己基甲基二氯硅烷 | 14799-94-1
中文名称
己基甲基二氯硅烷
中文别名
——
英文名称
n-hexylmethyldichlorosilane
英文别名
dichloromethylhexylsilane;Dichlor-hexyl-methyl-silan;hexyl methyl dichlorosilane;dichloro(hexyl)(methyl)silane;1-methyldichlorosilylhexane;1-hexyl-methyldichlorosilane;Hexyl-methyl-dichlorsilan;Hexylmethyldichlorosilane;dichloro-hexyl-methylsilane
CAS
14799-94-1
化学式
C7H16Cl2Si
mdl
MFCD00039295
分子量
199.196
InChiKey
KKRMHVJQWMXYBZ-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:<0°C
-
沸点:204-206 °C(lit.)
-
密度:0.992 g/mL at 20 °C(lit.)
-
闪点:85°C
计算性质
-
辛醇/水分配系数(LogP):3.25
-
重原子数:10
-
可旋转键数:5
-
环数:0.0
-
sp3杂化的碳原子比例:1.0
-
拓扑面积:0
-
氢给体数:0
-
氢受体数:0
安全信息
-
TSCA:Yes
-
危险等级:8
-
危险品标志:C
-
安全说明:S26,S28,S36/37/39,S45
-
危险类别码:R34
-
WGK Germany:1
-
危险品运输编号:UN 2987 8/PG 2
-
海关编码:2931900090
-
包装等级:II
-
危险类别:8
-
危险性防范说明:P260,P264,P280,P301+P330+P331,P303+P361+P353,P304+P340,P305+P351+P338,P310,P321,P363,P405,P501
-
危险性描述:H314
-
储存条件:存放于惰性气体中;避免湿气(否则可能导致分解)。
SDS
Dichlorohexylmethylsilane Revision number: 5
SAFETY DATA SHEET
Section 1. IDENTIFICATION
Product name: Dichlorohexylmethylsilane
Revision number: 5
Section 2. HAZARDS IDENTIFICATION
GHS classification
PHYSICAL HAZARDS
Category 4
Flammable liquids
Corrosive to metals Category 1
HEALTH HAZARDS
Skin corrosion/irritation Category 1B
Category 1
Serious eye damage/eye irritation
ENVIRONMENTAL HAZARDS Not classified
GHS label elements, including precautionary statements
Pictograms or hazard symbols
Signal word Danger
Hazard statements Combustible liquid
May be corrosive to metals
Causes severe skin burns and eye damage
Precautionary statements:
[Prevention] Keep away from flames and hot surfaces.
Keep only in original container.
Do not breathe dust/fume/gas/mist/vapours/spray.
Wash hands thoroughly after handling.
Wear protective gloves and eye/face protection.
IF INHALED: Remove victim to fresh air and keep at rest in a position comfortable for
[Response]
breathing.
IF SWALLOWED: Rinse mouth. Do NOT induce vomiting.
IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses,
if present and easy to do. Continue rinsing.
IF ON SKIN (or hair): Remove/Take off immediately all contaminated clothing. Rinse
skin with water/shower.
Wash contaminated clothing before reuse.
Immediately call a POISON CENTER or doctor/physician.
Absorb spillage to prevent material damage.
[Storage] Store in a well-ventilated place. Keep cool.
Store locked up.
Dichlorohexylmethylsilane
Section 2. HAZARDS IDENTIFICATION
[Disposal] Dispose of contents/container through a waste management company authorized by
the local government.
Section 3. COMPOSITION/INFORMATION ON INGREDIENTS
Substance/mixture: Substance
Components: Dichlorohexylmethylsilane
>95.0%(GC)
Percent:
CAS Number: 14799-94-1
Hexylmethyldichlorosilane
Synonyms:
Chemical Formula: C7H16Cl2Si
Section 4. FIRST AID MEASURES
Inhalation: Remove victim to fresh air and keep at rest in a position comfortable for breathing.
Immediately call a POISON CENTER or doctor/physician.
