1,4-二(苯乙二酰基)苯 | 3363-97-1
中文名称
1,4-二(苯乙二酰基)苯
中文别名
1,4-二苯氧乙酰基苯;1,4-双(苯乙二酰基)苯
英文名称
1,4-bis(phenylglyoxalyl)benzene
英文别名
1,4-di(phenyloxalyl)benzene;1,4-Bis(phenylglyoxaloyl)benzene;1-[4-(2-oxo-2-phenylacetyl)phenyl]-2-phenylethane-1,2-dione
CAS
3363-97-1
化学式
C22H14O4
mdl
MFCD00039561
分子量
342.351
InChiKey
FUEGWHHUYNHBNI-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:125°C
-
沸点:549.0±33.0 °C(Predicted)
-
密度:1.261±0.06 g/cm3(Predicted)
-
稳定性/保质期:
在常温常压下保持稳定
计算性质
-
辛醇/水分配系数(LogP):4.4
-
重原子数:26
-
可旋转键数:6
-
环数:3.0
-
sp3杂化的碳原子比例:0.0
-
拓扑面积:68.3
-
氢给体数:0
-
氢受体数:4
安全信息
-
危险等级:IRRITANT
-
危险品标志:Xn
-
WGK Germany:2
-
RTECS号:VO6250000
-
海关编码:2914399090
-
安全说明:S22,S29/56,S45
-
危险类别码:R42/43
-
危险性防范说明:P280,P305+P351+P338
-
危险性描述:H317,H319
SDS
1,4-Bisbenzil Revision number: 1
SAFETY DATA SHEET
Section 1. BASE INFORMATION
Product name: 1,4-Bisbenzil
Revision number: 1
Section 2. HAZARDS IDENTIFICATION
Classification of the GHS
PHYSICAL HAZARDS Not classified
HEALTH HAZARDS Not classified
Not classified
ENVIRONMENTAL HAZARDS
GHS label elements
None
Pictograms or hazard symbols
Signal word No signal word
None
Hazard statement
Precautionary statements None
Section 3. COMPOSITION/INFORMATION ON INGREDIENTS
Substance/mixture: Substance
Component(s): 1,4-Bisbenzil
Percent: >95.0%(GC)
CAS Number: 3363-97-1
Synonyms: 1,4-Bis(benzoylcarbonyl)benzene
Chemical Formula: C22H14O4
Section 4. FIRST AID MEASURES
Inhalation: Remove victim to fresh air and keep at rest in a position comfortable for breathing.
Get medical advice/attention if you feel unwell.
Skin contact: Remove/Take off immediately all contaminated clothing. Rinse skin with
water/shower. If skin irritation or rash occurs: Get medical advice/attention.
Eye contact: Rinse cautiously with water for several minutes. Remove contact lenses, if present
and easy to do. Continue rinsing. If eye irritation persists: Get medical
advice/attention.
Ingestion: Get medical advice/attention if you feel unwell. Rinse mouth.
A rescuer should wear personal protective equipment, such as rubber gloves and air-
Protection of first-aiders:
tight goggles.
Section 5. FIRE-FIGHTING MEASURES
Suitable extinguishing Dry chemical, foam, water spray, carbon dioxide.
media:
Specific methods: Fire-extinguishing work is done from the windward and the suitable fire-extinguishing
method according to the surrounding situation is used. Uninvolved persons should
evacuate to a safe place. In case of fire in the surroundings: Remove movable
containers if safe to do so.
1,4-Bisbenzil
Section 5. FIRE-FIGHTING MEASURES
Special protective When extinguishing fire, be sure to wear personal protective equipment.
equipment for firefighters:
Section 6. ACCIDENTAL RELEASE MEASURES
Personal precautions, Use personal protective equipment. Keep people away from and upwind of spill/leak.
protective equipment and Entry to non-involved personnel should be controlled around the leakage area by
emergency procedures: roping off, etc.
