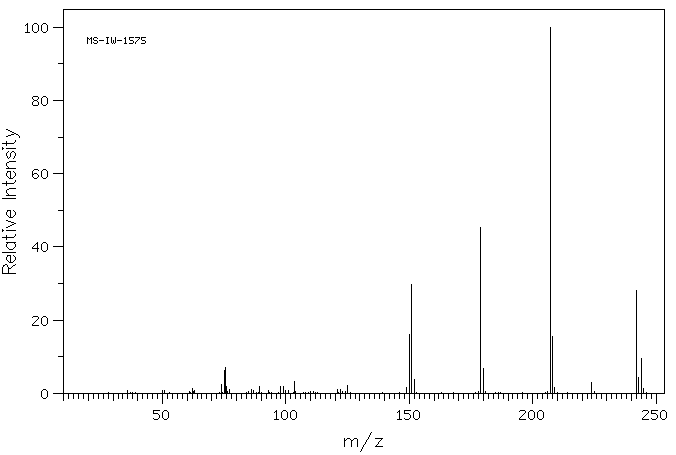
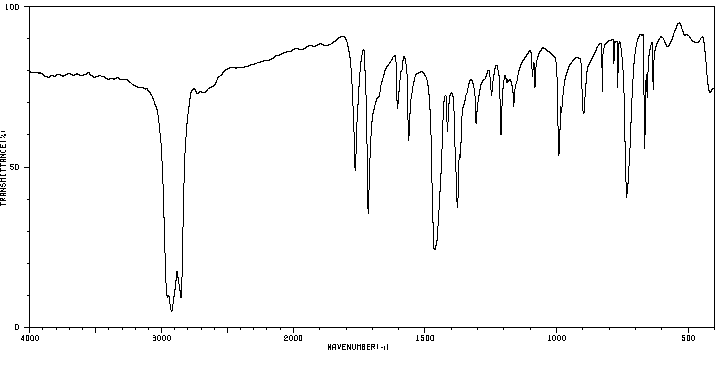
9-芴酮-4-甲酰氯 | 7071-83-2
中文名称
9-芴酮-4-甲酰氯
中文别名
9-氧代-4-芴甲酰氯
英文名称
9-fluorenone-4-carbonyl chloride
英文别名
9-oxo-9H-fluorene-4-carbonyl chloride;9-fluorenone-4-carboxylic acid chloride;9-oxofluorene-4-carbonyl chloride
CAS
7071-83-2
化学式
C14H7ClO2
mdl
MFCD00009931
分子量
242.661
InChiKey
XKCGWCQMEUASJF-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:139-141 °C (lit.)
-
沸点:420.1±24.0 °C(Predicted)
-
密度:1.417±0.06 g/cm3(Predicted)
计算性质
-
辛醇/水分配系数(LogP):3.6
-
重原子数:17
-
可旋转键数:1
-
环数:3.0
-
sp3杂化的碳原子比例:0.0
-
拓扑面积:34.1
-
氢给体数:0
-
氢受体数:2
安全信息
-
危险等级:8
-
危险类别码:R34,R40
-
危险品运输编号:UN 3261 8/PG 2
-
海关编码:2918300090
-
包装等级:III
-
安全说明:S26,S36/37/39,S45
-
WGK Germany:1
SDS
SECTION 1: Identification of the substance/mixture and of the company/undertaking
Product identifiers
Product name : 9-Fluorenone-4-carbonyl chloride
REACH No. : A registration number is not available for this substance as the substance
or its uses are exempted from registration, the annual tonnage does not
require a registration or the registration is envisaged for a later
registration deadline.
CAS-No. : 7071-83-2
Relevant identified uses of the substance or mixture and uses advised against
Identified uses : Laboratory chemicals, Manufacture of substances
SECTION 2: Hazards identification
Classification of the substance or mixture
Classification according to Regulation (EC) No 1272/2008
Skin corrosion (Category 1B), H314
Carcinogenicity (Category 2), H351
For the full text of the H-Statements mentioned in this Section, see Section 16.
