compositions containing the compounds and the use of the compounds in the prevention or treatment of disorders such as diabetes, metabolic syndrome, and obesity.
-
Use of Indole Derivatives as Nurr-1 Activators for the Application Thereof as a Medicament for the Treatment of Parkinson's Disease申请人:Amaudrut Jerome公开号:US20120232070A1公开(公告)日:2012-09-13Compounds derived from indole, notably useful in therapeutics, selected from: i) the compounds of formula: and ii) the pharmaceutically acceptable salts of the compounds of formula (I); in which R1, R2, R3, R4, R5, R6, R8, R9 and Cy have defined meanings, and the use of such compounds in pharmaceuticals for the treatment of neurodegenerative diseases, particularly Parkinson's disease.
-
FUSED HETEROCYCLIC DERIVATIVE AND USE THEREOF申请人:Sakai Nozomu公开号:US20090137595A1公开(公告)日:2009-05-28The present invention provides a fused heterocyclic derivative having a potent kinase inhibitory activity and use thereof. A compound represented by the formula (I): wherein each symbol is as defined in the specification, except a particular compound, or a salt thereof, and a pharmaceutical agent containing the compound or a prodrug thereof, which is a kinase (VEGFR, VEGFR2, PDGFR, Raf) inhibitor, an angiogenesis inhibitor, an agent for the prophylaxis or treatment of cancer, a cancer growth inhibitor or a cancer metastasis suppressor.
-
Kinase Inhibitors申请人:Wurster A. Julie公开号:US20070032478A1公开(公告)日:2007-02-08The present invention relates to organic molecules capable of modulating tyrosine kinase signal transduction in order to regulate, modulate and/or inhibit abnormal cell proliferation.本发明涉及有机分子,能够调节酪氨酸激酶信号传导,以调节、调控和/或抑制异常细胞增殖。