Environmental precautions: Prevent product from entering drains.
Methods and materials for Sweep dust to collect it into an airtight container, taking care not to disperse it.
containment and cleaning Adhered or collected material should be promptly disposed of, in accordance with
up: appropriate laws and regulations.
Section 7. HANDLING AND STORAGE
Handling
Handling is performed in a well ventilated place. Wear suitable protective equipment.
Technical measures:
Prevent dispersion of dust. Wash hands and face thoroughly after handling.
Use a local exhaust if dust or aerosol will be generated.
Advice on safe handling: Avoid contact with skin, eyes and clothing.
Storage
Storage conditions: Keep container tightly closed. Store in a cool and dark place.
Store away from incompatible materials such as oxidizing agents.
Packaging material: Law is followed.
Section 8. EXPOSURE CONTROLS / PERSONAL PROTECTION
Engineering controls: Install a closed system or local exhaust as possible so that workers should not be
exposed directly. Also install safety shower and eye bath.
Personal protective equipment
Respiratory protection: Dust respirator. Follow local and national regulations.
Protective gloves.
Hand protection:
Eye protection: Safety glasses. A face-shield, if the situation requires.
Skin and body protection: Protective clothing. Protective boots, if the situation requires.
Section 9. PHYSICAL AND CHEMICAL PROPERTIES
Physical state (20°C): Solid
Form: crystal - powder
Slightly pale yellow - Yellow
Color:
Odor: No data available
pH: No data available
Melting point/freezing point:125 °C
Boiling Point/Range: No data available
Flash Point: No data available
Explosive limits
Lower: No data available
No data available
Upper:
Density: No data available
No data available
Solubility:
Section 10. STABILITY AND REACTIVITY
Stability: Stable under proper conditions.
Reactivity: No special reactivity has been reported.
Incompartible materials: oxidizing agents
Hazardous Decomposition Carbon monoxide, Carbon dioxide
Products:
Section 11. TOXICOLOGICAL INFORMATION
Acute Toxicity: No data available
1,4-Bisbenzil
Section 11. TOXICOLOGICAL INFORMATION
Skin corrosion/irritation: No data available
Serious eye No data available
damage/irritation:
Germ cell mutagenicity: No data available
Carcinogenicity:
No data available
IARC =
NTP = No data available
No data available
Reproductive toxicity:
Section 12. ECOLOGICAL INFORMATION
Ecotoxicity:
Fish: No data available
Crustacea: No data available
Algae: No data available
Persistence / degradability: No data available
Bioaccumulative No data available
potential(BCF):
Mobillity in soil
log Pow: No data available
Soil adsorption (Koc): No data available
Henry's Law No data available
constant(PaM3/mol):
Section 13. DISPOSAL CONSIDERATIONS
Recycle to process, if possible. Consult your local regional authorities. You may be able to dissolve or mix material
with a combustible solvent and burn in a chemical incinerator equipped with an afterburner and scrubber system.
Observe all federal, state and local regulations when disposing of the substance.
Section 14. TRANSPORT INFORMATION
Hazards Class: Does not correspond to the classification standard of the United Nations
UN-No: Not Listed
Section 15. REGULATORY INFORMATION
Safe management ordinance of dangerous chemical product (State Council announces on January 26,
2002): Safe use and production, the storage of a dangerous chemical, transport, loading and unloading were
prescribed.
SECTION 16 - ADDITIONAL INFORMATION
N/A
SAFETY DATA SHEET
Section 1. BASE INFORMATION
Product name: 1,4-Bisbenzil
Revision number: 1
Section 2. HAZARDS IDENTIFICATION
Classification of the GHS
PHYSICAL HAZARDS Not classified
HEALTH HAZARDS Not classified
Not classified
ENVIRONMENTAL HAZARDS
GHS label elements
None
Pictograms or hazard symbols
Signal word No signal word
None
Hazard statement
Precautionary statements None
Section 3. COMPOSITION/INFORMATION ON INGREDIENTS
Substance/mixture: Substance
Component(s): 1,4-Bisbenzil
Percent: >95.0%(GC)
CAS Number: 3363-97-1
Synonyms: 1,4-Bis(benzoylcarbonyl)benzene
Chemical Formula: C22H14O4
Section 4. FIRST AID MEASURES
Inhalation: Remove victim to fresh air and keep at rest in a position comfortable for breathing.
