6-氯色酮 | 33533-99-2
中文名称
6-氯色酮
中文别名
——
英文名称
6-chloro-4H-chromen-4-one
英文别名
6-chlorochromone;6-chlorochromen-4-one
CAS
33533-99-2
化学式
C9H5ClO2
mdl
MFCD00191904
分子量
180.59
InChiKey
VFZQATFTQAZCMO-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:136-138 °C(lit.)
-
沸点:290.9±40.0 °C(Predicted)
-
密度:1.399±0.06 g/cm3(Predicted)
计算性质
-
辛醇/水分配系数(LogP):2.4
-
重原子数:12
-
可旋转键数:0
-
环数:2.0
-
sp3杂化的碳原子比例:0.0
-
拓扑面积:26.3
-
氢给体数:0
-
氢受体数:2
安全信息
-
危险品标志:Xn
-
危险类别码:R20/21/22
-
WGK Germany:3
-
海关编码:2914700090
-
安全说明:S26,S36
-
储存条件:室温
SDS
Version 1.2
Regulation (EC) No 1907/2006
1 - Product and Company Information
Product Name 6-CHLOROCHROMONE, 97%
2 - Hazards Identification
3 - Composition/Information on Ingredients
Product Name CAS # EC no Annex I
Index Number
6-CHLOROCHROMONE, 97% 33533-99-2 None None
Formula C9H5ClO2
Molecular Weight 180,5900 AMU
4 - First Aid Measures
AFTER INHALATION
If inhaled, remove to fresh air. If not breathing give
artificial respiration. If breathing is difficult, give oxygen.
AFTER SKIN CONTACT
In case of contact, immediately wash skin with soap and copious
amounts of water.
AFTER EYE CONTACT
In case of contact, immediately flush eyes with copious amounts
of water for at least 15 minutes.
AFTER INGESTION
If swallowed, wash out mouth with water provided person is
conscious. Call a physician.
5 - Fire Fighting Measures
EXTINGUISHING MEDIA
Suitable: Water spray. Carbon dioxide, dry chemical powder, or
appropriate foam.
ALDRICH www.molbase.com
SPECIAL RISKS
Specific Hazard(s): Emits toxic fumes under fire conditions.
SPECIAL PROTECTIVE EQUIPMENT FOR FIREFIGHTERS
Wear self-contained breathing apparatus and protective clothing
to prevent contact with skin and eyes.
6 - Accidental Release Measures
PROCEDURE(S) OF PERSONAL PRECAUTION(S)
Wear respirator, chemical safety goggles, rubber boots, and
heavy rubber gloves.
METHODS FOR CLEANING UP
Sweep up, place in a bag and hold for waste disposal. Avoid
raising dust. Ventilate area and wash spill site after material
pickup is complete.
7 - Handling and Storage
HANDLING
Directions for Safe Handling: Avoid inhalation. Avoid contact
with eyes, skin, and clothing. Avoid prolonged or repeated
exposure.
STORAGE
Conditions of Storage: Keep tightly closed. Store in a cool dry
place.
8 - Exposure Controls / Personal Protection
ENGINEERING CONTROLS
Safety shower and eye bath. Mechanical exhaust required.
GENERAL HYGIENE MEASURES
Wash thoroughly after handling. Wash contaminated clothing before
reuse.
PERSONAL PROTECTIVE EQUIPMENT
Respiratory Protection: Use respirators and components tested and
approved under appropriate government standards such as NIOSH (US)
or CEN (EU). Respiratory protection is not required. Where
protection is desired, use multi-purpose combination (US) or type
ABEK (EN 14387) respirator cartridges.
Hand Protection: Compatible chemical-resistant gloves.
Eye Protection: Chemical safety goggles.
9 - Physical and Chemical Properties
pH N/A
BP/BP Range N/A
MP/MP Range 136,000. - 138,000 °
C.