Skin contact: Remove/Take off immediately all contaminated clothing. Gently wash with plenty of
soap and water. Immediately call a POISON CENTER or doctor/physician.
Eye contact: Rinse cautiously with water for several minutes. Remove contact lenses, if present
and easy to do. Continue rinsing.Immediately call a POISON CENTER or
doctor/physician.
Immediately call a POISON CENTER or doctor/physician. Rinse mouth. Do NOT
Ingestion:
induce vomiting.
Protection of first-aiders: A rescuer should wear personal protective equipment, such as rubber gloves and air-
tight goggles.
Section 5. FIRE-FIGHTING MEASURES
Suitable extinguishing Dry chemical, carbon dioxide.
media:
Unsuitable extinguishing Water
media:
Specific hazards arising Take care as it may decompose upon combustion or in high temperatures to
generate poisonous fume.
from the chemical:
Precautions for firefighters: Fire-extinguishing work is done from the windward and the suitable fire-extinguishing
method according to the surrounding situation is used. Uninvolved persons should
evacuate to a safe place. In case of fire in the surroundings: Remove movable
containers if safe to do so.
Special protective When extinguishing fire, be sure to wear personal protective equipment.
equipment for firefighters:
Section 6. ACCIDENTAL RELEASE MEASURES
Personal precautions, Use extra personal protective equipment (self-contained breathing apparatus). Keep
protective equipment and people away from and upwind of spill/leak. Ensure adequate ventilation. Entry to non-
emergency procedures: involved personnel should be controlled around the leakage area by roping off, etc.
Environmental precautions: Prevent product from entering drains.
Methods and materials for Absorb spilled material in a suitable absorbent (e.g. rag, dry sand, earth, saw-dust).
containment and cleaning In case of large amount of spillage, contain a spill by bunding. Adhered or collected
up: material should be promptly disposed of, in accordance with appropriate laws and
regulations.
Prevention of secondary Do not allow contact with water. Remove all sources of ignition. Fire-extinguishing
hazards: devices should be prepared in case of a fire. Use spark-proof tools and explosion-
proof equipment.
Section 7. HANDLING AND STORAGE
Precautions for safe handling
Dichlorohexylmethylsilane
Section 7. HANDLING AND STORAGE
Technical measures: Handling is performed in a well ventilated place. Wear suitable protective equipment.
Prevent generation of vapour or mist. Keep away from flames and hot surfaces. Take
measures to prevent the build up of electrostatic charge. Use explosion-proof
equipment. Wash hands and face thoroughly after handling.
Use a closed system, ventilation.
Advice on safe handling: Avoid contact with skin, eyes and clothing.
May develop pressure. Open carefully.
Use corrosive resistant equipment.
Conditions for safe storage, including any
incompatibilities
Storage conditions: Keep container tightly closed. Store in a cool, dark and well-ventilated place.
Store under inert gas.
Protect from moisture.
Store locked up.
Store away from incompatible materials such as oxidizing agents.
Moisture-sensitive
Comply with laws. Keep only in original container.
Packaging material:
Section 8. EXPOSURE CONTROLS / PERSONAL PROTECTION
Engineering controls: Install a closed system or local exhaust. Also install safety shower and eye bath.
Personal protective equipment
Respiratory protection: Half or full facepiece respirator, self-contained breathing apparatus(SCBA), supplied
air respirator, etc. Use respirators approved under appropriate government standards
and follow local and national regulations.
Impervious gloves.
Hand protection:
Eye protection: Safety goggles. A face-shield, if the situation requires.
Skin and body protection: Impervious protective clothing. Protective boots, if the situation requires.
Section 9. PHYSICAL AND CHEMICAL PROPERTIES
Physical state (20°C): Liquid
Form: Clear
Colorless - Pale reddish yellow
Colour:
Odour: No data available
pH: No data available
Melting point/freezing point:No data available
Boiling point/range: 103°C/6.7kPa
Flash point: 85°C
Flammability or explosive
limits:
Lower: No data available
Upper: No data available
Vapour density: >1
Relative density: 0.99
Solubility(ies):
[Water] No data available
[Other solvents] No data available
Section 10. STABILITY AND REACTIVITY
Chemical stability: Stable under proper conditions.