Get medical advice/attention if you feel unwell.
Skin contact: Remove/Take off immediately all contaminated clothing. Rinse skin with
water/shower. If skin irritation or rash occurs: Get medical advice/attention.
Eye contact: Rinse cautiously with water for several minutes. Remove contact lenses, if present
and easy to do. Continue rinsing. If eye irritation persists: Get medical
advice/attention.
Ingestion: Get medical advice/attention if you feel unwell. Rinse mouth.
A rescuer should wear personal protective equipment, such as rubber gloves and air-
Protection of first-aiders:
tight goggles.
Section 5. FIRE-FIGHTING MEASURES
Suitable extinguishing Dry chemical, foam, water spray, carbon dioxide.
media:
Specific methods: Fire-extinguishing work is done from the windward and the suitable fire-extinguishing
method according to the surrounding situation is used. Uninvolved persons should
evacuate to a safe place. In case of fire in the surroundings: Remove movable
containers if safe to do so.
1,4-Bisbenzil
Section 5. FIRE-FIGHTING MEASURES
Special protective When extinguishing fire, be sure to wear personal protective equipment.
equipment for firefighters:
Section 6. ACCIDENTAL RELEASE MEASURES
Personal precautions, Use personal protective equipment. Keep people away from and upwind of spill/leak.
protective equipment and Entry to non-involved personnel should be controlled around the leakage area by
emergency procedures: roping off, etc.
Environmental precautions: Prevent product from entering drains.
Methods and materials for Sweep dust to collect it into an airtight container, taking care not to disperse it.
containment and cleaning Adhered or collected material should be promptly disposed of, in accordance with
up: appropriate laws and regulations.
Section 7. HANDLING AND STORAGE
Handling
Handling is performed in a well ventilated place. Wear suitable protective equipment.
Technical measures:
Prevent dispersion of dust. Wash hands and face thoroughly after handling.
Use a local exhaust if dust or aerosol will be generated.
Advice on safe handling: Avoid contact with skin, eyes and clothing.
Storage
Storage conditions: Keep container tightly closed. Store in a cool and dark place.
Store away from incompatible materials such as oxidizing agents.
Packaging material: Law is followed.
Section 8. EXPOSURE CONTROLS / PERSONAL PROTECTION
Engineering controls: Install a closed system or local exhaust as possible so that workers should not be
exposed directly. Also install safety shower and eye bath.
Personal protective equipment
Respiratory protection: Dust respirator. Follow local and national regulations.
Protective gloves.
Hand protection:
Eye protection: Safety glasses. A face-shield, if the situation requires.
Skin and body protection: Protective clothing. Protective boots, if the situation requires.
Section 9. PHYSICAL AND CHEMICAL PROPERTIES
Physical state (20°C): Solid
Form: crystal - powder
Slightly pale yellow - Yellow
Color:
Odor: No data available
pH: No data available
Melting point/freezing point:125 °C
Boiling Point/Range: No data available
Flash Point: No data available
Explosive limits
Lower: No data available
No data available
Upper:
Density: No data available
No data available
Solubility:
Section 10. STABILITY AND REACTIVITY
Stability: Stable under proper conditions.
Reactivity: No special reactivity has been reported.