Classification according to EU Directives 67/548/EEC or 1999/45/EC
C Corrosive R34
Xn Harmful R40
For the full text of the R-phrases mentioned in this Section, see Section 16.
Label elements
Labelling according Regulation (EC) No 1272/2008
Pictogram
Signal word Danger
Hazard statement(s)
H314 Causes severe skin burns and eye damage.
H351 Suspected of causing cancer.
Precautionary statement(s)
P280 Wear protective gloves/ protective clothing/ eye protection/ face
protection.
P305 + P351 + P338 IF IN EYES: Rinse cautiously with water for several minutes. Remove
contact lenses, if present and easy to do. Continue rinsing.
P310 Immediately call a POISON CENTER or doctor/ physician.
Supplemental Hazard none
Statements
Other hazards - none
SECTION 3: Composition/information on ingredients
Synonyms : 9-Oxo-4-fluorenecarbonyl chloride
Formula : C14H7ClO2
Molecular Weight : 242,66 g/mol
Hazardous ingredients according to Regulation (EC) No 1272/2008
Component Classification Concentration
9-Fluorenone-4-carbonyl chloride
CAS-No. 7071-83-2 Skin Corr. 1B; H314 <= 100 %
Methylene chloride
CAS-No. 75-09-2 Carc. 2; H351 1 - 10 %
EC-No. 200-838-9
Index-No. 602-004-00-3
Hazardous ingredients according to Directive 1999/45/EC
Component Classification Concentration
9-Fluorenone-4-carbonyl chloride
CAS-No. 7071-83-2 C, R34 <= 100 %
Methylene chloride
CAS-No. 75-09-2 Xn, Carc.Cat.3, R40 1 - 10 %
EC-No. 200-838-9
Index-No. 602-004-00-3
For the full text of the H-Statements and R-Phrases mentioned in this Section, see Section 16
SECTION 4: First aid measures
Description of first aid measures
General advice
Consult a physician. Show this safety data sheet to the doctor in attendance.
If inhaled
If breathed in, move person into fresh air. If not breathing, give artificial respiration. Consult a physician.
In case of skin contact
Take off contaminated clothing and shoes immediately. Wash off with soap and plenty of water. Consult a
physician.
In case of eye contact
Rinse thoroughly with plenty of water for at least 15 minutes and consult a physician.
If swallowed
Do NOT induce vomiting. Never give anything by mouth to an unconscious person. Rinse mouth with
water. Consult a physician.
Most important symptoms and effects, both acute and delayed
The most important known symptoms and effects are described in the labelling (see section 2.2) and/or in
section 11
Indication of any immediate medical attention and special treatment needed
no data available
SECTION 5: Firefighting measures
Extinguishing media
Suitable extinguishing media
Use water spray, alcohol-resistant foam, dry chemical or carbon dioxide.
Special hazards arising from the substance or mixture
Carbon oxides, Hydrogen chloride gas
Advice for firefighters
Wear self contained breathing apparatus for fire fighting if necessary.
Further information
no data available
SECTION 6: Accidental release measures
Personal precautions, protective equipment and emergency procedures
Use personal protective equipment. Avoid dust formation. Avoid breathing vapours, mist or gas. Ensure
adequate ventilation. Evacuate personnel to safe areas. Avoid breathing dust.
For personal protection see section 8.
Environmental precautions
Prevent further leakage or spillage if safe to do so. Do not let product enter drains.
Methods and materials for containment and cleaning up
Pick up and arrange disposal without creating dust. Sweep up and shovel. Keep in suitable, closed
containers for disposal.
Reference to other sections
For disposal see section 13.
SECTION 7: Handling and storage
Precautions for safe handling
Avoid contact with skin and eyes. Avoid formation of dust and aerosols.
Provide appropriate exhaust ventilation at places where dust is formed.
For precautions see section 2.2.
Conditions for safe storage, including any incompatibilities
Store in cool place. Keep container tightly closed in a dry and well-ventilated place.
Recommended storage temperature: 2 - 8 °C
Moisture sensitive.