Possibility of hazardous Decomposes in contact with water and liberates toxic gases.
reactions:
Conditions to avoid: Open flame, Moisture
Incompatible materials: Oxidizing agents
Hazardous decomposition Carbon monoxide, Carbon dioxide, Hydrogen chloride, Silicon oxides
products:
Dichlorohexylmethylsilane
Section 11. TOXICOLOGICAL INFORMATION
Acute Toxicity: No data available
Skin corrosion/irritation: No data available
Serious eye No data available
damage/irritation:
Germ cell mutagenicity: No data available
Carcinogenicity:
IARC = No data available
No data available
NTP =
Reproductive toxicity: No data available
Section 12. ECOLOGICAL INFORMATION
Ecotoxicity:
Fish: No data available
Crustacea: No data available
Algae: No data available
Persistence / degradability: No data available
No data available
Bioaccumulative
potential(BCF):
Mobility in soil
Log Pow: No data available
Soil adsorption (Koc): No data available
Henry's Law No data available
constant(PaM3/mol):
Section 13. DISPOSAL CONSIDERATIONS
Recycle to process, if possible. Consult your local regional authorities. You may be able to burn in a chemical
incinerator equipped with an afterburner and scrubber system. Observe all federal, state and local regulations when
disposing of the substance.
Section 14. TRANSPORT INFORMATION
Hazards Class: 8: Corrosive.
UN-No: 2987
Proper shipping name: Chlorosilanes, corrosive, n.o.s.
Packing group: II
Section 15. REGULATORY INFORMATION
Safe management ordinance of dangerous chemical product (State Council announces on January 26, 2002
and revised on February 16,2011): Safe use and production, the storage of a dangerous chemical, transport,
loading and unloading were prescribed.
SECTION 16 - ADDITIONAL INFORMATION
N/A
SAFETY DATA SHEET
Section 1. IDENTIFICATION
Product name: Dichlorohexylmethylsilane
Revision number: 5
Section 2. HAZARDS IDENTIFICATION
GHS classification
PHYSICAL HAZARDS
Category 4
Flammable liquids
Corrosive to metals Category 1
HEALTH HAZARDS
Skin corrosion/irritation Category 1B
Category 1
Serious eye damage/eye irritation
ENVIRONMENTAL HAZARDS Not classified
GHS label elements, including precautionary statements
Pictograms or hazard symbols
Signal word Danger
Hazard statements Combustible liquid
May be corrosive to metals
Causes severe skin burns and eye damage
Precautionary statements:
[Prevention] Keep away from flames and hot surfaces.
Keep only in original container.
Do not breathe dust/fume/gas/mist/vapours/spray.
Wash hands thoroughly after handling.
Wear protective gloves and eye/face protection.
IF INHALED: Remove victim to fresh air and keep at rest in a position comfortable for
[Response]
breathing.
IF SWALLOWED: Rinse mouth. Do NOT induce vomiting.
IF IN EYES: Rinse cautiously with water for several minutes. Remove contact lenses,
if present and easy to do. Continue rinsing.
IF ON SKIN (or hair): Remove/Take off immediately all contaminated clothing. Rinse
skin with water/shower.
Wash contaminated clothing before reuse.
Immediately call a POISON CENTER or doctor/physician.
Absorb spillage to prevent material damage.
[Storage] Store in a well-ventilated place. Keep cool.
Store locked up.
Dichlorohexylmethylsilane
Section 2. HAZARDS IDENTIFICATION
[Disposal] Dispose of contents/container through a waste management company authorized by
the local government.
Section 3. COMPOSITION/INFORMATION ON INGREDIENTS
Substance/mixture: Substance
Components: Dichlorohexylmethylsilane
>95.0%(GC)
Percent:
CAS Number: 14799-94-1
Hexylmethyldichlorosilane
Synonyms:
Chemical Formula: C7H16Cl2Si
Section 4. FIRST AID MEASURES
Inhalation: Remove victim to fresh air and keep at rest in a position comfortable for breathing.