Incompartible materials: oxidizing agents
Hazardous Decomposition Carbon monoxide, Carbon dioxide
Products:
Section 11. TOXICOLOGICAL INFORMATION
Acute Toxicity: No data available
1,4-Bisbenzil
Section 11. TOXICOLOGICAL INFORMATION
Skin corrosion/irritation: No data available
Serious eye No data available
damage/irritation:
Germ cell mutagenicity: No data available
Carcinogenicity:
No data available
IARC =
NTP = No data available
No data available
Reproductive toxicity:
Section 12. ECOLOGICAL INFORMATION
Ecotoxicity:
Fish: No data available
Crustacea: No data available
Algae: No data available
Persistence / degradability: No data available
Bioaccumulative No data available
potential(BCF):
Mobillity in soil
log Pow: No data available
Soil adsorption (Koc): No data available
Henry's Law No data available
constant(PaM3/mol):
Section 13. DISPOSAL CONSIDERATIONS
Recycle to process, if possible. Consult your local regional authorities. You may be able to dissolve or mix material
with a combustible solvent and burn in a chemical incinerator equipped with an afterburner and scrubber system.
Observe all federal, state and local regulations when disposing of the substance.
Section 14. TRANSPORT INFORMATION
Hazards Class: Does not correspond to the classification standard of the United Nations
UN-No: Not Listed
Section 15. REGULATORY INFORMATION
Safe management ordinance of dangerous chemical product (State Council announces on January 26,
2002): Safe use and production, the storage of a dangerous chemical, transport, loading and unloading were
prescribed.
SECTION 16 - ADDITIONAL INFORMATION
N/A
制备方法与用途
用途:用作聚酰亚胺中间体。
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 —— 1,4-bis (phenylacetyl)benzene 3363-92-6 C22H18O2 314.384 —— p-bis(phenacyl)benzene 24447-21-0 C22H18O2 314.384 2-羟基-1-[4-(2-羟基-2-苯基乙酰基)苯基]-2-苯基乙酮 1,4-Bis-(α-hydroxyphenacetyl)-benzol 59443-08-2 C22H18O4 346.383 1,4-二乙酰苯 1,4-Diacetylbenzene 1009-61-6 C10H10O2 162.188 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 2-羟基-1-[4-(2-羟基-2-苯基乙酰基)苯基]-2-苯基乙酮 1,4-Bis-(α-hydroxyphenacetyl)-benzol 59443-08-2 C22H18O4 346.383
反应信息
-
作为反应物:描述:1,4-二(苯乙二酰基)苯 以 甲酸 、 水 为溶剂, 反应 1.33h, 生成 1,4-bis(5-phenyl-2,4,6,8-tetraaza-3,7-dioxobicyclo<3.3.0>octan-1-yl)benzene参考文献:名称:Bakibaev, A. A.; Savchenko, T. I.; Filimonov, V. D., Journal of Organic Chemistry USSR (English Translation), 1988, vol. 24, # 12, p. 2331 - 2334摘要:DOI:
-
作为产物:描述:参考文献:名称:Bistetracyclones and Bishexaphenylbenzenes. II摘要:DOI:10.1021/jo01021a022
文献信息
-