Specific end use(s)
A part from the uses mentioned in section 1.2 no other specific uses are stipulated
SECTION 8: Exposure controls/personal protection
Control parameters
Components with workplace control parameters
Exposure controls
Appropriate engineering controls
Handle in accordance with good industrial hygiene and safety practice. Wash hands before breaks and
at the end of workday.
Personal protective equipment
Eye/face protection
Face shield and safety glasses Use equipment for eye protection tested and approved under
appropriate government standards such as NIOSH (US) or EN 166(EU).
Skin protection
Handle with gloves. Gloves must be inspected prior to use. Use proper glove removal technique
(without touching glove's outer surface) to avoid skin contact with this product. Dispose of
contaminated gloves after use in accordance with applicable laws and good laboratory practices.
Wash and dry hands.
The selected protective gloves have to satisfy the specifications of EU Directive 89/686/EEC and
the standard EN 374 derived from it.
Body Protection
Complete suit protecting against chemicals, The type of protective equipment must be selected
according to the concentration and amount of the dangerous substance at the specific workplace.
Respiratory protection
Where risk assessment shows air-purifying respirators are appropriate use a full-face particle
respirator type N100 (US) or type P3 (EN 143) respirator cartridges as a backup to engineering
controls. If the respirator is the sole means of protection, use a full-face supplied air respirator. Use
respirators and components tested and approved under appropriate government standards such
as NIOSH (US) or CEN (EU).
Control of environmental exposure
Prevent further leakage or spillage if safe to do so. Do not let product enter drains.
SECTION 9: Physical and chemical properties
Information on basic physical and chemical properties
a) Appearance Form: solid
b) Odour no data available
c) Odour Threshold no data available
d) pH no data available
e) Melting point/freezing Melting point/range: 139 - 141 °C - lit.
point
f) Initial boiling point and no data available
boiling range
g) Flash point no data available
h) Evapouration rate no data available
i) Flammability (solid, gas) no data available
j) Upper/lower no data available
flammability or
explosive limits
k) Vapour pressure no data available
l) Vapour density no data available
m) Relative density no data available
n) Water solubility no data available
o) Partition coefficient: n- log Pow: 3,07
octanol/water
p) Auto-ignition no data available
temperature
q) Decomposition no data available
temperature
r) Viscosity no data available
s) Explosive properties no data available
t) Oxidizing properties no data available
Other safety information
no data available
SECTION 10: Stability and reactivity
Reactivity
no data available
Chemical stability
Stable under recommended storage conditions.
Possibility of hazardous reactions
no data available
Conditions to avoid
Exposure to moisture.
Incompatible materials
Strong oxidizing agents, Strong bases
Hazardous decomposition products
Other decomposition products - no data available
In the event of fire: see section 5
SECTION 11: Toxicological information
Information on toxicological effects
Acute toxicity
no data available
Skin corrosion/irritation
no data available
Serious eye damage/eye irritation
no data available
Respiratory or skin sensitisation
no data available
Germ cell mutagenicity
no data available
Carcinogenicity
IARC: 2B - Group 2B: Possibly carcinogenic to humans (Methylene chloride)
Reproductive toxicity
no data available
Specific target organ toxicity - single exposure
no data available
Specific target organ toxicity - repeated exposure
no data available
Aspiration hazard
no data available
Additional Information
RTECS: Not available
burning sensation, Cough, wheezing, laryngitis, Shortness of breath, spasm, inflammation and edema of
the larynx, spasm, inflammation and edema of the bronchi, pneumonitis, pulmonary edema, Material is
extremely destructive to tissue of the mucous membranes and upper respiratory tract, eyes, and skin.
SECTION 12: Ecological information
Toxicity
no data available
Persistence and degradability
no data available
Bioaccumulative potential
no data available
Mobility in soil
no data available
Results of PBT and vPvB assessment
PBT/vPvB assessment not available as chemical safety assessment not required/not conducted
Other adverse effects
no data available
SECTION 13: Disposal considerations
Waste treatment methods
Product
Offer surplus and non-recyclable solutions to a licensed disposal company. Dissolve or mix the material
with a combustible solvent and burn in a chemical incinerator equipped with an afterburner and scrubber.
Contaminated packaging
Dispose of as unused product.
