2-叔丁基-5-甲基苯甲醚 | 88-40-4
中文名称
2-叔丁基-5-甲基苯甲醚
中文别名
——
英文名称
2-tert-butyl-5-methylanisole
英文别名
2-t-Butyl-5-methyl-anisol;Benzene, 1-(1,1-dimethylethyl)-2-methoxy-4-methyl-;1-tert-butyl-2-methoxy-4-methylbenzene
CAS
88-40-4
化学式
C12H18O
mdl
MFCD00009855
分子量
178.274
InChiKey
NFDFEFJTYRAHEM-UHFFFAOYSA-N
BEILSTEIN
——
EINECS
——
-
物化性质
-
计算性质
-
ADMET
-
安全信息
-
SDS
-
制备方法与用途
-
上下游信息
-
文献信息
-
表征谱图
-
同类化合物
-
相关功能分类
-
相关结构分类
物化性质
-
熔点:23-24 °C(Solv: ligroine (8032-32-4))
-
沸点:228 °C
-
密度:0.907±0.06 g/cm3(Predicted)
-
LogP:4.758 (est)
计算性质
-
辛醇/水分配系数(LogP):3.5
-
重原子数:13
-
可旋转键数:2
-
环数:1.0
-
sp3杂化的碳原子比例:0.5
-
拓扑面积:9.2
-
氢给体数:0
-
氢受体数:1
安全信息
-
海关编码:2909309090
SDS
| Name: | 2-t-Butyl-5-methyl-methoxybenzene Material Safety Data Sheet |
| Synonym: | Benzene, 1-(1,1-dimethylethyl)-2-methoxy-4-methyl- |
| CAS: | 88-40-4 |
Synonym:Benzene, 1-(1,1-dimethylethyl)-2-methoxy-4-methyl-
Section 2 - COMPOSITION, INFORMATION ON INGREDIENTS
| CAS# | Chemical Name | content | EINECS# |
| 88-40-4 | 2-t-Butyl-5-methyl-methoxybenzene | 100 | 201-827-1 |
Risk Phrases: 36/37/38
Section 3 - HAZARDS IDENTIFICATION
EMERGENCY OVERVIEW
Irritating to eyes, respiratory system and skin.
Potential Health Effects
Eye:
Causes eye irritation. May cause chemical conjunctivitis.
Skin:
Causes skin irritation.
Ingestion:
May cause gastrointestinal irritation with nausea, vomiting and diarrhea.
Inhalation:
Causes respiratory tract irritation. Can produce delayed pulmonary edema.
Chronic:
No information found.
Section 4 - FIRST AID MEASURES
Eyes: Immediately flush eyes with plenty of water for at least 15 minutes, occasionally lifting the upper and lower eyelids. Get medical aid.
Skin:
Get medical aid. Flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes. Wash clothing before reuse.
Ingestion:
Never give anything by mouth to an unconscious person. Get medical aid. Do NOT induce vomiting. If conscious and alert, rinse mouth and drink 2-4 cupfuls of milk or water.
Inhalation:
Remove from exposure and move to fresh air immediately. If not breathing, give artificial respiration. If breathing is difficult, give oxygen. Get medical aid.
Notes to Physician:
Treat symptomatically and supportively.
Section 5 - FIRE FIGHTING MEASURES
General Information:
As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear. During a fire, irritating and highly toxic gases may be generated by thermal decomposition or combustion.
Extinguishing Media:
Use water spray, dry chemical, carbon dioxide, or appropriate foam.
Section 6 - ACCIDENTAL RELEASE MEASURES
General Information: Use proper personal protective equipment as indicated in Section 8.
Spills/Leaks:
Vacuum or sweep up material and place into a suitable disposal container. Clean up spills immediately, observing precautions in the Protective Equipment section. Avoid generating dusty conditions.
Provide ventilation.