A Bifunctional Iron Nanocomposite Catalyst for Efficient Oxidation of Alkenes to Ketones and 1,2-Diketones作者:Tao Song、Zhiming Ma、Peng Ren、Youzhu Yuan、Jianliang Xiao、Yong YangDOI:10.1021/acscatal.9b05197日期:2020.4.17sites of Fe–Nx and Fe phosphate, as oxidation and Lewis acid sites, were simultaneously integrated into a hierarchical N,P-dual doped porous carbon. As a bifunctional catalyst, it exhibited high efficiency for direct oxidative cleavage of alkenes into ketones or their oxidation into 1,2-diketones with a broad substrate scope and high functional group tolerance using TBHP as the oxidant in water under
-
One-pot cascade synthesis of α-diketones from aldehydes and ketones in water by using a bifunctional iron nanocomposite catalyst作者:Tao Song、Xin Zhou、Xiaoxue Wang、Jianliang Xiao、Yong YangDOI:10.1039/d0gc03739g日期:——A new methodology for the synthesis of α-diketones was reported via a one-pot cascade process from aldehydes and ketones catalyzed by a bifunctional iron nanocomposite using H2O2 as a green oxidant in water. The one-pot strategy showed excellent catalytic stability, comprehensive suitability of substrates and important practical utility for directly synthesizing biologically active and medicinally
-
Discovery of 4-<i>tert</i>-Butyl-2,6-dimethylphenylsulfur Trifluoride as a Deoxofluorinating Agent with High Thermal Stability as Well as Unusual Resistance to Aqueous Hydrolysis, and Its Diverse Fluorination Capabilities Including Deoxofluoro-Arylsulfinylation with High Stereoselectivity作者:Teruo Umemoto、Rajendra P. Singh、Yong Xu、Norimichi SaitoDOI:10.1021/ja106343h日期:2010.12.29groups to CF(3) groups, in high yields. 1k also converts C(=S) and CH(3)SC(=S)O groups to CF(2) and CF(3)O groups, respectively, in high yields. In addition, 1k effects highly stereoselective deoxofluoro-arylsulfinylation of diols and amino alcohols to give fluoroalkyl arylsulfinates and arylsulfinamides, with complete inversion of configuration at fluorine and the simultaneous, selective formation of学术和工业领域都非常需要多功能、安全、货架稳定和易于处理的氟化剂,因为由于氟原子的独特作用,氟化化合物在许多领域引起了相当大的兴趣,例如药物发现当结合到分子中时。本文描述了许多取代且热稳定的三氟化苯硫的合成、性质和反应性,特别是 4-叔丁基-2,6-二甲基苯硫三氟化物(Fluolead,1k),作为一种结晶固体,在接触时具有惊人的高稳定性与当前的试剂(例如 DAST 及其类似物)相比,具有水和作为脱氧氟化剂的优越效用。1k 上的取代基在热稳定性和水解稳定性、氟化反应性、并阐明了它所经历的高产氟化机理。除了醇、醛和可烯醇化酮的氟化外,1k 还可以高产率顺利地将不可烯醇化的羰基转化为 CF(2) 基团,将羧基转化为 CF(3) 基团。1k 还以高产率分别将 C(=S) 和 CH(3)SC(=S)O 基团转化为 CF(2) 和 CF(3)O 基团。此外,1k 可实现二醇和氨基醇的高度立体选择性脱
-
Electrochemical synthesis of 1,2-diketones from alkynes under transition-metal-catalyst-free conditions作者:Jie Zhou、Xiang-Zhang Tao、Jian-Jun Dai、Chen-Guang Li、Jun Xu、Hong-Mei Xu、Hua-Jian XuDOI:10.1039/c9cc03996a日期:——We report an electrochemical protocol for the direct oxidation of internal alkynes in air to provide 1,2-diketones. A variety of functional groups and heterocycle-containing substrates can be tolerated well under mild conditions.
