2-(2-萘硫代)乙腈 | 5324-69-6
中文名称
2-(2-萘硫代)乙腈
中文别名
——
英文名称
2-(naphthalen-2-ylthio)acetonitrile
英文别名
2-(naphthalenylthio)acetonitrile;2-Naphthalenylthioacetonitrile;2-Naphthylthioacetonitrile;2-naphthalen-2-ylsulfanylacetonitrile
CAS
5324-69-6
化学式
C12H9NS
mdl
MFCD00067878
分子量
199.276
InChiKey
PFMHSJMACRLMEX-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:83 °C
计算性质
-
辛醇/水分配系数(LogP):3.8
-
重原子数:14
-
可旋转键数:2
-
环数:2.0
-
sp3杂化的碳原子比例:0.083
-
拓扑面积:49.1
-
氢给体数:0
-
氢受体数:2
安全信息
-
危险品标志:Xn
-
安全说明:S36/37
-
危险类别码:R20/21/22
-
海关编码:2930909090
SDS
| Name: | 2-(2-Naphthylthio)acetonitrile 97% Material Safety Data Sheet |
| Synonym: | |
| CAS: | 5324-69-6 |
Synonym:
Section 2 - COMPOSITION, INFORMATION ON INGREDIENTS
| CAS# | Chemical Name | content | EINECS# |
| 5324-69-6 | 2-(2-Naphthylthio)acetonitrile | 97% | unlisted |
Risk Phrases: 20/21/22
Section 3 - HAZARDS IDENTIFICATION
EMERGENCY OVERVIEW
Harmful by inhalation, in contact with skin and if swallowed.
Potential Health Effects
Eye:
May cause eye irritation.
Skin:
May cause skin irritation. Harmful if absorbed through the skin.
Ingestion:
Harmful if swallowed. May cause irritation of the digestive tract.
Inhalation:
Harmful if inhaled. May cause respiratory tract irritation.
Chronic:
Not available.
Section 4 - FIRST AID MEASURES
Eyes: Flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid.
Skin:
Get medical aid. Flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes.
Ingestion:
Get medical aid. Wash mouth out with water.
Inhalation:
Remove from exposure and move to fresh air immediately. If not breathing, give artificial respiration. If breathing is difficult, give oxygen. Get medical aid.
Notes to Physician:
Section 5 - FIRE FIGHTING MEASURES
General Information:
As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear.
Extinguishing Media:
Use water spray, dry chemical, carbon dioxide, or chemical foam.
Section 6 - ACCIDENTAL RELEASE MEASURES
General Information: Use proper personal protective equipment as indicated in Section 8.
Spills/Leaks:
Vacuum or sweep up material and place into a suitable disposal container.
Section 7 - HANDLING and STORAGE
Handling:
Avoid breathing dust, vapor, mist, or gas. Avoid contact with skin and eyes.
Storage:
Store in a cool, dry place. Store in a tightly closed container.
Section 8 - EXPOSURE CONTROLS, PERSONAL PROTECTION
Engineering Controls:
Use adequate ventilation to keep airborne concentrations low.
Exposure Limits CAS# 5324-69-6: Personal Protective Equipment Eyes: Not available.
Skin:
Wear appropriate protective gloves to prevent skin exposure.
Clothing:
Wear appropriate protective clothing to prevent skin exposure.
Respirators:
Follow the OSHA respirator regulations found in 29 CFR 1910.134 or European Standard EN 149. Use a NIOSH/MSHA or European Standard EN 149 approved respirator if exposure limits are exceeded or if irritation or other symptoms are experienced.
Section 9 - PHYSICAL AND CHEMICAL PROPERTIES
Physical State: Solid
Color: off-white
Odor: Not available.
pH: Not available.
Vapor Pressure: Not available.
Viscosity: Not available.
Boiling Point: Not available.
Freezing/Melting Point: 83 - 86 deg C
Autoignition Temperature: Not available.
Flash Point: Not available.
Explosion Limits, lower: Not available.