Section 7 - HANDLING and STORAGE
Handling:
Minimize dust generation and accumulation. Avoid contact with eyes, skin, and clothing. Keep container tightly closed. Avoid ingestion and inhalation. Use with adequate ventilation. Wash clothing before reuse.
Storage:
Store in a tightly closed container. Store in a cool, dry, well-ventilated area away from incompatible substances.
Section 8 - EXPOSURE CONTROLS, PERSONAL PROTECTION
Engineering Controls:
Facilities storing or utilizing this material should be equipped with an eyewash facility and a safety shower. Use adequate ventilation to keep airborne concentrations low.
Exposure Limits CAS# 88-40-4: Personal Protective Equipment Eyes: Wear appropriate protective eyeglasses or chemical safety goggles as described by OSHA's eye and face protection regulations in 29 CFR 1910.133 or European Standard EN166.
Skin:
Wear appropriate protective gloves to prevent skin exposure.
Clothing:
Wear appropriate protective clothing to prevent skin exposure.
Respirators:
A respiratory protection program that meets OSHA's 29 CFR 1910.134 and ANSI Z88.2 requirements or European Standard EN 149 must be followed whenever workplace conditions warrant respirator use.
Section 9 - PHYSICAL AND CHEMICAL PROPERTIES
Physical State: Crystals
Color: white
Odor: Not available.
pH: Not available.
Vapor Pressure: Not available.
Viscosity: Not available.
Boiling Point: 222-224 deg C
Freezing/Melting Point: 23-34 deg C
Autoignition Temperature: Not available.
Flash Point: Not available.
Explosion Limits, lower: Not available.
Explosion Limits, upper: Not available.
Decomposition Temperature:
Solubility in water:
Specific Gravity/Density: 0.9331g/ml
Molecular Formula: C12H18O
Molecular Weight: 178.27
Section 10 - STABILITY AND REACTIVITY
Chemical Stability:
Stable at room temperature in closed containers under normal storage and handling conditions.
Conditions to Avoid:
Dust generation.
Incompatibilities with Other Materials:
Strong oxidizing agents.
Hazardous Decomposition Products:
Carbon monoxide, carbon dioxide.
Hazardous Polymerization: Will not occur.
Section 11 - TOXICOLOGICAL INFORMATION
RTECS#:
CAS# 88-40-4 unlisted.
LD50/LC50:
Not available.
Carcinogenicity:
2-t-Butyl-5-methyl-methoxybenzene - Not listed by ACGIH, IARC, or NTP.
Section 12 - ECOLOGICAL INFORMATION
Section 13 - DISPOSAL CONSIDERATIONS
Products which are considered hazardous for supply are classified as Special Waste and the disposal of such chemicals is covered by regulations which may vary according to location. Contact a specialist disposal company or the local waste regulator for advice. Empty containers must be decontaminated before returning for recycling.
Section 14 - TRANSPORT INFORMATION
IATA
Not regulated as a hazardous material.
IMO
Not regulated as a hazardous material.
RID/ADR
Not regulated as a hazardous material.
Section 15 - REGULATORY INFORMATION
European/International Regulations
European Labeling in Accordance with EC Directives
Hazard Symbols: XI
Risk Phrases:
R 36/37/38 Irritating to eyes, respiratory system
and skin.
Safety Phrases:
S 36/37/39 Wear suitable protective clothing, gloves
and eye/face protection.
WGK (Water Danger/Protection)
CAS# 88-40-4: No information available.
Canada
CAS# 88-40-4 is listed on Canada's DSL List.
CAS# 88-40-4 is not listed on Canada's Ingredient Disclosure List.
US FEDERAL
TSCA
CAS# 88-40-4 is listed on the TSCA inventory.