-
Nature of the Nucleophilic Oxygenation Reagent Is Key to Acid-Free Gold-Catalyzed Conversion of Terminal and Internal Alkynes to 1,2-Dicarbonyls作者:Alexey Yu. Dubovtsev、Nikolay V. Shcherbakov、Dmitry V. Dar’in、Vadim Yu. KukushkinDOI:10.1021/acs.joc.9b02785日期:2020.1.172,3-Dichloropyridine N-oxide, a novel oxygen transfer reagent, allows the conductance of the gold(I)-catalyzed oxidation of alkynes to 1,2-dicarbonyls in the absence of any acid additives and under mild conditions to furnish the target species, including those derivatized by highly acid-sensitive groups. The developed strategy is effective for a wide range of alkyne substrates such as terminal- and
表征谱图
-
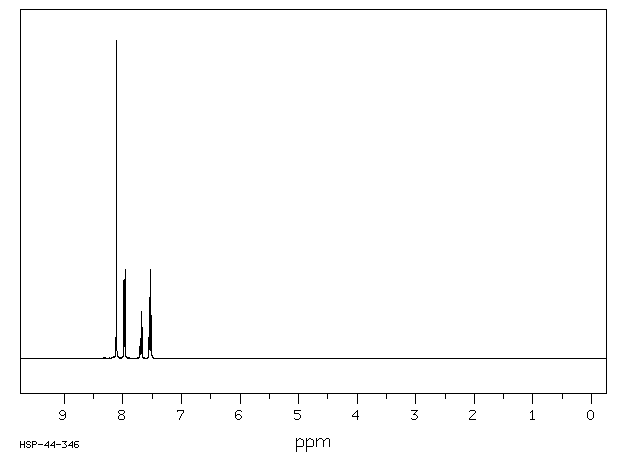
氢谱1HNMR
-
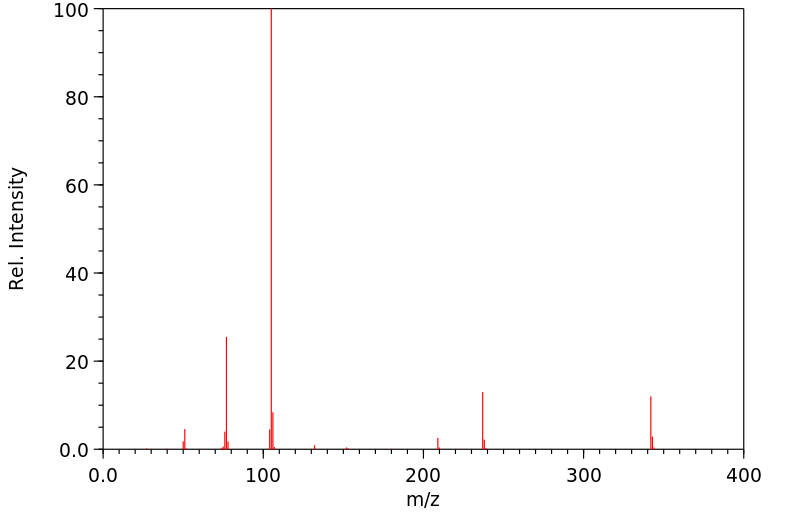
质谱MS
-
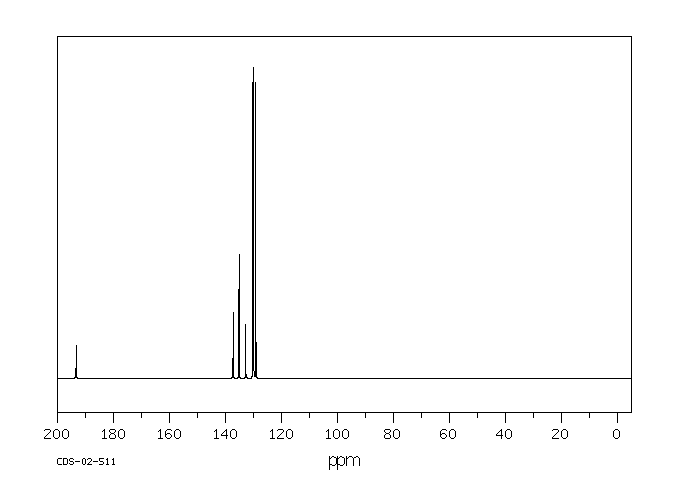
碳谱13CNMR
-
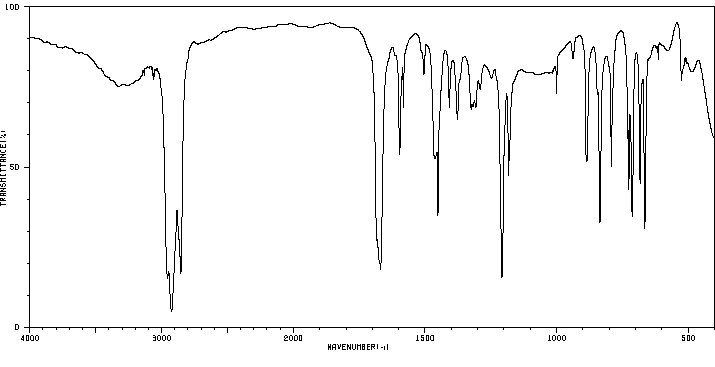
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(E,Z)-他莫昔芬N-β-D-葡糖醛酸
(E/Z)-他莫昔芬-d5
(4S,5R)-4,5-二苯基-1,2,3-恶噻唑烷-2,2-二氧化物-3-羧酸叔丁酯