Flash Point N/A
Flammability N/A
Autoignition Temp N/A
Oxidizing Properties N/A
Explosive Properties N/A
Explosion Limits N/A
Vapor Pressure N/A
ALDRICH www.molbase.com
Partition Coefficient N/A
Viscosity N/A
Vapor Density N/A
Saturated Vapor Conc. N/A
Evaporation Rate N/A
Bulk Density N/A
Decomposition Temp. N/A
Solvent Content N/A
Water Content N/A
Surface Tension N/A
Conductivity N/A
Miscellaneous Data N/A
Solubility N/A
10 - Stability and Reactivity
STABILITY
Materials to Avoid: Strong oxidizing agents.
HAZARDOUS DECOMPOSITION PRODUCTS
Hazardous Decomposition Products: Carbon monoxide, Carbon dioxide,
Hydrogen chloride gas.
11 - Toxicological Information
SIGNS AND SYMPTOMS OF EXPOSURE
To the best of our knowledge, the chemical, physical, and
toxicological properties have not been thoroughly investigated.
ROUTE OF EXPOSURE
Skin Contact: May cause skin irritation.
Eye Contact: May cause eye irritation.
Multiple Routes: May be harmful by inhalation, ingestion, or
skin absorption.
12 - Ecological Information
No data available.
13 - Disposal Considerations
SUBSTANCE DISPOSAL
Dissolve or mix the material with a combustible solvent and burn
in a chemical incinerator equipped with an afterburner and
scrubber. Observe all federal, state, and local environmental
regulations.
14 - Transport Information
RID/ADR
Non-hazardous for road transport.
IMDG
Non-hazardous for sea transport.
IATA
Non-hazardous for air transport.
15 - Regulatory Information
ALDRICH www.molbase.com
Caution: Substance not yet fully tested (EU).
COUNTRY SPECIFIC INFORMATION
Germany
WGK: 3
Self-Classification
16 - Other Information
WARRANTY
The above information is believed to be correct but does not
purport to be all inclusive and shall be used only as a guide. The
information in this document is based on the present state of our
knowledge and is applicable to the product with regard to
appropriate safety precautions. It does not represent any
guarantee of the properties of the product. Inc.,
shall not be held liable for any damage resulting from handling or
from contact with the above product. See reverse side of invoice
or packing slip for additional terms and conditions of sale.
Copyright 2010 Co. License granted to make
unlimitedpaper copies for internal use only.
DISCLAIMER
For R&D use only. Not for drug, household or other uses.
ALDRICH www.molbase.com
SECTION 16 - ADDITIONAL INFORMATION
N/A
Regulation (EC) No 1907/2006
1 - Product and Company Information
Product Name 6-CHLOROCHROMONE, 97%
2 - Hazards Identification
3 - Composition/Information on Ingredients
Product Name CAS # EC no Annex I
Index Number
6-CHLOROCHROMONE, 97% 33533-99-2 None None
Formula C9H5ClO2
Molecular Weight 180,5900 AMU
4 - First Aid Measures
AFTER INHALATION
If inhaled, remove to fresh air. If not breathing give
artificial respiration. If breathing is difficult, give oxygen.
AFTER SKIN CONTACT
In case of contact, immediately wash skin with soap and copious
amounts of water.
AFTER EYE CONTACT
In case of contact, immediately flush eyes with copious amounts
of water for at least 15 minutes.
AFTER INGESTION
If swallowed, wash out mouth with water provided person is
conscious. Call a physician.
5 - Fire Fighting Measures
EXTINGUISHING MEDIA
Suitable: Water spray. Carbon dioxide, dry chemical powder, or
appropriate foam.
ALDRICH www.molbase.com
SPECIAL RISKS
Specific Hazard(s): Emits toxic fumes under fire conditions.
SPECIAL PROTECTIVE EQUIPMENT FOR FIREFIGHTERS
Wear self-contained breathing apparatus and protective clothing
to prevent contact with skin and eyes.
6 - Accidental Release Measures
PROCEDURE(S) OF PERSONAL PRECAUTION(S)
Wear respirator, chemical safety goggles, rubber boots, and
heavy rubber gloves.
METHODS FOR CLEANING UP
Sweep up, place in a bag and hold for waste disposal. Avoid
raising dust. Ventilate area and wash spill site after material
pickup is complete.