Immediately call a POISON CENTER or doctor/physician.
Skin contact: Remove/Take off immediately all contaminated clothing. Gently wash with plenty of
soap and water. Immediately call a POISON CENTER or doctor/physician.
Eye contact: Rinse cautiously with water for several minutes. Remove contact lenses, if present
and easy to do. Continue rinsing.Immediately call a POISON CENTER or
doctor/physician.
Immediately call a POISON CENTER or doctor/physician. Rinse mouth. Do NOT
Ingestion:
induce vomiting.
Protection of first-aiders: A rescuer should wear personal protective equipment, such as rubber gloves and air-
tight goggles.
Section 5. FIRE-FIGHTING MEASURES
Suitable extinguishing Dry chemical, carbon dioxide.
media:
Unsuitable extinguishing Water
media:
Specific hazards arising Take care as it may decompose upon combustion or in high temperatures to
generate poisonous fume.
from the chemical:
Precautions for firefighters: Fire-extinguishing work is done from the windward and the suitable fire-extinguishing
method according to the surrounding situation is used. Uninvolved persons should
evacuate to a safe place. In case of fire in the surroundings: Remove movable
containers if safe to do so.
Special protective When extinguishing fire, be sure to wear personal protective equipment.
equipment for firefighters:
Section 6. ACCIDENTAL RELEASE MEASURES
Personal precautions, Use extra personal protective equipment (self-contained breathing apparatus). Keep
protective equipment and people away from and upwind of spill/leak. Ensure adequate ventilation. Entry to non-
emergency procedures: involved personnel should be controlled around the leakage area by roping off, etc.
Environmental precautions: Prevent product from entering drains.
Methods and materials for Absorb spilled material in a suitable absorbent (e.g. rag, dry sand, earth, saw-dust).
containment and cleaning In case of large amount of spillage, contain a spill by bunding. Adhered or collected
up: material should be promptly disposed of, in accordance with appropriate laws and
regulations.
Prevention of secondary Do not allow contact with water. Remove all sources of ignition. Fire-extinguishing
hazards: devices should be prepared in case of a fire. Use spark-proof tools and explosion-
proof equipment.
Section 7. HANDLING AND STORAGE
Precautions for safe handling
Dichlorohexylmethylsilane
Section 7. HANDLING AND STORAGE
Technical measures: Handling is performed in a well ventilated place. Wear suitable protective equipment.
Prevent generation of vapour or mist. Keep away from flames and hot surfaces. Take
measures to prevent the build up of electrostatic charge. Use explosion-proof
equipment. Wash hands and face thoroughly after handling.
Use a closed system, ventilation.
Advice on safe handling: Avoid contact with skin, eyes and clothing.
May develop pressure. Open carefully.
Use corrosive resistant equipment.
Conditions for safe storage, including any
incompatibilities
Storage conditions: Keep container tightly closed. Store in a cool, dark and well-ventilated place.
Store under inert gas.
Protect from moisture.
Store locked up.
Store away from incompatible materials such as oxidizing agents.
Moisture-sensitive
Comply with laws. Keep only in original container.
Packaging material:
Section 8. EXPOSURE CONTROLS / PERSONAL PROTECTION
Engineering controls: Install a closed system or local exhaust. Also install safety shower and eye bath.
Personal protective equipment
Respiratory protection: Half or full facepiece respirator, self-contained breathing apparatus(SCBA), supplied
air respirator, etc. Use respirators approved under appropriate government standards
and follow local and national regulations.
Impervious gloves.
Hand protection:
Eye protection: Safety goggles. A face-shield, if the situation requires.
Skin and body protection: Impervious protective clothing. Protective boots, if the situation requires.