(4S,4''S,5R,5''R)-2,2''-(1-甲基亚乙基)双[4,5-二氢-4,5-二苯基恶唑]
(4R,5S)-4,5-二苯基-1,2,3-恶噻唑烷-2,2-二氧化物-3-羧酸叔丁酯
(4R,4''R,5S,5''S)-2,2''-(1-甲基亚乙基)双[4,5-二氢-4,5-二苯基恶唑]
(1R,2R)-2-(二苯基膦基)-1,2-二苯基乙胺
鼓槌石斛素
黄子囊素
高黄绿酸
顺式白藜芦醇三甲醚
顺式白藜芦醇
顺式己烯雌酚
顺式-白藜芦醇3-O-beta-D-葡糖苷酸
顺式-桑皮苷A
顺式-曲札芪苷
顺式-二苯乙烯
顺式-beta-羟基他莫昔芬
顺式-a-羟基他莫昔芬
顺式-3,4',5-三甲氧基-3'-羟基二苯乙烯
顺式-1-(3-甲基-2-萘基)-2-(2-萘基)乙烯
顺式-1,2-双(三甲基硅氧基)-1,2-双(4-溴苯基)环丙烷
顺式-1,2-二苯基环丁烷
顺-均二苯乙烯硼酸二乙醇胺酯
顺-4-硝基二苯乙烯
顺-1-异丙基-2,3-二苯基氮丙啶
非洲李(PRUNUSAFRICANA)树皮提取物
阿非昔芬
阿里可拉唑
阿那曲唑二聚体
阿托伐他汀环氧四氢呋喃
阿托伐他汀环氧乙烷杂质
阿托伐他汀环(氟苯基)钠盐杂质
阿托伐他汀环(氟苯基)烯丙基酯
阿托伐他汀杂质D
阿托伐他汀杂质94
阿托伐他汀杂质7
阿托伐他汀杂质5
阿托伐他汀内酰胺钠盐杂质
阿托伐他汀中间体M4
阿奈库碘铵
锌(II)(苯甲醛)(四苯基卟啉)
银松素
铜酸盐(5-),[m-[2-[2-[1-[4-[2-[4-[[4-[[4-[2-[4-[4-[2-[2-(羧基-kO)苯基]二氮烯基-kN1]-4,5-二氢-3-甲基-5-(羰基-kO)-1H-吡唑-1-基]-2-硫代苯基]乙烯基]-3-硫代苯基]氨基]-6-(苯基氨基)-1,3,5-三嗪-2-基]氨基]-2-硫代苯基]乙烯基]-3-硫代
铒(III) 离子载体 I
铀,二(二苯基甲酮)四碘-
钾钠2,2'-[(E)-1,2-乙烯二基]二[5-({4-苯胺基-6-[(2-羟基乙基)氨基]-1,3,5-三嗪-2-基}氨基)苯磺酸酯](1:1:1)
钠{4-[氧代(苯基)乙酰基]苯基}甲烷磺酸酯
钠;[2-甲氧基-5-[2-(3,4,5-三甲氧基苯基)乙基]苯基]硫酸盐
钠4-氨基二苯乙烯-2-磺酸酯