Explosion Limits, upper: Not available.
Decomposition Temperature:
Solubility in water:
Specific Gravity/Density:
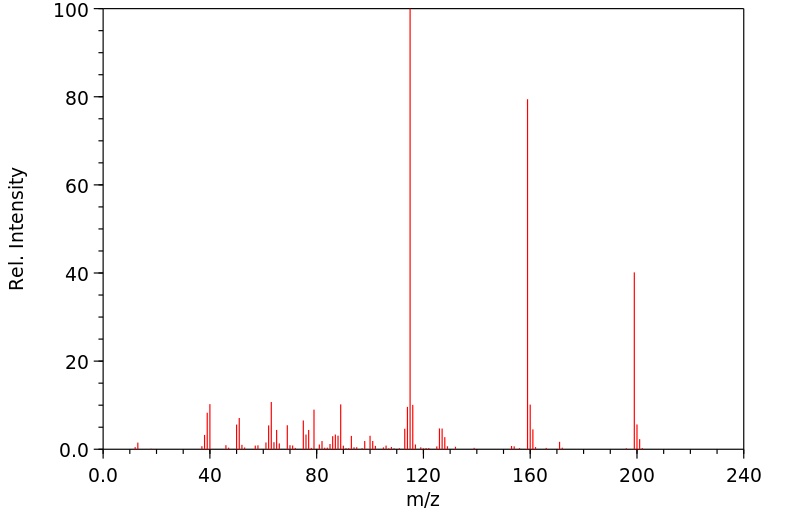
Molecular Formula: C12H9NS
Molecular Weight: 199
Section 10 - STABILITY AND REACTIVITY
Chemical Stability:
Not available.
Conditions to Avoid:
Incompatible materials.
Incompatibilities with Other Materials:
Oxidizing agents, reducing agents.
Hazardous Decomposition Products:
Hydrogen cyanide, nitrogen oxides, carbon monoxide, oxides of sulfur, carbon dioxide.
Hazardous Polymerization: Has not been reported
Section 11 - TOXICOLOGICAL INFORMATION
RTECS#:
CAS# 5324-69-6 unlisted.
LD50/LC50:
Not available.
Carcinogenicity:
2-(2-Naphthylthio)acetonitrile - Not listed by ACGIH, IARC, or NTP.
Section 12 - ECOLOGICAL INFORMATION
Section 13 - DISPOSAL CONSIDERATIONS
Dispose of in a manner consistent with federal, state, and local regulations.
Section 14 - TRANSPORT INFORMATION
IATA
Shipping Name: NITRILES, SOLID, TOXIC, N.O.S.*
Hazard Class: 6.1
UN Number: 3276
Packing Group: III
IMO
Shipping Name: NITRILES, TOXIC, N.O.S.
Hazard Class: 6.1
UN Number: 3276
Packing Group: III
RID/ADR
Shipping Name: NITRILES, TOXIC, N.O.S.
Hazard Class: 6.1
UN Number: 3276
Packing group: III
Section 15 - REGULATORY INFORMATION
European/International Regulations
European Labeling in Accordance with EC Directives
Hazard Symbols: XN
Risk Phrases:
R 20/21/22 Harmful by inhalation, in contact with
skin and if swallowed.
Safety Phrases:
S 36/37 Wear suitable protective clothing and
gloves.
WGK (Water Danger/Protection)
CAS# 5324-69-6: No information available.
Canada
None of the chemicals in this product are listed on the DSL/NDSL list.
CAS# 5324-69-6 is not listed on Canada's Ingredient Disclosure List.
US FEDERAL
TSCA
CAS# 5324-69-6 is not listed on the TSCA inventory.
It is for research and development use only.