Section 9. PHYSICAL AND CHEMICAL PROPERTIES
Physical state (20°C): Liquid
Form: Clear
Colorless - Pale reddish yellow
Colour:
Odour: No data available
pH: No data available
Melting point/freezing point:No data available
Boiling point/range: 103°C/6.7kPa
Flash point: 85°C
Flammability or explosive
limits:
Lower: No data available
Upper: No data available
Vapour density: >1
Relative density: 0.99
Solubility(ies):
[Water] No data available
[Other solvents] No data available
Section 10. STABILITY AND REACTIVITY
Chemical stability: Stable under proper conditions.
Possibility of hazardous Decomposes in contact with water and liberates toxic gases.
reactions:
Conditions to avoid: Open flame, Moisture
Incompatible materials: Oxidizing agents
Hazardous decomposition Carbon monoxide, Carbon dioxide, Hydrogen chloride, Silicon oxides
products:
Dichlorohexylmethylsilane
Section 11. TOXICOLOGICAL INFORMATION
Acute Toxicity: No data available
Skin corrosion/irritation: No data available
Serious eye No data available
damage/irritation:
Germ cell mutagenicity: No data available
Carcinogenicity:
IARC = No data available
No data available
NTP =
Reproductive toxicity: No data available
Section 12. ECOLOGICAL INFORMATION
Ecotoxicity:
Fish: No data available
Crustacea: No data available
Algae: No data available
Persistence / degradability: No data available
No data available
Bioaccumulative
potential(BCF):
Mobility in soil
Log Pow: No data available
Soil adsorption (Koc): No data available
Henry's Law No data available
constant(PaM3/mol):
Section 13. DISPOSAL CONSIDERATIONS
Recycle to process, if possible. Consult your local regional authorities. You may be able to burn in a chemical
incinerator equipped with an afterburner and scrubber system. Observe all federal, state and local regulations when
disposing of the substance.
Section 14. TRANSPORT INFORMATION
Hazards Class: 8: Corrosive.
UN-No: 2987
Proper shipping name: Chlorosilanes, corrosive, n.o.s.
Packing group: II
Section 15. REGULATORY INFORMATION
Safe management ordinance of dangerous chemical product (State Council announces on January 26, 2002
and revised on February 16,2011): Safe use and production, the storage of a dangerous chemical, transport,
loading and unloading were prescribed.
SECTION 16 - ADDITIONAL INFORMATION
N/A
上下游信息
反应信息
-
作为反应物:参考文献:名称:硅上甲基的催化功能化:铱催化的甲基氯硅烷的 C(sp3)-H 硼酸化摘要:在 80 °C 的环己烷中,甲基氯硅烷的甲基与双(频哪醇)二硼在铱催化反应中发生 CH 硼酰化,从而选择性地生成(硼基甲基)氯硅烷。DOI:10.1021/ja307956w
-
作为产物:参考文献:名称:四-三苯基膦-铂(0),催化剂摘要:四-三苯基膦-铂(0)选择性催化硅烷加成至末端烯烃双键。该催化剂既不发生内部双键的氢化硅烷化也不发生异构化。DOI:10.1002/hlca.19710540510
文献信息
-
Catalytic Synthesis of Functional Silicon-Stereogenic Silanes through<i>Candida antarctica</i>Lipase B Catalyzed Remote Desymmetrization of Silicon-Centered Diols作者:Xing Lu、Li Li、Wei Yang、Kezhi Jiang、Ke-Fang Yang、Zhan-Jiang Zheng、Li-Wen XuDOI:10.1002/ejoc.201300932日期:2013.9A series of silicon-containing diols are synthesized and used in lipase-catalyzed remote desymmetrization. This synthetic method is valuable in the construction of optically active silicon-stereogenic organosilicon compounds. Good enantioselectivities of the remote desymmetrization was achieved with Candida antarctica lipase B (CAL-B) (up to 90:10er).