7 - Handling and Storage
HANDLING
Directions for Safe Handling: Avoid inhalation. Avoid contact
with eyes, skin, and clothing. Avoid prolonged or repeated
exposure.
STORAGE
Conditions of Storage: Keep tightly closed. Store in a cool dry
place.
8 - Exposure Controls / Personal Protection
ENGINEERING CONTROLS
Safety shower and eye bath. Mechanical exhaust required.
GENERAL HYGIENE MEASURES
Wash thoroughly after handling. Wash contaminated clothing before
reuse.
PERSONAL PROTECTIVE EQUIPMENT
Respiratory Protection: Use respirators and components tested and
approved under appropriate government standards such as NIOSH (US)
or CEN (EU). Respiratory protection is not required. Where
protection is desired, use multi-purpose combination (US) or type
ABEK (EN 14387) respirator cartridges.
Hand Protection: Compatible chemical-resistant gloves.
Eye Protection: Chemical safety goggles.
9 - Physical and Chemical Properties
pH N/A
BP/BP Range N/A
MP/MP Range 136,000. - 138,000 °
C.
Flash Point N/A
Flammability N/A
Autoignition Temp N/A
Oxidizing Properties N/A
Explosive Properties N/A
Explosion Limits N/A
Vapor Pressure N/A
ALDRICH www.molbase.com
Partition Coefficient N/A
Viscosity N/A
Vapor Density N/A
Saturated Vapor Conc. N/A
Evaporation Rate N/A
Bulk Density N/A
Decomposition Temp. N/A
Solvent Content N/A
Water Content N/A
Surface Tension N/A
Conductivity N/A
Miscellaneous Data N/A
Solubility N/A
10 - Stability and Reactivity
STABILITY
Materials to Avoid: Strong oxidizing agents.
HAZARDOUS DECOMPOSITION PRODUCTS
Hazardous Decomposition Products: Carbon monoxide, Carbon dioxide,
Hydrogen chloride gas.
11 - Toxicological Information
SIGNS AND SYMPTOMS OF EXPOSURE
To the best of our knowledge, the chemical, physical, and
toxicological properties have not been thoroughly investigated.
ROUTE OF EXPOSURE
Skin Contact: May cause skin irritation.
Eye Contact: May cause eye irritation.
Multiple Routes: May be harmful by inhalation, ingestion, or
skin absorption.
12 - Ecological Information
No data available.
13 - Disposal Considerations
SUBSTANCE DISPOSAL
Dissolve or mix the material with a combustible solvent and burn
in a chemical incinerator equipped with an afterburner and
scrubber. Observe all federal, state, and local environmental
regulations.
14 - Transport Information
RID/ADR
Non-hazardous for road transport.
IMDG
Non-hazardous for sea transport.
IATA
Non-hazardous for air transport.
15 - Regulatory Information
ALDRICH www.molbase.com
Caution: Substance not yet fully tested (EU).
COUNTRY SPECIFIC INFORMATION
Germany
WGK: 3
Self-Classification
16 - Other Information
WARRANTY
The above information is believed to be correct but does not
purport to be all inclusive and shall be used only as a guide. The
information in this document is based on the present state of our
knowledge and is applicable to the product with regard to
appropriate safety precautions. It does not represent any
guarantee of the properties of the product. Inc.,
shall not be held liable for any damage resulting from handling or
from contact with the above product. See reverse side of invoice
or packing slip for additional terms and conditions of sale.
Copyright 2010 Co. License granted to make
unlimitedpaper copies for internal use only.
DISCLAIMER
For R&D use only. Not for drug, household or other uses.