SECTION 16 - ADDITIONAL INFORMATION
N/A
上下游信息
-
上游原料
中文名称 英文名称 CAS号 化学式 分子量 6-叔丁基间甲酚 2-tert-Butyl-5-methylphenol 88-60-8 C11H16O 164.247 -
下游产品
中文名称 英文名称 CAS号 化学式 分子量 6-叔丁基间甲酚 2-tert-Butyl-5-methylphenol 88-60-8 C11H16O 164.247 —— 2,4-di-tert-butyl-5-methyl-anisole 874521-29-6 C16H26O 234.382 —— 2-Brom-4-tert.-butyl-5-methoxy-toluol 26822-00-4 C12H17BrO 257.17 3-甲氧基-4-叔丁基苯甲酸 4-tert-butyl-3-methoxybenzoic acid 79822-46-1 C12H16O3 208.257 —— 2-iodo-5-methoxy-4-t-butyl-toluene 113137-99-8 C12H17IO 304.171 —— 2-Methyl-5-tert.-butyl-hydrochinon 2349-76-0 C11H16O2 180.247 —— 5-tert-butyl-4-methoxy-2-methyl-benzaldehyde 873990-70-6 C13H18O2 206.285 —— 2-tert-butyl-4-chloromethyl-5-methyl-anisole 26821-98-7 C13H19ClO 226.746 1-叔丁基-2-甲氧基-4-甲基-5-硝基苯 2-tert-butyl-5-methyl-4-nitro-anisole 71850-79-8 C12H17NO3 223.272
反应信息
-
作为反应物:描述:2-叔丁基-5-甲基苯甲醚 在 boron triiodide-N,N-diethylaniline 作用下, 以 苯 为溶剂, 反应 3.0h, 以71%的产率得到6-叔丁基间甲酚参考文献:名称:使用三碘化硼-N,N-二乙基苯胺络合物裂解醚和双乙酸双酯摘要:由硼烷:N,N-二乙基苯胺和碘生成的三碘化硼-N,N-二乙基苯胺络合物在温和的条件下以适当的收率裂解醚并从相应的双乙酸双酯衍生物中再生羧醛。DOI:10.1016/s0040-4039(00)97220-4
-
作为产物:描述:间甲酚 在 aluminum (III) chloride 、 苄基三甲基氯化铵 、 sodium hydroxide 作用下, 以 二氯甲烷 、 水 为溶剂, 反应 6.0h, 生成 2-叔丁基-5-甲基苯甲醚参考文献:名称:一种葵子麝香的合成方法摘要:本发明提供了一种葵子麝香的合成方法,该合成方法包括如下步骤:(1)将间甲酚、相转移催化剂、强碱的水溶液、氯甲烷进行混合,反应后生成间甲基苯甲醚;(2)将催化剂与溶剂进行混合,在其中滴加所述的间甲基苯甲醚,滴加0.5‑2小时,然后滴加叔丁基氯,滴加0.5‑2小时,滴加完成后,反应3‑5小时后生成对叔丁基甲氧基甲苯。本发明所述的葵子麝香的合成方法将间甲酚与氯甲烷在强碱的水溶液中合成间甲基甲醚(碱水溶液补加氢氧化钠过滤出氯化钠后循环套用),相对于传统的生产工艺,即提高了收率又减少废水的处理,节省了人力资源,减少能耗,更加安全可靠。公开号:CN107417537A
文献信息
-
PYRROLOPYRAZINE-SPIROCYCLIC PIPERIDINE AMIDES AS MODULATORS OF ION CHANNELS申请人:Hadida Ruah Sara Sabina公开号:US20120196869A1公开(公告)日:2012-08-02The invention relates to pyrrolopyrazine-spirocyclic piperidine amide compounds useful as inhibitors of ion channels. The invention also provides pharmaceutically acceptable compositions comprising the compounds of the invention and methods of using the compositions in the treatment of various disorders.
-
[EN] PYRAN-SPIROCYCLIC PIPERIDINE AMIDES AS MODULATORS OF ION CHANNELS<br/>[FR] AMIDES DE PIPÉRIDINE SPIROCYCLIQUES PYRANES UTILISÉS EN TANT QUE MODULATEURS DE CANAUX IONIQUES申请人:VERTEX PHARMA公开号:WO2013109521A1公开(公告)日:2013-07-25The invention relates to pyran spirocyclic piperidine amide compounds useful as inhibitors of ion channels. The invention also provides pharmaceutically acceptable compositions comprising the compounds of the invention and methods of using the compositions in the treatment of various disorders.