-
[EN] PROCESS FOR THE STEPWISE SYNTHESIS OF SILAHYDROCARBONS<br/>[FR] PROCÉDÉ DE SYNTHÈSE SÉQUENTIELLE DE SILAHYDROCARBURES申请人:MOMENTIVE PERFORMANCE MAT INC公开号:WO2021243137A1公开(公告)日:2021-12-02The invention relates to a process for the stepwise synthesis of silahydrocarbons bearing up to four different organyl substituents at the silicon atom, wherein the process includes at least one step a) of producing a bifunctional hydridochlorosilane by a redistribution reaction, selective chlorination of hydridosilanes with an ether/HCI reagent, or by selective chlorination of hydridosilanes with SiCI4, at least one step b) of submitting a bifunctional hydridochloromonosilane to a hydrosilylation reaction, at least one step c) of hydrogenation of a chloromonosilane, and a step d) in which a silahydrocarbon compound is obtained in a hydrosilylation reaction.
-
Catalytic oxidation of diorganosilanes to 1,1,3,3-tetraorganodisiloxanes with gold nanoparticle assembly at the water–chloroform interface作者:Ravi Shankar、Asmita Sharma、Bhawana Jangir、Manchal Chaudhary、Gabriele Kociok-KöhnDOI:10.1039/c8nj04223c日期:——The formation of the spherical self-assembly of gold nanoparticles (AuNPs) of 200 ± 20 nm size at the water–chloroform interface is achieved by employing the cyclotetrasiloxane [RSCH2CH2SiMeO]4 (R = CH2CH2OH) as the stabilizing ligand. The interfacially stabilized AuNPs act as a versatile catalyst for selective hydrolytic oxidation of only one of the Si–H bonds in secondary organosilanes, RR1SiH2 (R
-