ALDRICH www.molbase.com
SECTION 16 - ADDITIONAL INFORMATION
N/A
制备方法与用途
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 6-氯-3-甲酰基色酮 6-Chloro-4-oxo-4H-chromene-3-carbaldehyde 42248-31-7 C10H5ClO3 208.601 6-氯查尔酮-3-羧酸 6-chloro-4-oxo-4H-chromene-3-carboxylic acid 51085-92-8 C10H5ClO4 224.6 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 —— 3,6-dichloro-4H-chromen-4-one 68661-31-4 C9H4Cl2O2 215.036 —— 6-chloro-3-hydroxy-4H-chromen-4-one 73484-71-6 C9H5ClO3 196.59 6-氯黄酮 6-chloroflavone 10420-73-2 C15H9ClO2 256.688
反应信息
-
作为反应物:描述:参考文献:名称:用 9-BBN 还原色酮合成 2H-Chromenes摘要:通过 9-硼双环 [3.3.1] 壬烷的 1,2-加成,色素被区域选择性地还原为 2H-1-苯并吡喃。虽然过渡金属配合物对反应没有催化作用,但仅通过使用氯化钯(II),才能在相似程度上获得2H-1-苯并吡喃和二氢-1-苯并吡喃。此外,使用其他有机硼烷还原色酮不会产生 2H-1-苯并吡喃,而是通过仅还原烯烃部分产生色酮。DOI:10.1246/bcsj.74.967
-
作为产物:描述:参考文献:名称:一种有效的TBHP / TBAI介导的协议,可通过氧化CC键从苯并吡喃-4-酮合成4 H-铬-4-酮摘要:摘要已经开发了一种无过渡金属且高效的TBHP / TBAI介导的方案,用于通过氧化C-C键形成从苯并吡喃-4-酮合成4 H -chromen-4-酮。它在催化量的碘化四丁基铵和氧化剂叔丁基过氧化氢(TBHP,癸烷中的5-6 M)存在下进行,以良好的收率获得了相应的产物。此外,已经观察到TBHP的浓度增加到30mol%大大增加了4 H的收率。-chromen-4-ones,进一步增加将导致产率降低。该反应的机理涉及最初生成叔丁氧基自由基,其通过氧化单电子转化被转化为碘代色烷-4-酮。之后,将碘化氢从碘代chroman-4-one中除去,得到所需的产物,即4 H -chromen-4-ones。此外,这是通过脱氢反应由n -Bu 4 NI / TBHP介导的C–C键的罕见例子。 图形摘要DOI:10.1007/s00706-020-02576-8
文献信息
-
Chemoselective Hydrosilylation of the α,β-Site Double Bond in α,β- and α,β,γ,δ-Unsaturated Ketones Catalyzed by Macrosteric Borane Promoted by Hexafluoro-2-propanol作者:Xiao-Yu Zhan、Hua Zhang、Yu Dong、Jian Yang、Shuai He、Zhi-Chuan Shi、Lei Tang、Ji-Yu WangDOI:10.1021/acs.joc.0c00568日期:2020.5.15The B(C6F5)3-catalyzed chemoselective hydrosilylation of α,β- and α,β,γ,δ-unsaturated ketones into the corresponding non-symmetric ketones in mild reaction conditions is developed. Nearly 55 substrates including those bearing reducible functional groups such as alkynyl, alkenyl, cyano, and aromatic heterocycles are chemoselectively hydrosilylated in good to excellent yields. Isotope-labeling studies
-
Novel Bicyclic Pyridinones申请人:Pettersson Martin Youngjin公开号:US20120252758A1公开(公告)日:2012-10-04Compounds and pharmaceutically acceptable salts of the compounds are disclosed, wherein the compounds have the structure of Formula I as defined herein. Corresponding pharmaceutical compositions, methods of treatment, methods of synthesis, and intermediates are also disclosed.所述化合物及其药用可接受的盐被披露,其中所述化合物具有如本文所定义的Formula I的结构。相应的药物组合物、治疗方法、合成方法和中间体也被披露。
-
AgSbF6-controlled diastereodivergence in alkyne hydroarylation: facile access to Z- and E-alkenyl arenes作者:Minsik Min、Donghee Kim、Sungwoo HongDOI:10.1039/c4cc03602f日期:——AgSbF6-controlled diastereodivergent hydroarylation reactions were developed. Unprecedented and remarkable switching of the E/Z-stereoselectivity could be obtained by adjusting the AgSbF6 loading.开发了AgSbF6控制的立体多样性氢芳基化反应。通过调整AgSbF6的负载量,可以实现前所未有的E/Z立体选择性的显著切换。
-
Metal-free oxidative decarbonylative alkylation of chromones using aliphatic aldehydes作者:Rongzhen Chen、Jin-Tao Yu、Jiang ChengDOI:10.1039/c8ob00720a日期:——A decarbonylative alkylation of chromones via radical conjugate addition under metal-free conditions was developed using aliphatic aldehydes as alkylating reagents. A series of 2-tertiary, secondary, and even primary alkylated chromanones were obtained in moderate to excellent yields.