SECTION 14: Transport information
UN number
ADR/RID: 3261 IMDG: 3261 IATA: 3261
UN proper shipping name
ADR/RID: CORROSIVE SOLID, ACIDIC, ORGANIC, N.O.S. (9-Fluorenone-4-carbonyl chloride)
IMDG: CORROSIVE SOLID, ACIDIC, ORGANIC, N.O.S. (9-Fluorenone-4-carbonyl chloride)
IATA: Corrosive solid, acidic, organic, n.o.s. (9-Fluorenone-4-carbonyl chloride)
Transport hazard class(es)
ADR/RID: 8 IMDG: 8 IATA: 8
Packaging group
ADR/RID: II IMDG: II IATA: II
Environmental hazards
ADR/RID: no IMDG Marine pollutant: no IATA: no
Special precautions for user
no data available
SECTION 15 - REGULATORY INFORMATION
N/A
SECTION 16 - ADDITIONAL INFORMATION
N/A
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 9-芴酮-4-甲酸 9-oxo-9H-fluorene-4-carboxylic acid 6223-83-2 C14H8O3 224.216 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 4-氰基-9-芴酮 4-cyano-9-fluorenone 4269-20-9 C14H7NO 205.216 9-氧代-9h-芴-4-羧酰胺 9-oxofluorene-4-carboxamide 42135-38-6 C14H9NO2 223.231 —— 4-fluorenecarbaldehyde 82979-62-2 C14H10O 194.233 9-芴酮-4-甲酸 9-oxo-9H-fluorene-4-carboxylic acid 6223-83-2 C14H8O3 224.216 9-氧代-9h-芴-4-羧酸甲酯 9-oxo-fluorene-4-carboxylic acid methyl ester 4269-19-6 C15H10O3 238.243 —— [9,9-bis-(4-methoxy-3-methyl-phenyl)-fluoren-4-yl]-(4-methoxy-3-methyl-phenyl)-ketone 500360-94-1 C38H34O4 554.686 —— 9-oxo-fluorene-4-carboxylic acid-(2-diethylamino-ethyl ester) —— C20H21NO3 323.392 —— N-[3-(4-morpholinyl)propyl]-9-oxo-9H-fluorene-4-carboxamide —— C21H22N2O3 350.417 —— N-(3-aminopyridin-4-yl)-9-oxofluorene-4-carboxamide 915138-74-8 C19H13N3O2 315.331 —— 4-iodo-9H-fluoren-9-one 883-33-0 C13H7IO 306.102 —— N-methyl-9-fluorenone-1-carboxamide 1193434-68-2 C15H11NO2 237.258 —— ((2S,3R)-3,6-Dihydro-6-oxo-(2E-prop-1-enyl-2H-pyran-3-yl))-9-oxo-9H-fluorene-4-amide 247188-38-1 C22H17NO4 359.381 - 1
- 2
反应信息
-
作为反应物:描述:9-芴酮-4-甲酰氯 在 吡啶 、 溶剂黄146 、 三乙胺 作用下, 以 二氯甲烷 、 苯 为溶剂, 反应 26.0h, 生成 9-fluorenylidenemalononitrile-4-carboxylic acid N-octadecylamide参考文献:名称:具有多种可寻址特性的芴衍生物:合成,表征和反应性摘要:从目标化合物N十八烷基9氧代9 H芴4羧酰胺(3)合成了一系列新的芴衍生物,从而通过与各种试剂反应生成了功能化的五元和六元杂环衍生物。允许合成的化合物与环氧丙烷反应以提供独特的表面活性。通过光谱和分析工具阐明并确认了新合成化合物的化学结构。合成的化合物具有良好的表面活性,并且这些芴基表面活性剂的生物降解在7天后大于96%。DOI:10.1007/s11743-017-1958-4
-
作为产物:描述:参考文献:名称:Pick, Monatshefte fur Chemie, 1904, vol. 25, p. 980摘要:DOI:
文献信息
-