Dehydrohalogenative coupling reaction of organic halides with silanes申请人:Korea Institute of Science and Technology公开号:US06251057B1公开(公告)日:2001-06-26The present invention relates to methods for making the compounds of formula I which is a dehydrohalogenative coupling of hydrochlorosilanes of formula II with organic halides of formula III in the presence of a Lewis base catalyst. R3CH2SiR1Cl2 (I) HSiR1Cl2 (II) R2CH2X (III) In formulas I and II, R1 represents a hydrogen, chloro, or methyl; in formula III, X represents a chloro or bromo; in formula III, R2 can be selected from the group consisting of a C1-17 alkyl, a C1-10 fluorinated alkyl with partial or full fluorination, a C1-5 alkenyl groups, a silyl group containing alkyls, (CH2)nSiMe3-mClm wherein n is 0 to 2 and m is 0 to 3, aromatic groups, Ar(R′)1 wherein Ar is C6-14 aromatic hydrocarbon, R′ is a C1-4 alkyl, halogen, alkoxy, or vinyl, and q is 0 to 5, a haloalkyl group, (CH2)pX wherein p is 1 to 9 and X is a chloro or bromo; or an aromatic hydrocarbon, Ar CH2X wherein Ar is C6-14 aromatic hydrocarbon and X is a chloro or bromo. in formula I, R3 is the same as R2 in formula III and further, R3 can also be (CH2)pSiR1Cl2 or ArCH2SiR1Cl2 when R2 in formula III is (CH2)pX or ArCH2X, because of the coupling reaction of X with the compound of formula II.本发明涉及一种制备式I化合物的方法,该方法是在Lewis碱催化剂存在下,通过式II的氢氯硅烷与式III的有机卤化物的脱氢卤代偶联而得到的。 R3 SiR1Cl2 (I) HSiR1Cl2 (II) R2 X (III) 在式I和II中,R1代表氢、氯或甲基;在式III中,X代表氯或溴;在式III中,R2可以选择自C1-17烷基、C1-10氟化烷基(部分或完全氟化)、C1-5烯基、含有矽烷基的矽基团(CH2)nSiMe3-mClm,其中n为0至2,m为0至3,芳香族基团、Ar(R′)1,其中Ar为C6-14芳香烃烃基,R′为C1-4烷基、卤素、烷氧基或乙烯基,q为0至5,卤代烷基、( )pX,其中p为1至9,X为氯或溴;或芳香烃基、Ar X,其中Ar为C6-14芳香烃烃基,X为氯或溴。在式I中,R3与式III中的R2相同,此外,R3也可以是( )pSiR1Cl2或Ar SiR1Cl2,当式III中的R2为( )pX或Ar X时,因为X与式II化合物的偶联反应。
-