-
An Efficient Microwave-Assisted Propylphosphonic Anhydride (T3P®)-Mediated One-Pot Chromone Synthesis via Enaminones作者:Manoranjan Behera、C. Balakrishna、Venu Kandula、Ramakrishna Gudipati、Satyanarayana Yennam、P. DeviDOI:10.1055/s-0036-1591898日期:2018.5An efficient synthesis of 4H-chromene-4-ones via enamino ketones, with cyclization by using T3P® under microwave heating is described. This is the first report for the synthesis of chromones by using T3P®. Significant features of this method include short reaction times and high-purity products.
表征谱图
-
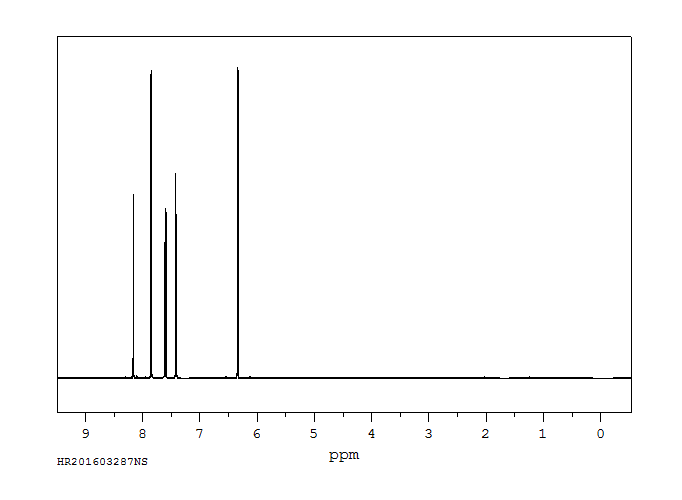
氢谱1HNMR
-
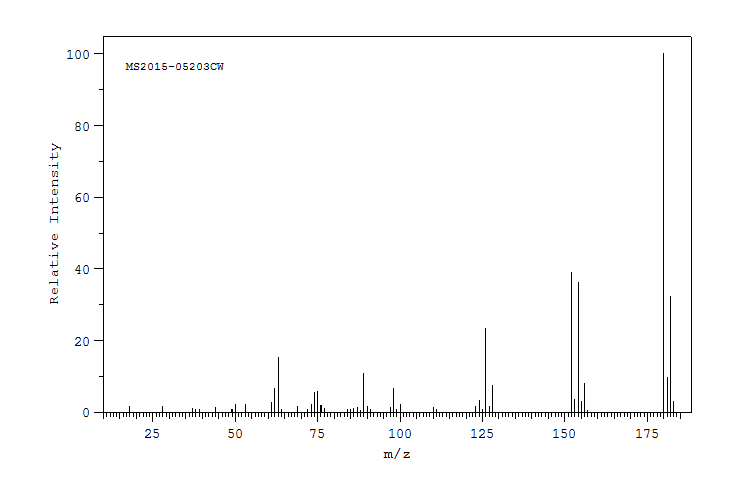
质谱MS
-
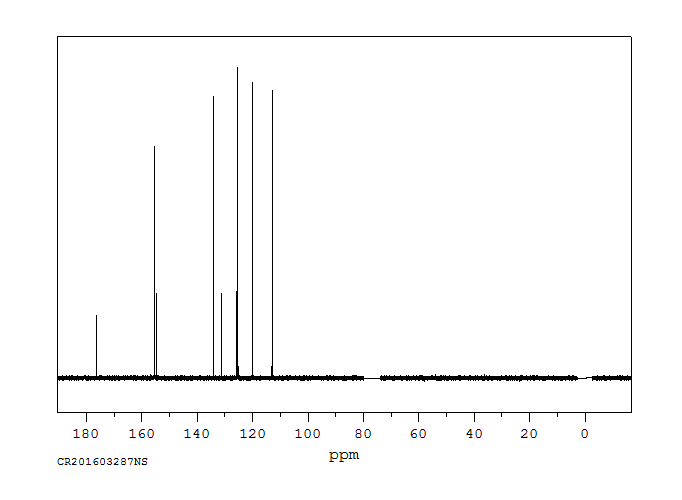
碳谱13CNMR
-
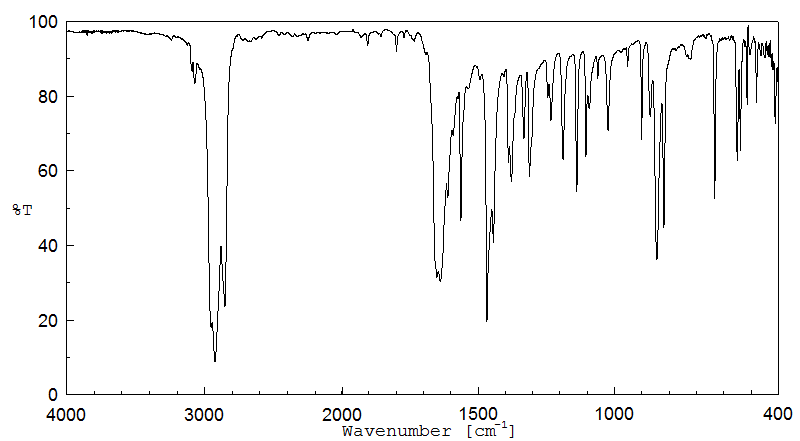