-
[EN] 3-OXO-TETRAHYDRO-FURO[3,2-B]PYRROL-4(5H)-YL) DERIVATIVES I<br/>[FR] DÉRIVÉS 3-OXO-TÉTRAHYDRO-FURO [3,2-B] PYRROL -4 (5H)-YL)申请人:GRUENENTHAL GMBH公开号:WO2015161928A1公开(公告)日:2015-10-29The invention relates to amidic oxotetrahydro-2H-furo[3.2-b]pyrrol-4(5H)-yl) derivatives as dual CatS/K inhibitors, to pharmaceutical compositions containing these compounds and also to these compounds for use in the treatment and/or prophylaxis of pain and further diseases and/or disorders.
表征谱图
-
氢谱1HNMR
-
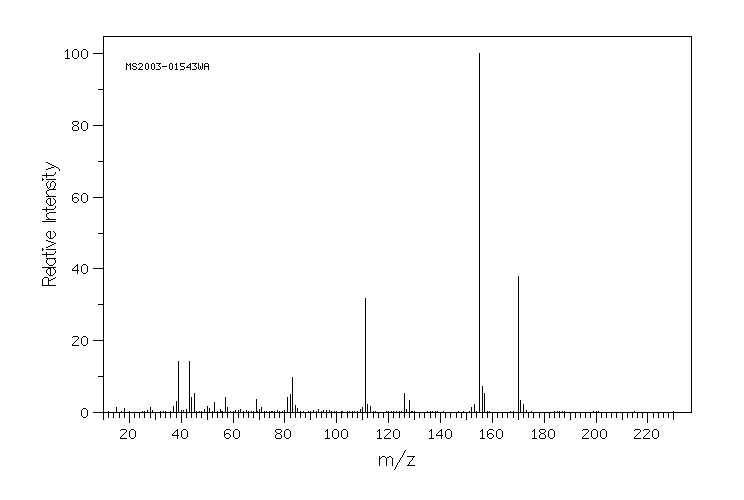
质谱MS
-
碳谱13CNMR
-
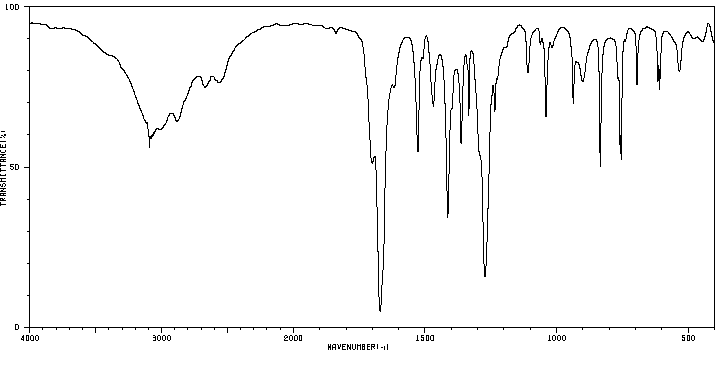
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
阿罗洛尔
阿替卡因
阿克兰酯
锡烷,(5-己基-2-噻吩基)三甲基-
邻氨基噻吩(2盐酸)
辛基5-(1,3-二氧戊环-2-基)-2-噻吩羧酸酯
辛基4,6-二溴噻吩并[3,4-b]噻吩-2-羧酸酯
辛基2-甲基异巴豆酸酯
血管紧张素IIAT2受体激动剂
葡聚糖凝胶LH-20
苯螨噻
苯并[c]噻吩-1-羧酸,5-溴-4,5,6,7-四氢-3-(甲硫基)-4-羰基-,乙基酯
苯并[b]噻吩-2-胺
苯并[b]噻吩-2-胺
苯基-[5-(4,4,5,5-四甲基-[1,3,2]二氧杂硼烷-2-基)-噻吩-2-基亚甲基]-胺
苯基-(5-氯噻吩-2-基)甲醇
苯乙酸,-α--[(1-羰基-2-丙烯-1-基)氨基]-
苯乙酰胺,3,5-二氨基-a-羟基-2,4,6-三碘-
苯乙脒,2,6-二氯-a-羟基-
腈氨噻唑
聚(3-丁基噻吩-2,5-二基),REGIOREGULAR
硝呋肼
硅烷,(3-己基-2,5-噻吩二基)二[三甲基-
硅噻菌胺
盐酸阿罗洛尔
盐酸阿罗洛尔
盐酸多佐胺
甲酮,[5-(1-环己烯-1-基)-4-(2-噻嗯基)-1H-吡咯-3-基]-2-噻嗯基-
甲基5-甲酰基-4-甲基-2-噻吩羧酸酯
甲基5-乙氧基-3-羟基-2-噻吩羧酸酯
甲基5-乙基-3-肼基-2-噻吩羧酸酯
甲基5-(氯甲酰基)-2-噻吩羧酸酯
甲基5-(氯乙酰基)-2-噻吩羧酸酯
甲基5-(氨基甲基)噻吩-2-羧酸酯
甲基5-(4-甲氧基苯基)-2-噻吩羧酸酯
甲基5-(4-甲基苯基)-2-噻吩羧酸酯
甲基5-(1,3-二氧戊环-2-基)-2-噻吩羧酸酯
甲基4-硝基-2-噻吩羧酸酯
甲基4-氰基-5-(4,6-二氨基吡啶-2-基)偶氮-3-甲基噻吩-2-羧酸酯
甲基4-氨基-5-(甲硫基)-2-噻吩羧酸酯
甲基4-{[(2E)-2-(4-氰基苯亚甲基)肼基]磺酰}噻吩-3-羧酸酯
甲基4-(氯甲酰基)-3-噻吩羧酸酯
甲基4-(氨基磺酰基氨基)-3-噻吩羧酸酯
甲基3-甲酰氨基-4-甲基-2-噻吩羧酸酯
甲基3-氨基-5-异丙基-2-噻吩羧酸酯
甲基3-氨基-5-(4-溴苯基)-2-噻吩羧酸酯
甲基3-氨基-4-苯基-5-(三氟甲基)-2-噻吩羧酸酯
甲基3-氨基-4-氰基-5-甲基-2-噻吩羧酸酯
甲基3-氨基-4-丙基-2-噻吩羧酸酯
甲基3-[[(4-甲氧基苯基)亚甲基氨基]氨基磺酰基]噻吩-2-羧酸酯