-
QUINOLONE COMPOUND申请人:OTSUKA PHARMACEUTICAL CO., LTD.公开号:US20140179675A1公开(公告)日:2014-06-26The present invention provides a compound represented by the formula (I) wherein X is a hydrogen atom or a fluorine atom; R is a hydrogen atom or alkyl; R 1 is (1) cyclopropyl optionally substituted by 1 to 3 halogen atoms or (2) phenyl optionally substituted by 1 to 3 halogen atoms; R 2 is alkyl, alkoxy, haloalkoxy, a halogen atom, cyano, etc.; and R 3 is 7-oxo-7,8-dihydro-1,8-naphthyridinyl, 3-pyridyl, etc., or a salt thereof. The compound of the present invention has excellent antimicrobial activity against Clostridium difficile and is useful for the prevention or treatment of intestinal infection such as Clostridium difficile -associated diarrhea.
-
HETEROARYLS AND USES THEREOF申请人:MILLENNIUM PHARMACEUTICALS, INC.公开号:US20150225422A1公开(公告)日:2015-08-13The present invention provides a compound of formula I: and pharmaceutically acceptable salts thereof, wherein X, R 1 , R 2 , R 3 , R 4 , R 5 , L 1 , L 2 , m, and n, are as described in the specification. Such compounds are inhibitors of VPS34 and thus useful for treating proliferative, inflammatory, or cardiovascular disorders.
-