SECTION 16 - ADDITIONAL INFORMATION
N/A
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 2-萘硫醇 2-Naphthalenethiol 91-60-1 C10H8S 160.24 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 —— N-Hydroxy-2-(naphthalen-2-ylsulfanyl)-acetamidine 79506-81-3 C12H12N2OS 232.306 2-(2-萘磺酰基)乙腈 2-(naphthalen-2-ylsulfonyl)acetonitrile 32083-60-6 C12H9NO2S 231.275
反应信息
-
作为反应物:描述:参考文献:名称:基于尿肽的布朗斯台德碱的设计,合成及在(对苯磺酰基)乙腈催化对映选择性合成β-氨基腈中的应用摘要:在亚胺上添加氰基烷基部分是制备β-氨基腈的一种非常有吸引力的方法。我们为立体选择性合成β-氨基腈提供了一种高效的有机催化方法,其中成功的关键是将基于尿素肽的布朗斯台德碱催化剂与(芳基磺酰基)乙腈结合使用作为乙腈阴离子的合成当量。该方法使人们能够以良好的收率和出色的对映选择性获得各种β-氨基腈,并拓宽了可用于合成的立体选择性曼尼希型方法。DOI:10.1002/chem.201304877
-
作为产物:描述:2-萘硫醇 以54%的产率得到参考文献:名称:CERVENA I.; HRUBANTOVA M.; BARTOSOVA M.; PROTIVA M., COLLECT. CZECH. CHEM. COMMUN., 1981, 46, NO 5, 1188-1198摘要:DOI:
文献信息
-
Cs2CO3-promoted carbon–sulfur bond construction via cross dehydrogenative coupling of thiophenols with acetonitrile作者:Qian Chen、Yulin Huang、Xiaofeng Wang、Chunxiao Wen、Xinxing Yan、Jiekun ZengDOI:10.1016/j.tetlet.2017.08.067日期:2017.10construction of carbon–sulfur bonds has been achieved via halogen-free Cs2CO3-promoted cross dehydrogenative coupling (CDC) of thiophenols with acetonitrile. This transformation provides a straightforward route to the synthesis of sulfenylated acetonitriles in up to 80% yield.
-
Novel naphthalenyl-3H-1,2,3,5-oxathiadiazole 2-oxides useful as申请人:American Home Products Corporation公开号:US04895861A1公开(公告)日:1990-01-23This invention relates to novel substituted 3H-1,2,3,5-oxathiadiazole 2-oxides, processes for their preparation, to methods for using the compounds, and to pharmaceutical preparations thereof. The compounds have pharmaceutical properties which render them beneficial for the treatment of diabetes mellitus and associated conditions.这项发明涉及新型取代的3H-1,2,3,5-噁硫二唑-2-氧化物,其制备方法,以及使用这些化合物的方法,以及其药物制剂。这些化合物具有药理特性,使它们对于治疗糖尿病及相关疾病有益。
-
Iron-catalysed carbene-transfer reactions of diazo acetonitrile作者:Claire Empel、Katharina J. Hock、Rene M. KoenigsDOI:10.1039/c8ob01991f日期:——A continuous-flow protocol for the synthesis of diazo acetonitrile was developed. It was further applied in iron-catalysed insertion reactions of diazo acetonitrile into N–H and S–H bonds to yield valuable α-substituted acetonitrile, including gram-scale synthesis.