Metathesis-active ligands enable a catalytic functional group metathesis between aroyl chlorides and aryl iodides作者:Yong Ho Lee、Bill MorandiDOI:10.1038/s41557-018-0078-8日期:2018.10for the most part, relied on relatively strong driving forces which often require highly reactive reagents to generate irreversibly a desired product in high yield and selectivity. These approaches generally prevent the use of the same catalytic strategy to perform the reverse reaction. Here we describe a catalytic functional group metathesis approach to interconvert, under CO-free conditions, two当前用于官能团互变的方法大部分依赖于相对强的驱动力,该驱动力通常需要高反应性的试剂以高收率和选择性不可逆地产生所需产物。这些方法通常阻止使用相同的催化策略进行逆反应。在这里,我们描述了一种催化官能团复分解方法,可在无CO条件下相互转化两类重要的亲电试剂,通常用于制备药物和农用化学品:芳酰氯(ArCOCl)和芳基碘化物(ArI)。我们的反应设计依赖于关键的可逆配体C–P键裂解事件的实施,这可以实现非清白的,复分解活性膦配体介导两个不同亲电试剂之间快速的芳基转移。除了为ArCOCl和ArI的相互转化提供一种实用且更安全的方法外,这种类型的配体非无毒性为开发使用合成相关的芳基亲电试剂的各种官能团复分解反应提供了一个蓝图。
-
Palladium-catalysed carboformylation of alkynes using acid chlorides as a dual carbon monoxide and carbon source作者:Yong Ho Lee、Elliott H. Denton、Bill MorandiDOI:10.1038/s41557-020-00621-x日期:2021.2Hydroformylation, a reaction that installs both a C–H bond and an aldehyde group across an unsaturated substrate, is one of the most important catalytic reactions in both industry and academia. Given the synthetic importance of creating new C–C bonds, the development of carboformylation reactions, wherein a new C–C bond is formed instead of a C–H bond, would bear enormous synthetic potential to rapidly
-
Scavenger assisted combinatorial process for preparing libraries of amides, carbamates and sulfonamides申请人:ELI LILLY AND COMPANY公开号:EP0825164A2公开(公告)日:1998-02-25This invention relates to a novel solution phase process for the preparation of amide, carbamate, and sulfonamide combinatorial libraries. These libraries have utility for drug discovery and are used to form wellplate components of novel assay kits.这项发明涉及一种用于制备酰胺、碳酸酯和磺酰胺组合库的新型溶液相过程。这些库在药物发现中具有实用价值,并用于形成新型检测套件的微孔板组件。
-
Cytokine production inhibitors申请人:Terragen Discovery Inc.公开号:US06197811B1公开(公告)日:2001-03-06A compound selected from the group consisting of a 5,6-dihydro-&agr;-pyrone of formula (I): wherein bond a is oriented either or ; X is O or NH; R1 is selected from the group consisting of: a group R3(O)C— wherein R3 is selected from the group consisting of: (i) a group of formula R4—CH═CH— wherein R4 is C1-C20 alkyl, C2-C20 alkenyl, an aryl group, or a 5- or 6-membered unsaturated heterocyclic ring containing one or two O, N or S atoms, (ii) C2-C20 alkyl; and (iii) an aryl group or a substituent which is a fused ring system of formula (1) or (2): wherein each of R′ and R″, which are the same or different and may occupy