A resin-promoted Williamson's alkyl aryl ether synthesis. Methylation of 2-tert-butyl-5-methylphenol with methyl chloride.作者:YASUMASA HAMADA、NOBUHARU KATO、YUKO KAKAMU、TAKAYUKI SHIOIRIDOI:10.1248/cpb.29.2246日期:——The use of easily recoverable tri-n-butylphosphoniummethylated polystyrene resin (phosphonium resin catalyst) under triphasic reaction conditions was proposed in Williamson's alkyl aryl ether synthesis. O-Methylation of 2-tert-butyl-5-methylphenol with methyl chloride was easily carried out in the presence of the phosphonium resin catalyst under basic conditions to give 2-tert-butyl-5-methylphenyl methyl ether in excellent yield. A few other phenols were also O-alkylated under similar triphasic reaction conditions to give alkyl aryl ethers.
表征谱图
-
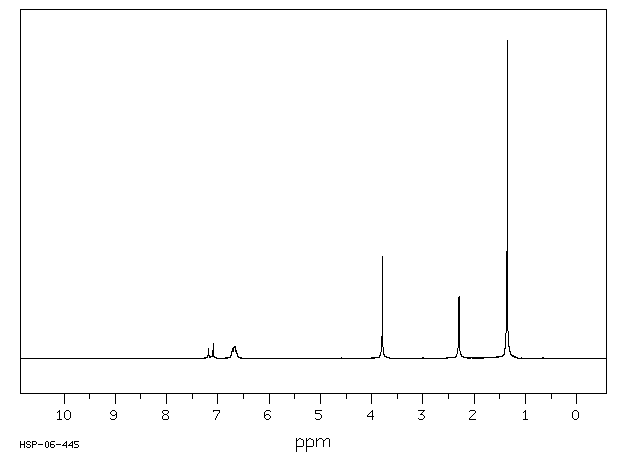
氢谱1HNMR
-
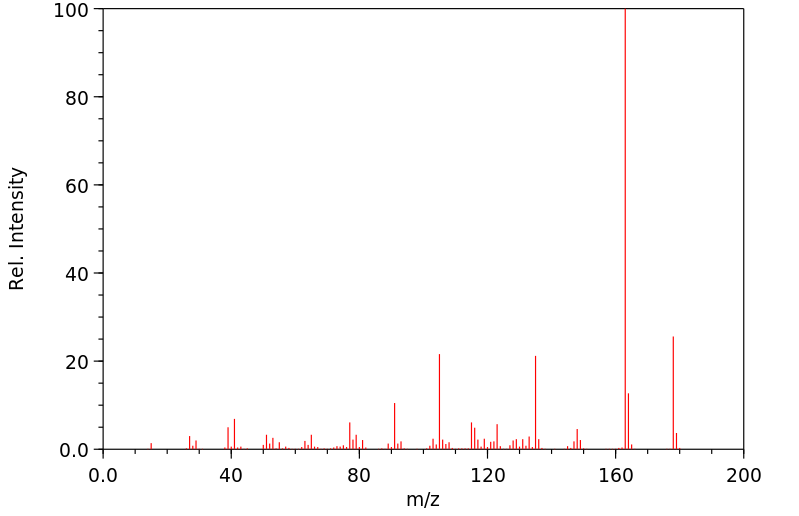
质谱MS
-
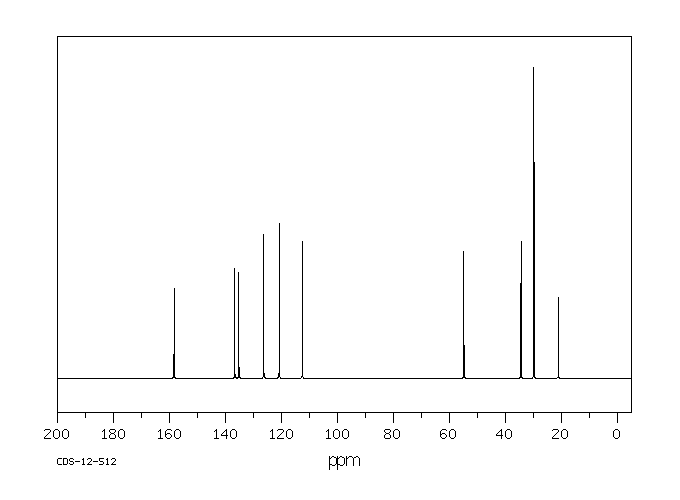
碳谱13CNMR
-
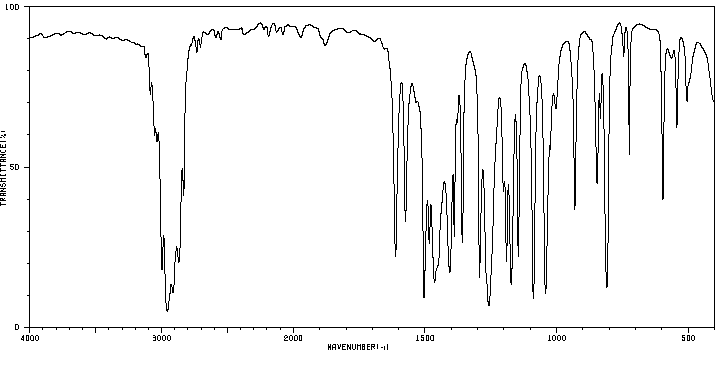
红外IR
-
拉曼Raman
-
峰位数据
-
峰位匹配
-
表征信息
同类化合物
(βS)-β-氨基-4-(4-羟基苯氧基)-3,5-二碘苯甲丙醇
(S,S)-邻甲苯基-DIPAMP
(S)-(-)-7'-〔4(S)-(苄基)恶唑-2-基]-7-二(3,5-二-叔丁基苯基)膦基-2,2',3,3'-四氢-1,1-螺二氢茚
(S)-盐酸沙丁胺醇
(S)-3-(叔丁基)-4-(2,6-二甲氧基苯基)-2,3-二氢苯并[d][1,3]氧磷杂环戊二烯
(S)-2,2'-双[双(3,5-三氟甲基苯基)膦基]-4,4',6,6'-四甲氧基联苯
(S)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(R)富马酸托特罗定
(R)-(-)-盐酸尼古地平
(R)-(-)-4,12-双(二苯基膦基)[2.2]对环芳烷(1,5环辛二烯)铑(I)四氟硼酸盐
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[((6-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(4-叔丁基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-7-双(3,5-二叔丁基苯基)膦基7''-[(3-甲基吡啶-2-基甲基)氨基]-2,2'',3,3''-四氢-1,1''-螺双茚满