-
Synthesis of arylthioacetamidoximes as potential antidepressants作者:Irena Červená、Marta Hrubantová、Marie Bartošová、Miroslav ProtivaDOI:10.1135/cccc19811188日期:——
Reactions of thiophenol and 16 of its derivatives with chloroacetonitrile afforded arylthioacetonitriles
IVb-XXb which were treated with hydroxylamine and gave arylthioacetamidoximesIVa-XXa . CompoundsVIIa-IXa, XIIa, XVIa andXVIIIa exhibited antireserpine activity in the test of ptosis in mice and compoundsIXa andXVIIa in the test of reserpine hypothermia in mice. -
COMPOSITION, SYNTHESIS, AND USE OF A NEW CLASS OF ISONITRILES申请人:DUQUESNE UNIVERSITY OF THE HOLY GHOST公开号:US20150239835A1公开(公告)日:2015-08-27This invention relates to novel isonitriles, including arylthio isonitriles, and methods for their preparation. The isonitriles include a conjugated ring system. The structure is designed with the flexibility to have multiple substitution patterns. The isonitriles may be used in applications including, but not limited to, pharmaceutical compositions.本发明涉及新型异腈,包括芳基硫代异腈,以及它们的制备方法。异腈包括一个共轭环系统。该结构被设计为具有多种取代模式的灵活性。异腈可用于包括但不限于制药组合物在内的应用中。
表征谱图
-
氢谱1HNMR
-
质谱MS
-
碳谱13CNMR
-
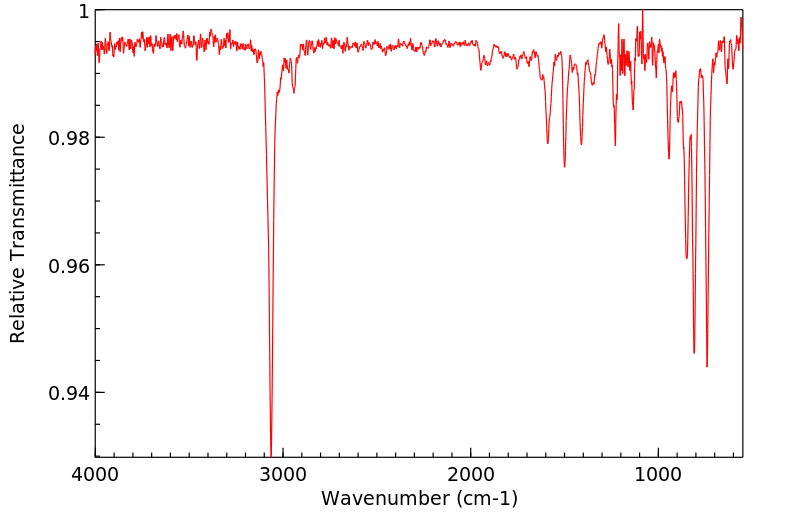
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(S)-溴烯醇内酯
(R)-3,3''-双([[1,1''-联苯]-4-基)-[1,1''-联萘]-2,2''-二醇
(3S,3aR)-2-(3-氯-4-氰基苯基)-3-环戊基-3,3a,4,5-四氢-2H-苯并[g]吲唑-7-羧酸
(3R,3’’R,4S,4’’S,11bS,11’’bS)-(+)-4,4’’-二叔丁基-4,4’’,5,5’’-四氢-3,3’’-联-3H-二萘酚[2,1-c:1’’,2’’-e]膦(S)-BINAPINE
(11bS)-2,6-双(3,5-二甲基苯基)-4-羟基-4-氧化物-萘并[2,1-d:1'',2''-f][1,3,2]二氧磷
(11bS)-2,6-双(3,5-二氯苯基)-4羟基-4-氧-二萘并[2,1-d:1'',2''-f][1,3,2]二氧磷杂七环
(11bR)-2,6-双[3,5-双(1,1-二甲基乙基)苯基]-4-羟基-4-氧化物-二萘并[2,1-d:1'',2''-f][1,3,2]二氧杂磷平
黄胺酸
马兜铃对酮
马休黄钠盐一水合物
马休黄
食品黄6号
食品红40铝盐色淀
飞龙掌血香豆醌
颜料黄101
颜料红70
颜料红63
颜料红53:3
颜料红5
颜料红48单钠盐
颜料红48:2
颜料红4
颜料红261
颜料红258
颜料红220
颜料红22
颜料红214
颜料红2
颜料红19
颜料红185
颜料红184
颜料红170
颜料红148
颜料红147
颜料红146
颜料红119
颜料红114
颜料红 9
颜料红 21
颜料橙7
颜料橙46
颜料橙38
颜料橙3
颜料橙22
颜料橙2
颜料橙17
颜料橙 5
颜料棕1
顺式-阿托伐醌-d5
雄甾烷-3,17-二酮