any position on ring a or ring b of said fused ring system, is H or C1-C6 alkyl; a group ArCH2— wherein Ar is an aryl group; and a group R5O—CH2— wherein R5 is C1-C6 alkyl optionally interrupted by one or two O atoms; and R2 is CH3 or, when R1 is a group of formula (A) as defined below, R2 is R6OOC— or R6NOC— wherein R6 is C1-C6 alkyl; and the pharmaceutically and veterinarily acceptable salts and esters thereof; with the exception of compounds wherein bond a is oriented , X is O and R1 is a group of formula (A): wherein R11 and R21 are H when R2 in formula (I) is CO2H or CH3, or one of R11 and R21 is H and the other is OH when R2 in formula (I) is CO2H.从以下组中选择的一种化合物,为式(I)的5,6-二氢-α-吡喃:其中键a朝向任一或;X为O或NH;R1从以下组中选择:一组R3(O)C—,其中R3从以下组中选择:(i)一个式R4—CH═CH—的基团,其中R4为C1-C20烷基,C2-C20烯基,芳基,或含有一个或两个O、N或S原子的5-或6-成员不饱和杂环环,(ii)C2-C20烷基;以及(iii)芳基或一个式(1)或(2)的融合环系统的取代基:其中R′和R″分别为H或C1-C6烷基,它们相同或不同且可以占据所述融合环系统的环a或环b上的任意位置;一组Ar —,其中Ar为芳基;和一组R5O—CH2—,其中R5为C1-C6烷基,可选地被一个或两个O原子中断;R2为CH3或者当R1为下述式(A)的基团时,R2为R6OOC—或R6NOC—,其中R6为C1-C6烷基;以及其药学和兽医学上可接受的盐和酯;除了键a朝向,X为O且R1为式(A)的基团的化合物。
-
Bichromophoric Perylene Derivatives: Energy Transfer from Non-Fluorescent Chromophores作者:Heinz Langhals、Sigrid SaulichDOI:10.1002/1521-3765(20021216)8:24<5630::aid-chem5630>3.0.co;2-z日期:2002.12.16The energy of excitation at non-fluorescent chromophores such as fluorenone and anthraquinone has been trapped by a fast energy transfer to the highly fluorescent perylene bisimides. To this end, anthraquinone, fluorenone and anthracene derivatives have been linked to the perylene bisimides by non-conjugating spacers and fluorescence quantum yield of such assemblies have been determined as a function
表征谱图
-
氢谱1HNMR
-
质谱MS
-
碳谱13CNMR
-
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(S)-2-N-Fmoc-氨基甲基吡咯烷盐酸盐
(2S,4S)-Fmoc-4-三氟甲基吡咯烷-2-羧酸
黎芦碱
鳥胺酸
魏因勒卜链接剂
雷迪帕韦二丙酮合物
雷迪帕韦中间体6
雷迪帕韦
雷迪帕维中间体
雷迪帕维中间体
雷尼托林
锰(2+)二{[乙酰基(9H-芴-2-基)氨基]氧烷负离子}
醋酸丁酸纤维素
达托霉素杂质
赖氨酸杂质4
试剂9,9-Dioctyl-9H-fluoren-2-amine
螺[环戊烷-1,9'-芴]
螺[环庚烷-1,9'-芴]
螺[环己烷-1,9'-芴]
螺[3.3]庚烷-2,6-二-(2',2'',7',7''-四碘螺芴)
螺-(金刚烷-2,9'-芴)
螺(环己烷-1,9'-芴)-3-酮
藜芦托素
荧蒽 反式-2,3-二氢二醇
草甘膦-FMOC
英地卡胺
苯芴醇杂质A
苯甲酸-(芴-9-基-苯基-甲基酯)
苯甲酸-(9-苯基-芴-9-基酯)
苯并[b]芴铯盐
苯并[a]芴酮
苯基芴胺
苯基(9-苯基-9-芴基)甲醇
苯(甲)醛,9H-芴-9-亚基腙
苯(甲)醛,4-羟基-3-甲氧基-,(3-甲基-9H-茚并[2,1-c]吡啶-9-亚基)腙
芴甲氧羰酰胺
芴甲氧羰酰基高苯丙氨酸
芴甲氧羰酰基肌氨酸
芴甲氧羰酰基环己基甘氨酸
芴甲氧羰酰基正亮氨酸
芴甲氧羰酰基D-环己基甘氨酸
芴甲氧羰酰基D-Β环己基丙氨酸
芴甲氧羰酰基-O-三苯甲基丝氨酸
芴甲氧羰酰基-D-正亮氨酸
芴甲氧羰酰基-6-氨基己酸
芴甲氧羰基-高丝氨酸内酯
芴甲氧羰基-缬氨酸-1-13C
芴甲氧羰基-叔丁基二甲基硅-D-丝氨酸
芴甲氧羰基-beta-赖氨酰酸(叔丁氧羰基)
芴甲氧羰基-S-叔丁基-L-半胱氨酸五氟苯基脂