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(S)-8-氟苯并二氢吡喃-4-胺
(2S)-6-氟-3,4-二氢-4-氧代-2H-1-苯并吡喃-2-甲酸
(2R)-6-氟-3,4-二氢-4-氧代-2H-1-苯并吡喃-2-甲酸
龙胆根素
龙胆SHAN酮
齿阿米素
黑色素-1
黄天精
麥角黃酮酸
鲍迪木醌
高邻苯二甲酸酐
高芒果苷
高氯酸罗丹明640
马佐卡林
香豆素-340
香豆素 339
食用色素红色105号
颜料红90:1铝色淀[CI45380:3]
颜料红172铝色淀[CI45430:1]
雏菊叶龙胆酮
降阿赛里奥; 1,3,6,7-四羟基氧杂蒽酮
阿米醇
阿米凯林
阿米凯林
阿扎那托
阿巴哌酮
阿尼地坦
阿尼地坦
阿匹氯铵
锌离子荧光探针-4
锆(2+)二[2-(2,4,5,7-四溴-3,6-二羟基氧杂蒽-9-基)-3,4,5,6-四氯苯甲酸酯]
铁力木呫吨酮-B
铁力木吨酮A
钠6'-羟基-5-[2-[4-(2-甲基-3-氧代-7H-咪唑并[1,2-d]吡嗪-6-基)苯氧基]乙基硫代氨基甲酰氨基]-3-氧代螺[2-苯并呋喃-1,9'-氧杂蒽]-3'-醇
钠4-{[6'-(二乙基氨基)-3'-羟基-3-氧代-3H-螺[2-苯并呋喃-1,9'-氧杂蒽]-2'-基]偶氮}-3-羟基-1-萘磺酸酯
钠2-(2,7-二氯-9H-氧杂蒽-9-基)苯甲酸酯
钙黄绿素乙酰甲酯
钙荧光探针Fluo-8,AM
钙荧光探针Fluo-4,AM
钙离子荧光探针
钙柑子
采木(HAEMATOXYLONCAMPECHIANUM)木质提取物
酸性媒介桃红3BM
邻苯三酚红
远志山酮III
转移核糖核酸(面包酵母)
赤藓红B异硫氰酸酯异构体II
赤藓红
诺大麻
西伯尔链接剂