(R)-(+)-4,7-双(3,5-二-叔丁基苯基)膦基-7“-[(吡啶-2-基甲基)氨基]-2,2”,3,3'-四氢1,1'-螺二茚满
(R)-3-(叔丁基)-4-(2,6-二苯氧基苯基)-2,3-二氢苯并[d][1,3]氧杂磷杂环戊烯
(R)-2-[((二苯基膦基)甲基]吡咯烷
(R)-1-[3,5-双(三氟甲基)苯基]-3-[1-(二甲基氨基)-3-甲基丁烷-2-基]硫脲
(N-(4-甲氧基苯基)-N-甲基-3-(1-哌啶基)丙-2-烯酰胺)
(5-溴-2-羟基苯基)-4-氯苯甲酮
(5-溴-2-氯苯基)(4-羟基苯基)甲酮
(5-氧代-3-苯基-2,5-二氢-1,2,3,4-oxatriazol-3-鎓)
(4S,5R)-4-甲基-5-苯基-1,2,3-氧代噻唑烷-2,2-二氧化物-3-羧酸叔丁酯
(4S,4''S)-2,2''-亚环戊基双[4,5-二氢-4-(苯甲基)恶唑]
(4-溴苯基)-[2-氟-4-[6-[甲基(丙-2-烯基)氨基]己氧基]苯基]甲酮
(4-丁氧基苯甲基)三苯基溴化磷
(3aR,8aR)-(-)-4,4,8,8-四(3,5-二甲基苯基)四氢-2,2-二甲基-6-苯基-1,3-二氧戊环[4,5-e]二恶唑磷
(3aR,6aS)-5-氧代六氢环戊基[c]吡咯-2(1H)-羧酸酯
(2Z)-3-[[(4-氯苯基)氨基]-2-氰基丙烯酸乙酯
(2S,3S,5S)-5-(叔丁氧基甲酰氨基)-2-(N-5-噻唑基-甲氧羰基)氨基-1,6-二苯基-3-羟基己烷
(2S,2''S,3S,3''S)-3,3''-二叔丁基-4,4''-双(2,6-二甲氧基苯基)-2,2'',3,3''-四氢-2,2''-联苯并[d][1,3]氧杂磷杂戊环
(2S)-(-)-2-{[[[[3,5-双(氟代甲基)苯基]氨基]硫代甲基]氨基}-N-(二苯基甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[((1S,2S)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2S)-2-[[[[[[((1R,2R)-2-氨基环己基]氨基]硫代甲基]氨基]-N-(二苯甲基)-N,3,3-三甲基丁酰胺
(2-硝基苯基)磷酸三酰胺
(2,6-二氯苯基)乙酰氯
(2,3-二甲氧基-5-甲基苯基)硼酸
(1S,2S,3S,5S)-5-叠氮基-3-(苯基甲氧基)-2-[(苯基甲氧基)甲基]环戊醇
(1S,2S,3R,5R)-2-(苄氧基)甲基-6-氧杂双环[3.1.0]己-3-醇
(1-(4-氟苯基)环丙基)甲胺盐酸盐
(1-(3-溴苯基)环丁基)甲胺盐酸盐
(1-(2-氯苯基)环丁基)甲胺盐酸盐
(1-(2-氟苯基)环丙基)甲胺盐酸盐
(1-(2,6-二氟苯基)环丙基)甲胺盐酸盐
(-)-去甲基西布曲明
龙蒿油
龙胆酸钠
龙胆酸叔丁酯
龙胆酸
龙胆紫-d6
龙胆紫