Hydrosilane synthesis via catalytic hydrogenolysis of halosilanes using a metal-ligand bifunctional iridium catalyst作者:Teruo Beppu、Kei Sakamoto、Yumiko Nakajima、Kazuhiro Matsumoto、Kazuhiko Sato、Shigeru ShimadaDOI:10.1016/j.jorganchem.2018.05.024日期:2018.8halosilanes was catalysed by iridium amido complexes to produce hydrosilanes. Selective monohydrogenolysis of di- and trichlorosilanes similarly proceeded, resulting in the formation of chlorohydrosilanes (R2SiHCl or RSiHCl2) as synthetically important building blocks for various organosilicon compounds. A mechanistic study supported the in-situ formation of an iridium hydride species as a key intermediate
表征谱图
-
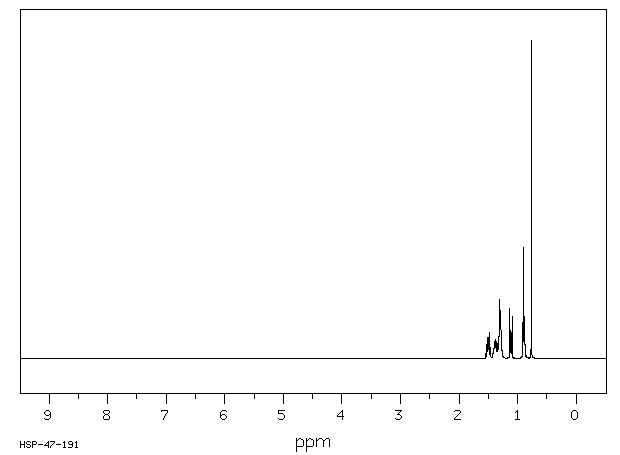
氢谱1HNMR
-
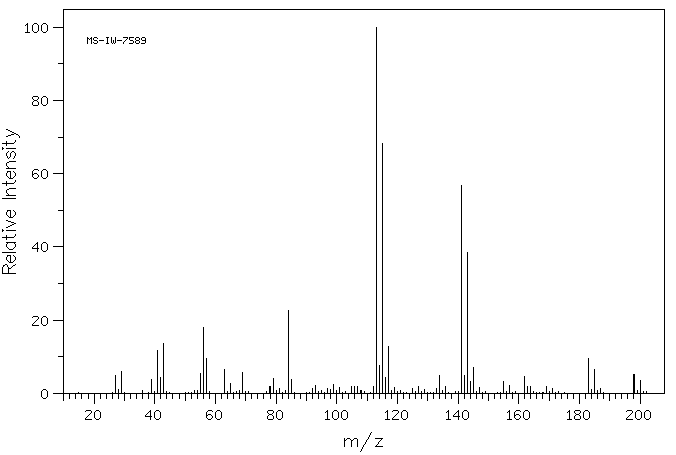
质谱MS
-
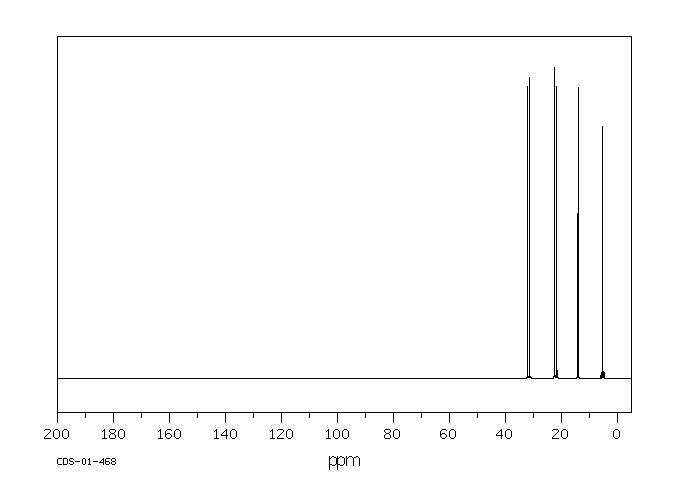
碳谱13CNMR
-
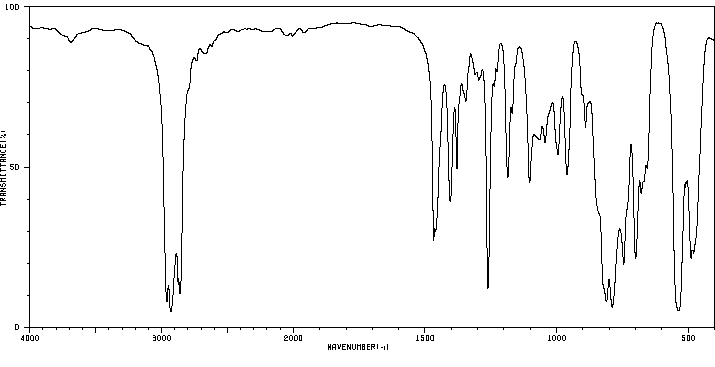
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(2-溴乙氧基)-特丁基二甲基硅烷
鲸蜡基聚二甲基硅氧烷
骨化醇杂质DCP
马沙骨化醇中间体
马来酸双(三甲硅烷)酯
顺式-二氯二(二甲基硒醚)铂(II)
顺-N-(1-(2-乙氧基乙基)-3-甲基-4-哌啶基)-N-苯基苯酰胺
降钙素杂质13
降冰片烯基乙基三甲氧基硅烷
降冰片烯基乙基-POSS
间-氨基苯基三甲氧基硅烷
镓,二(1,1-二甲基乙基)甲基-
镁,氯[[二甲基(1-甲基乙氧基)甲硅烷基]甲基]-
锑,二溴三丁基-
铷,[三(三甲基甲硅烷基)甲基]-
铂(0)-1,3-二乙烯-1,1,3,3-四甲基二硅氧烷
钾(4-{[二甲基(2-甲基-2-丙基)硅烷基]氧基}-1-丁炔-1-基)(三氟)硼酸酯(1-)
金刚烷基乙基三氯硅烷
酰氧基丙基双封头
达格列净杂质
辛醛,8-[[(1,1-二甲基乙基)二甲基甲硅烷基]氧代]-
辛甲基-1,4-二氧杂-2,3,5,6-四硅杂环己烷
辛基铵甲烷砷酸盐
辛基衍生化硅胶(C8)ZORBAX?LP100/40C8
辛基硅三醇
辛基甲基二乙氧基硅烷
辛基三甲氧基硅烷
辛基三氯硅烷
辛基(三苯基)硅烷
辛乙基三硅氧烷
路易氏剂-3
路易氏剂-2
路易士剂
试剂Cyanomethyl[3-(trimethoxysilyl)propyl]trithiocarbonate
试剂3-[Tris(trimethylsiloxy)silyl]propylvinylcarbamate
试剂3-(Trimethoxysilyl)propylvinylcarbamate
试剂2-(Trimethylsilyl)cyclopent-2-en-1-one
试剂11-Azidoundecyltriethoxysilane
西甲硅油杂质14
衣康酸二(三甲基硅基)酯
苯胺,4-[2-(三乙氧基甲硅烷基)乙基]-
苯磺酸,羟基-,盐,单钠聚合甲醛,1,3,5-三嗪-2,4,6-三胺和脲
苯甲醇,a-[(三苯代甲硅烷基)甲基]-
苯并磷杂硅杂英,5,10-二氢-10,10-二甲基-5-苯基-
苯基二甲基氯硅烷
苯基二甲基乙氧基硅
苯基二甲基(2'-甲氧基乙氧基)硅烷
苯基乙酰氧基三甲基硅烷
苯基三辛基硅烷
苯基三甲氧基硅烷